Abstract
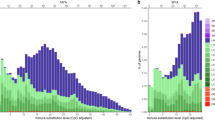
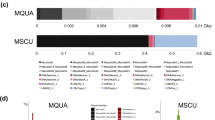
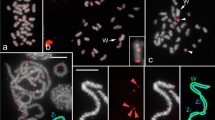
Over the past decades, transposable elements (TEs) have been shown to play important roles shaping genome architecture and as major promoters of genetic diversification and evolution of species. Likewise, TE accumulation is tightly linked to heterochromatinization and centromeric dynamics, which can ultimately contribute to speciation. Despite growing efforts to characterize the repeat landscape of species, few studies have focused on mapping the accumulation profiles of TEs on chromosomes. The few studies on repeat accumulation profiles in populations are biased towards model organisms and inbred lineages. Here, we present a cytomolecular analysis of six mobilome-extracted elements on multiple individuals from a population of a species of wild-captured beetle, Dichotomius schiffleri, aiming to investigate patterns of TE accumulation and uncover possible trends of their chromosomal distribution. Compiling TE distribution data from several individuals allowed us to make generalizations regarding variation of TEs at the gross chromosome level unlikely to have been achieved using a single individual, or even from a whole-genome assembly. We found that (1) transposable elements have differential accumulation profiles on D. schiffleri chromosomes and (2) specific chromosomes have their own TE accumulation landscape. The remarkable variability of their genomic distribution suggests that TEs are likely candidates to contribute to the evolution of heterochromatin architecture and promote high genetic variability in species that otherwise display conserved karyotypes. Therefore, this variation likely contributed to genome evolution and species diversification in Dichotomius.



Similar content being viewed by others
Data availability
DNA sequences of the clones used in this study are deposited in GenBank (https://www.ncbi.nlm.nih.gov/nuccore/)
Abbreviations
- TEs:
-
Transposable elements
- TP:
-
Transposase region
- RT:
-
Reverse transcriptase
- FISH:
-
Fluorescence in situ hybridization
- Xyr :
-
“Rod” configuration of sex chromosomes
- IBAMA :
-
Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (Brazilian Institute of Environment and Renewable Natural Resources)
- SISBIO :
-
Sistema de Autorização e Informação em Biodiversidade (Biodiversity Authorization and Information System)
- CAPES:
-
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (Brazilian Funding Organization - Coordination for the Improvement of Higher Education Personnel)
- PNPD:
-
National Postdoctoral Program
- FACEPE:
-
Fundação de Amparo a Ciência e Tecnologia do Estado de Pernambuco (Brazilian Funding Organization - Science and Technology Foundation of the State of Pernambuco)
- CNPq:
-
Conselho Nacional de Desenvolvimento Científico e Tecnológico (Brazilian Funding Organization - National Council for Scientific and Technological Development)
- LBGI :
-
Laboratório de Biodiversidade e Genética de Insetos (Biodiversity and Insect Genetics Laboratory)
References
Amorim IC, Costa RGC, Xavier C, Moura RC (2018) Characterization and chromosomal mapping of the DgmarMITE transposon in populations of Dichotomius (Luederwaldtinia) sericeus species complex (Coleoptera: Scarabaeidae). Genet Mol Biol 41:419–425. https://doi.org/10.1590/1678-4685-gmb-2017-0230
Amorim IC, Melo ES, Moura RC, Wallau GL (2020) Diverse mobilome of Dichotomius (Luederwaldtinia) schiffleri (Coleoptera: Scarabaeidae) reveals long-range horizontal transfer events of DNA transposons. Mol Gen Genomics 295:1339–1353. https://doi.org/10.1007/s00438-020-01703-8
Barrón MG, Fiston-Lavier A-S, Petrov DA, González J (2014) Population Genomics of Transposable Elements in Drosophila. Annu Rev Genet 48:561–581. https://doi.org/10.1146/annurev-genet-120213-092359
Bartolomé C, Maside X (2004) The lack of recombination drives the fixation transposable elements on the fourth chromosome of Drosophila melanogaster. Genet Res 83:91–100. https://doi.org/10.1017/S0016672304006755
Blumenstiel JP (2019) Birth, school, work, death, and resurrection: the life stages and dynamics of transposable element proliferation. Genes 10:336. https://doi.org/10.3390/genes10050336
Bourgeois Y, Boissinot S (2019) On the population dynamics of junk: a review on the population genomics of transposable elements. Genes 10:419. https://doi.org/10.3390/genes10060419
Brandes A, Heslop-Harrison JS, Kamm A, Kubis S, Doudrick RL, Schmidt T (1997) Comparative analysis of the chromosomal and genomic organization of Ty1-copia-like retrotransposons in pteridophytes, gymnosperms and angiosperms. Plant Mol Biol 33:11–21. https://doi.org/10.1023/A:1005797222148
Brown JD, O’Neill RJ (2010) Chromosomes, conflict, and epigenetics: chromosomal speciation revisited. Annu Rev Genomics Hum Genet 11:291–316. https://doi.org/10.1146/annurev-genom-082509-141554
Cabral-de-Mello DC (2015) Beetles (Coleoptera). In: Sharakhov I v (ed) Protocols for cytogenetic mapping of arthropod genomes. CRC Press Taylor & Fracis Group, Boca Raton
Cabral-de-Mello DC, Moura RC, Martins C (2010) Chromosomal mapping of repetitive DNAs in the beetle Dichotomius geminatus provides the first evidence for an association of 5S rRNA and histone H3 genes in insects, and repetitive DNA similarity between the B chromosome and A complement. Heredity 104:393–400. https://doi.org/10.1038/hdy.2009.126
Cabral-de-Mello DC, Moura RC, Melo AS, Martins C (2011a) Evolutionary dynamics of heterochromatin in the genome of Dichotomius beetles based on chromosomal analysis. Genetica 139:315–325. https://doi.org/10.1007/s10709-011-9551-7
Cabral-de-Mello DC, Moura RC, Martins C (2011b) Cytogenetic mapping of rRNAs and histone H3 genes in 14 species of Dichotomius (Coleoptera, Scarabaeidae, Scarabaeinae) beetles. Cytogenet Genome Res 134:127–135. https://doi.org/10.1159/000326803
Cáceres M, Puig M, Ruiz A (2001) Molecular Characterization of Two Natural Hotspots in the Drosophila buzzatii Genome Induced by Transposon Insertions. Genome Res 11:1353–1364. https://doi.org/10.1101/gr.174001
Casacuberta E (2017) Drosophila: retrotransposons making up telomeres. Viruses 9(7):192. https://doi.org/10.3390/v9070192
Chalopin D, Naville M, Plard F, Galiana D, Volff JN (2015) Comparative analysis of transposable elements highlights mobilome diversity and evolution in vertebrates. Genome Biol Evol 7:567–580. https://doi.org/10.1093/gbe/evv005
Chang CH, Chavan A, Palladino J, Wei X, Martins NMC, Santinello B, Chen CC, Erceg J, Beliveau BJ, Wu CT, Larracuente AM, Mellone BG (2019) Islands of retroelements are major components of Drosophila centromeres. PLoS Biol 17(5):e3000241. https://doi.org/10.1371/journal.pbio.3000241
Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95:118–128. https://doi.org/10.1038/sj.hdy.6800697
Crespi B, Nosil P (2013) Conflictual speciation: species formation via genomic conflict. Trends Ecol Evol 28:48–57. https://doi.org/10.1016/j.tree.2012.08.015
Deniz Ö, Frost JM, Branco MR (2019) Regulation of transposable elements by DNA modifications. Nat Rev Genet 20(7):417–431. https://doi.org/10.1038/s41576-019-0106-6
Dimitri P (1997) Constitutive heterochromatin and transposable elements in Drosophila melanogaster. Genetica 100:85–93
Dimitri P, Junakovic N (1999) Revising the selfish DNA hypothesis new evidence on accumulation of transposable elements in heterochromatin the bulk of the eukaryotic genome is composed of families of repetitive sequences that are genetically silent. Tig 15:123–124
Eichler EE, Sankoff D (2003) Structural dynamics of eukaryotic chromosome evolution. Science 301:793–797. https://doi.org/10.1126/science.1086132
Elliott TA, Gregory TR (2015) Do larger genomes contain more diverse transposable elements? BMC Evol Biol 15:1–10. https://doi.org/10.1186/s12862-015-0339-8
Feschotte C, Pritham EJ (2007) DNA Transposons and the evolution of eukaryotic genomes. Annu Rev Genet 41:331–368. https://doi.org/10.1146/annurev.genet.40.110405.090448
Fujiwara H, Osanai M, Matsumoto T, Kojima KK (2005) Telomere-specific non-LTR retrotransposons and telomere maintenance in the silkworm, Bombyx mori. Chromosom Res 13:455–467. https://doi.org/10.1007/s10577-005-0990-9
Gilbert C, Peccoud J, Cordaux R (2021) Transposable elements and the evolution of insects. Annu Rev Entomol 66:355–372. https://doi.org/10.1146/annurev-ento-070720-074650
Gorinšek B, Gubensek F, Kordis D (2004) Evolutionary genomics of chromoviruses in eukaryotes. Mol Biol Evol 21(5):781–798. https://doi.org/10.1093/molbev/msh057
Grzˇan T, Despot-Slade E, Mestrović N, Plohl M, Mravinac B (2020) CenH3 distribution reveals extended centromeres in the model beetle Tribolium castaneum. PLoS Genet 16(10):e1009115. https://doi.org/10.1371/journal.pgen.1009115
Harvey-Clark C (2011) IACUC challenges in invertebrate research. ILAR J 52:213–220. https://doi.org/10.1093/ilar.52.2.213
Hernandez-Hernandez EM, Fernández-Medina RD, Navarro-Escalante L, Nuñez J, Benavides-Machado P, Carareto CM (2017) Genome-wide analysis of transposable elements in the coffee berry borer Hypothenemus hampei (Coleoptera: Curculionidae): description of novel families. Mol Gen Genomics 292(3):565–583. https://doi.org/10.1007/s00438-017-1291-7
Jensen-Seaman MI, Furey TS, Payseur BA, Lu Y, Roskin KM, Chen CF, Chen CF, Thomas MA, Haussler D, Jacob HJ (2004) Comparative recombination rates in the rat, mouse, and human genomes. Genome Res 14(4):528–538. https://doi.org/10.1101/gr.1970304
Junakovic N, Terrinoni A, Di Franco C et al (1998) Accumulation of transposable elements in the heterochromatin and on the Y chromosome of Drosophila simulans and Drosophila melanogaster. J Mol Evol 46:661–668. https://doi.org/10.1007/PL00006346
Kaback DB, Barber D, Mahon J, Lamb J, You J (1999) Chromosome size-dependent control of meiotic reciprocal recombination in Saccharomyces cerevisiae: the role of crossover interference. Genetics 152(4):1475–1486
Katoh K, Rozewicki J, Yamada KD (2018) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1166. https://doi.org/10.1093/bib/bbx108
Kent TV, Uzunović J, Wright SI (2017) Coevolution between transposable elements and recombination. Philo Transac Royal Soc B: Biol Sci 372(1736):20160458. https://doi.org/10.1098/rstb.2016.0458
Kidwell MG, Lisch DR (2001) Perspective: transposable elements, parasitic Dna, and genome evolution. Evolution 55:1–24. https://doi.org/10.1111/j.0014-3820.2001.tb01268.x
Klein SJ, O’Neill RJ (2018) Transposable elements: genome innovation, chromosome diversity, and centromere conflict. Chromosom Res 26:5–23. https://doi.org/10.1007/s10577-017-9569-5
Kubo Y, Okazaki S, Anzai T, Fujiwara H (2001) Structural and phylogenetic analysis of TRAS, telomeric repeat-specific non-LTR retrotransposon families in Lepidopteran insects. Mol Biol Evol 18:848–857. https://doi.org/10.1093/oxfordjournals.molbev.a003866
Levis RW, Ganesan R, Houtchens K, Tolar LA, Sheen FM (1993) Transposons in place of telomeric repeats at a Drosophila telomere. Cell 75:1083–1093. https://doi.org/10.1016/0092-8674(93)90318-K
Llorens C, Futami R, Covelli L, Domínguez-Escriba L, Viu JM, Tamarit D et al (2011) The Gypsy Database (GyDB) of mobile genetic elements: release 2.0. Nucleic Acids Res 39:D70–D74. https://doi.org/10.1093/nar/gkq1061
Maumus F, Fiston-Lavier AS, Quesneville H (2015) Impact of transposable elements on insect genomes and biology. Curr Opin Insect Sci 7:30–36. https://doi.org/10.1016/j.cois.2015.01.001
Montgomery EA, Huang SM, Langley CH, Judd BH (1991) Chromosome rearrangement by ectopic recombination in Drosophila melanogaster: genome structure and evolution. Genetics 129:1085–1098
Montiel EE, Cabrero J, Camacho JPM, López-León MD (2012) Gypsy, RTE and Mariner transposable elements populate Eyprepocnemis plorans genome. Genetica 140:365–374. https://doi.org/10.1007/s10709-012-9686-1
Neumann P, Navrátilová A, Koblížková A, Kejnovský E, Hřibová E, Hobza R, Widmer A, Doležel J, Macas J (2011) Plant centromeric retrotransposons: a structural and cytogenetic perspective. Mob DNA 2(1):4. https://doi.org/10.1186/1759-8753-2-4
Nunes RV, Vaz-de-Mello FZ (2019) Taxonomic revision of Dichotomius (Cephagonus) Luederwaldt 1929 and the taxonomic status of remaining Dichotomius Hope 1838 subgenera (Coleoptera: Scarabaeidae: Scarabaeinae: Dichotomiini). J Nat Hist 53:2231–2351. https://doi.org/10.1080/00222933.2019.1692088
O’Neill MJ, O’Neill RJ (2018) Sex chromosome repeats tip the balance towards speciation. Mol Ecol 27:3783–3798. https://doi.org/10.1111/mec.14577
Oliveira SG, Moura RC, Martins C (2012) B chromosome in the beetle Coprophanaeus cyanescens (Scarabaeidae): emphasis in the organization of repetitive DNA sequences. BMC Genet 13:1. https://doi.org/10.1186/1471-2156-13-96
Oliveira SG, Cabral-de-Mello DC, Moura RC, Martins C (2013) Chromosomal organization and evolutionary history of Mariner transposable elements in Scarabaeinae coleopterans. Mol Cytogenet 6:54. https://doi.org/10.1186/1755-8166-6-54
Palacios-Gimenez OM, Bueno D, Cabral-de-Mello DC (2014) Chromosomal mapping of two Mariner-like elements in the grasshopper Abracris flavolineata (Orthoptera: Acrididae) reveals enrichment in euchromatin. Eur J Entomol 111(3):329–334. https://doi.org/10.14411/eje.2014.052
Petersen M, Armisén D, Gibbs RA, Hering L, Khila A, Mayer G, Richards S, Niehuis O, Misof B (2019) Diversity and evolution of the transposable element repertoire in arthropods with particular reference to insects. BMC Evol Biol 19:1–15. https://doi.org/10.1186/s12862-018-1324-9
Plohl M, Luchetti A, Meštrović N, Mantovani B (2008) Satellite DNAs between selfishness and functionality: structure, genomics and evolution of tandem repeats in centromeric (hetero) chromatin. Gene 409:72–82. https://doi.org/10.1016/j.gene.2007.11.013
Presting GG (2018) Centromeric retrotransposons and centromere function. Curr Opin Genet Dev 49:79–84. https://doi.org/10.1016/j.gde.2018.03.004
Pritham EJ, Putliwala T, Feschotte C (2007) Mavericks, a novel class of giant transposable elements widespread in eukaryotes and related to DNA viruses. Gene 390:3–17. https://doi.org/10.1016/j.gene.2006.08.008
Quesneville H, Nouaud D, Anxolabéhère D (2006) P elements and MITE relatives in the whole genome sequence of Anopheles gambiae. BMC Genomics 7:214. https://doi.org/10.1186/1471-2164-7-214
Rahman R, Chirn GW, Kanodia A, Sytnikova YA, Brembs B, Bergman CM, Lau NC (2015) Unique transposon landscapes are pervasive across Drosophila melanogaster genomes. Nucleic Acids Res 43:10655–10672. https://doi.org/10.1093/nar/gkv1193
Ray DA, Grimshaw JR, Halsey MK, Korstian JM, Osmanski AB, Sullivan KAM, Wolf KA, Reddy H, Foley N, Stevens RD, Knisbacher BA, Levy O, Counterman B, Edelman NB, Mallet J (2019) Simultaneous TE analysis of 19 heliconiine butterflies yields novel insights into rapid TE-based genome diversification and multiple SINE births and deaths. Genome Biol Evol 11:2162–2177. https://doi.org/10.1093/gbe/evz125
Rosolen LA, Vicari MR, Almeida MC (2018) Accumulation of transposable elements in autosomes and giant sex chromosomes of Omophoita (Chrysomelidae: Alticinae). Cytogen Genome Res 156(4):215–222. https://doi.org/10.1159/000495199
Sambrook J, Russel DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
Santini S, Cavallini A, Natali L, Minelli S, Maggini F, Cionini P (2002) Ty1/copia- and Ty3/gypsy-like DNA sequences in Helianthus species. Chromosoma 111:192–200. https://doi.org/10.1007/s00412-002-0196-2
Serrato-Capuchina A, Matute DR (2018) The role of transposable elements in speciation. Genes 9:254. https://doi.org/10.3390/genes9050254
Silva GMD, Bione EG, Cabral-de-Mello DC, Moura RC, Simões ZLP, Souza MJ (2009) Comparative cytogenetics of three species of Dichotomius (Coleoptera, Scarabaeidae). Genet Mol Biol 32(2):276–280. https://doi.org/10.1590/S1415-47572009005000040
Sotero-Caio CG, Platt RN, Suh A, Ray DA (2017) Evolution and diversity of transposable elements in vertebrate genomes. Genome Biol Evol 9:161–177. https://doi.org/10.1093/gbe/evw264
Termolino P, Cremona G, Consiglio MF, Conicella C (2016) Insights into epigenetic landscape of recombination-free regions. Chromosoma 125:301–308. https://doi.org/10.1007/s00412-016-0574-9
Underwood CJ, Choi K (2019) Heterogeneous transposable elements as silencers, enhancers and targets of meiotic recombination. Chromosoma 128:279–296. https://doi.org/10.1007/s00412-019-00718-4
Valois MC, Vaz-De-Mello FZ, Silva FAB (2017) Taxonomic revision of the Dichotomius sericeus (Harold, 1867) species group (Coleoptera: Scarabaeidae: Scarabaeinae). Zootaxa 4277:503–530. https://doi.org/10.11646/zootaxa.4277.4.3
Van-Lume B, Mata-Sucre Y, Báez M, Ribeiro T, Huettel B, Gagnon E, Leitch IJ, Pedrosa-Harand A, Lewis GP, Souza G (2019) Evolutionary convergence or homology? Comparative cytogenomics of Caesalpinia group species (Leguminosae) reveals diversification in the pericentromeric heterochromatic composition. Planta 250(6):2173–2186. https://doi.org/10.1007/s00425-019-03287-z
Wells JN, Feschotte C (2020) A field guide to eukaryotic transposable elements. Annu Rev Genet 23:539–561. https://doi.org/10.1146/annurev-genet-040620-022145
Wicker T, Sabot F, Hua-Van A et al (2007) A unified classification system for eukaryotic transposable elements. Nat Rev Genet 8:973–982. https://doi.org/10.1038/nrg2165
Xavier C, Cabral-de-Mello DC, Moura RC (2014) Heterochromatin and molecular characterization of DsmarMITE transposable element in the beetle Dichotomius schiffleri (Coleoptera: Scarabaeidae). Genetica 142:575–581. https://doi.org/10.1007/s10709-014-9805-2
Yoshida K, Ishikawa A, Toyoda A, Shigenobu S, Fujiyama A, Kitano J (2019) Functional divergence of a heterochromatin-binding protein during stickleback speciation. Mol Ecol 28:1563–1578. https://doi.org/10.1111/mec.14841
Zhang RG, Wang ZX, Ou S, Li GY (2019) TEsorter: lineage-level classification of transposable elements using conserved protein domains, vol 800177. bioRxiv, Cold Spring Harbor. https://doi.org/10.1101/800177
Code availability
Accession codes of GenBank: MT636862, MT636863, MT636866, MT636868, MT636869 and MT636871.
Funding
Costa RGCC was supported with a Master’s scholarship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES); Sotero-Caio CG and Amorim IC were supported with postdoctoral fellowships PNPD/CAPES, process numbers 88882.306325/2018-01 and 88882.317331/2019-01, respectively. This work was funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) with the grant PQ-2 to Moura RC, process number 305298/2014-3; and by Fundação de Amparo à Ciência e Tecnologia de Pernambuco (FACEPE), APQ process number 0777-2.02/15.
Author information
Authors and Affiliations
Contributions
ICA performed research, analyzed the data, produced the images, and helped writing of the manuscript; CGSC helped analyzing the data and wrote the paper; RGCC performed all FISH experiments, helped analyzing the data, and helped writing the first version of the manuscript; CX performed research, analyzed the data, and helped writing of the manuscript; RCM conceived and designed the study and analyzed the data and helped writing of the manuscript. All authors read and approve the manuscript submission
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Andreas Houben
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 813 kb)
Rights and permissions
About this article
Cite this article
Amorim, I.C., Sotero-Caio, C.G., Costa, R.G.C. et al. Comprehensive mapping of transposable elements reveals distinct patterns of element accumulation on chromosomes of wild beetles. Chromosome Res 29, 203–218 (2021). https://doi.org/10.1007/s10577-021-09655-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-021-09655-4