Abstract
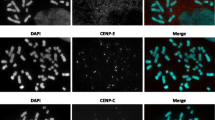
Twenty-five dicentric small supernumerary marker chromosomes (sSMC) derived from #13/21, #14, #15, #18, and #22 were studied by immunohistochemistry for their centromeric activity. Centromere protein (CENP)-B was applied as marker for all centromeres and CENP-C to label the active ones. Three different ‘predominant’ activation patterns could be observed, i.e., centric fusion or either only one or all two centromeres were active. In one inherited case, the same activation pattern was found in mother and son. In acrocentric-derived sSMC, all three activation patterns could be present. In contrary, in chromosome 18-derived sSMC, only the fusion type was observed. In concordance with previous studies a certain centromeric plasticity was observed in up to 13% of the cells of an individual case. Surprisingly, the obtained data suggests a possible influence of the sSMC carrier’s gender on the implementation of the predominant activation pattern; especially, only one active centromere was found more frequently in female than in male carriers. Also, it might be suggested that dicentric sSMC with one active centromere could be less stable than such with two active ones—centromeric plasticity might have an influence here, as well. Also, centromere activity in acrocentric-derived dicentrics could be influenced by heteromorphisms of the corresponding short arms. Finally, evidence is provided that the closer the centromeres of a dicentric are and if they are not fused, the more likely it was that both of them became active. In concordance and refinement with previous studies, a distance of 1.4 Mb up to about 13 Mb the two active centromere state was favored, while centromeric distance of over ∼15 Mb lead to inactivation of one centromere. Overall, here, the first and largest ever undertaken study in dicentric sSMC is presented, providing evidence that the centromeric activation pattern is, and parental origin may be of interest for their biology. Influence of mechanisms similar or identical to meiotic imprinting in the centromeric regions of human chromosomes might be present. Furthermore, centromeric activation pattern could be at least in parts meaningful for the clinical outcome of dicentric sSMC, as sSMC stability and mosaicism can make the difference between clinically normal and abnormal phenotypes.




Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- CENP:
-
Centromere protein
- MB:
-
Megabasepair
- sSMC:
-
Small supernumerary marker chromosome
References
Ando S, Yang H, Nozaki N, Okazaki T, Yoda K (2002) CENP-A, -B, and -C chromatin complex that contains the I-type alpha-satellite array constitutes the prekinetochore in HeLa cells. Mol Cell Biol 22:2229–2241
Bischoff FR, Maier G, Tilz G, Ponstingl H (1990) A 47-kDa human nuclear protein recognized by antikinetochore autoimmune sera is homologous with the protein encoded by RCC1, a gene implicated in onset of chromosome condensation. Proc Natl Acad Sci U S A 87:8617–8621
Bonati MT, Finelli P, Giardino D, Gottardi G, Roberts W, Larizza L (2005) Trisomy 15q25.2-qter in an autistic child: genotype-phenotype correlations. Am J Med Genet A 133:184–188
Cooke CA, Bernat RL, Earnshaw WC (1990) CENP-B: a major human centromere protein located beneath the kinetochore. J Cell Biol 110:1475–1488
Daniel A, Lam-Po-Tang PR (1976) Structure and inheritance of some heterozygous Robertsonian translocation in man. J Med Genet 13:381–388
Dewald GW, Boros SJ, Conroy MM, Dahl RJ, Spurbeck JL, Vitek HA (1979) A tdic(5;15)(p31;p11) chromosome showing variation for constriction in the centromeric regions in a patient with the cri du chat syndrome. Cytogenet Cell Genet 24:15–26
Earnshaw WC, Migeon BR (1985) Three related centromere proteins are absent from the inactive centromere of a stable isodicentric chromosome. Chromosoma 92:290–296
Earnshaw WC, 3rd Ratrie Ratrie, Stetten G (1989) Visualization of centromere proteins CENP-B and CENP-C on a stable dicentric chromosome in cytological spreads. Chromosoma 98:1–12
Friedrich U, Caprani M, Niebuhr E, Therkelsen AJ, Jørgensen AL (1996) Extreme variant of the short arm of chromosome 15. Hum Genet 97:710–713
Fukagawa T, Brown WR (1997) Efficient conditional mutation of the vertebrate CENP-C gene. Hum Mol Genet 6:2301–2308
Fukagawa T, Pendon C, Morris J, Brown W (1999) CENP-C is necessary but not sufficient to induce formation of a functional centromere. EMBO J 18:4196–4209
Guichet A, Boisseau P, Ingster O, Guardiola P, Couteleau A, Bonneau D (2009) Large inv dup(15) characterized by FISH and aCGH in a child with no abnormal phenotype at 2 years old. Chromosome Res 17(1):178, 11.4
Haaf T, Schmid M (1990) Y isochromosome associated with a mosaic karyotype and inactivation of the centromere. Hum Genet 85:486–490
Higgins AW, Gustashaw KM, Willard HF (2005) Engineered human dicentric chromosomes show centromere plasticity. Chromosome Res 13:745–762
Ing PS, Smith SD (1983) Cytogenetic studies of a patient with mosaicism of isochromosome 13q and a dicentric (Y;13) translocation showing differential centromeric activity. Clin Genet 24:194–199
Lau YF, Wertelecki W, Pfeiffer RA, Arrighi FE (1979) Cytological analyses of 14p+ variant by means of N-banding and combinations of silver staining and chromosome bandings. Hum Genet 46:75–82
Liehr T (2006) Familial small supernumerary marker chromosomes are predominantly inherited via the maternal line. Genet Med 8:459–462
Liehr T (2010) Small supernumerary marker chromosome homepage. Retrieved from http://www.med.uni-jena.de/fish/sSMC/00START.htm Accessed 9 April 2010
Liehr T, Weise A (2007) Frequency of small supernumerary marker chromosomes in prenatal, newborn, developmentally retarded and infertility diagnostics. Int J Mol Med 19:719–731
Liehr T, Claussen U, Starke H (2004) Small supernumerary marker chromosomes (sSMC) in humans. Cytogenet Genome Res 107:55–67
Liehr T, Mrasek K, Weise A, Dufke A, Rodríguez L, Martínez Guardia N, Sanchís A, Vermeesch JR, Ramel C, Polityko A, Haas OA, Anderson J, Claussen U, von Eggeling F, Starke H (2006) Small supernumerary marker chromosomes—progress towards a genotype-phenotype correlation. Cytogenet Genome Res 112:23–34
Loitzsch A, Bartsch O (2006) Healthy 12-year-old boy with mosaic inv dup(15)(q13). Am J Med Genet A 140:640–643
Masumoto H, Masukata H, Muro Y, Nozaki N, Okazaki T (1989) A human centromere antigen (CENP-B) interacts with a short specific sequence in alphoid DNA, a human centromeric satellite. J Cell Biol 109:1963–1973
Merry DE, Pathak S, Hsu TC, Brinkley BR (1985) Anti-kinetochore antibodies: use as probes for inactive centromeres. Am J Hum Genet 37:425–430
Moroi Y, Peebles C, Fritzler MJ, Steigerwald J, Tan EM (1980) Autoantibody to centromere (kinetochore) in scleroderma sera. Proc Natl Acad Sci U S A 77:1627–1631
Murmann AE, Conrad DF, Mashek H, Curtis CA, Nicolae RI, Ober C, Schwartz S (2009) Inverted duplications on acentric markers: mechanism of formation. Hum Mol Genet 18:2241–2256
Niebuhr E (1972) Dicentric and monocentric Robertsonian translocations in man. Humangenetik 16:217–226
Page SL, Shaffer LG (1998) Chromosome stability is maintained by short intercentromeric distance in functionally dicentric human Robertsonian translocations. Chromosome Res 6:115–122
Palmer DK, O'Day K, Trong HL, Charbonneau H, Margolis RL (1991) Purification of the centromere-specific protein CENP-A and demonstration that it is a distinctive histone. Proc Natl Acad Sci U S A 88:3734–3738
Peretti D, Maraschio P, Lambiase S, Lo Curto F, Zuffardi O (1986) Indirect immunofluorescence of inactive centromeres as indicator of centromeric function. Hum Genet 73:12–16
Reddy KS, Sulcova V (1998) The mobile nature of acrocentric elements illustrated by three unusual chromosome variants. Hum Genet 102:653–662
Rivera H, Zuffardi O, Maraschio P, Caiulo A, Anichini C, Scarinci R, Vivarelli R (1989) Alternate centromere inactivation in a pseudodicentric (15;20)(pter;pter) associated with a progressive neurological disorder. J Med Genet 26:626–630
Saitoh H, Tomkiel J, Cooke CA, 3rd Ratrie H, Maurer M, Rothfield NF, Earnshaw WC (1992) CENP-C an autoantigen in scleroderma, is a component of the human inner kinetochore plate. Cell 115:115–125
Schmid M, Nanda I, Steinlein C, Epplen JT (1994) Amplification of (GACA)n simple repeats in an exceptional 14p+ marker chromosome. Hum Genet 93:375–382
Sheth F, Ewers E, Kosyakova N, Weise A, Sheth J, Patil S, Ziegler M, Liehr T (2009) A neocentric isochromosome Yp present as additional small supernumerary marker chromosome–evidence against U-type exchange mechanism? Cytogenet Genome Res 125:115–116
Sullivan BA, Willard HF (1998) Stable dicentric X chromosomes with two functional centromeres. Nat Genet 20:227–228
Therman E, Trunca C, Kuhn EM, Sarto GE (1986) Dicentric chromosomes and the inactivation of the centromere. Hum Genet 72:191–195
Trifonov V, Seidel J, Starke H, Martina P, Beensen V, Ziegler M, Hartmann I, Heller A, Nietzel A, Claussen U, Liehr T (2003) Enlarged chromosome 13 p-arm hiding a cryptic partial trisomy 6p22.2-pter. Prenat Diagn 23:427–430
Verma RS, Kleyman SM, Conte RA (1996) Molecular characterization of an unusual variant of the short arm of chromosome 15 by FISH-technique. Jpn J Hum Genet 41:307–311
Wandall A (1989) Kinetochore development in two dicentric chromosomes in man. A light and electron microscopic study. Hum Genet 82:137–141
Wandall A (1994) A stable dicentric chromosome: both centromeres develop kinetochores and attach to the spindle in monocentric and dicentric configuration. Chromosoma 103:56–62
Acknowledgments
Supported in parts by the DAAD and the Prochance 2008, Jena.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans-Joachim Lipps.
Rights and permissions
About this article
Cite this article
Ewers, E., Yoda, K., Hamid, A.B. et al. Centromere activity in dicentric small supernumerary marker chromosomes. Chromosome Res 18, 555–562 (2010). https://doi.org/10.1007/s10577-010-9138-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-010-9138-7