Abstract
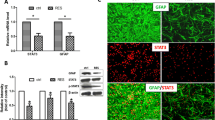
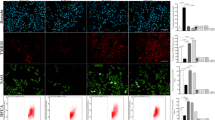
To study the inhibitory effect of Hepatocyte growth factor (HGF) on the responsive hyperplasia of damaged astrocytes in vitro. We prepared damaged model of astrocytes to simulate the responsive hyperplasia of damaged astrocytes in vivo by culturing astrocytes in vitro; After the first day of Ad-HGF transfection, astrocytes were scratched, then after the first, the third, and the fifth day of scratch, we detect the expression amount of astrocytes specific glial fibrillary acidic protein (GFAP) and the ratio of S-phase cells with flow cytometry, both of which can reflect the proliferation status of damaged astrocytes; After HGF was added in scratched astrocytes, the activity of SPK and MAPK (P42/44) were detected by autoradiography and immunoblotting test; After adding different concentrations of HGF protein in astrocytes cultured in different serum concentrations and adding diverse concentrations of HGF protein, SPK and SPK inhibitor DMS in scratched astrocytes, we detect cell proliferation with 3H-TDR incorporation. The first day after Ad-HGF transfected astrocytes were scratched, the amount of GFAP secreted by astrocytes were decreased significantly (P < 0.05), and the cells in S phase were declined obviously. HGF has bidirectional regulation on SPK of scratched astrocytes: increases the SPK activity when HGF in low dose, while inhibits when in high dose. In addition, DMS can block the signal passage; HGF had no effects on MAPK (P42/44) of damaged astrocytes cells. In conclusion, after the transfection of Ad-HGF, it can inhibit the responsive hyperplasia of damaged astrocytes by the means of blocking SPK passage.







Similar content being viewed by others
References
Abe K, Saito H (2000) The p44/42 mitogen-activated protein kinase cascade is involved in the induction and maintenance of astrocyte stellation mediated by protein kinase C. Neurosci Res 36(3):251–257
Chattopadhyay N, Tfelt-Hansen J, Brown EM (2002) PKC, p42/44 MAPK and p38 MAPK regulate hepatocyte growth factor secretion from human astrocytoma cells. Brain Res Mol Brain Res 102(1–2):73–82
Du F, Qian ZM, Zhu L, Wu XM et al (2010) Purity, cell viability, expression of GFAP and bystin in astrocytes cultured by different procedures. J Cell Biochem 109(1):30–37
Eng LF, Stocklin E, Lee YL et al (1986) Astrocyte culture on nitrocellulose membranes and plastic: detection of cytoskeletal proteins and mRNAs by immunocytochemistry and in situ hybridization. J Neurosci Res 16(1):239–250
Fitch MT, Silver J (2008) CNS injury, glial scars, and inflammation: inhibitory extracellular matrices and regeneration failure. Exp Neurol 209(2):294–301
Fitch MT, Doller C, Combs CK et al (1999) Cellular and molecular mechanisms of glial scarring and progressive cavitation: in vivo and in vitro analysis of inflammation-induced secondary injury after CNS trauma. J Neurosci 19(19):8182–8198
Gylys KH, Fein JA, Cole GM (2000) Quantitative characterization of crude synaptosomal fraction (P-2) components by flow cytometry. J Neurosci Res 61(2):186–192
Ha X, Li Y, Lao M et al (2003) Effect of human hepatocyte growth factor on promoting wound healing and preventing scar formation by adenovirus-mediated gene transfer. Chin Med J (Engl) 116(7):1029–1033
Jackson AB, Dijkers M, DeVivo MJ et al (2004) A demographic profile of new traumatic spinal cord injuries: change and stability over 30 years. Arch Phys Med Rehab 85(11):1740–1748
Kishimoto Y, Hirano S, Kitani Y et al (2010) Chronic vocal fold scar restoration with hepatocyte growth factor hydrogel. Laryngoscope 120(1):108–113
Kornyei Z, Czirok A, Vicsek T et al (2000) Proliferative and migratory responses of astrocytes to in vitro injury. J Neurosci Res 61(4):421–429
Liesi P, Kauppila T (2002) Induction of type IV collagen and other basement-membrane-associated proteins after spinal cord injury of the adult rat may participate in formation of the glial scar. Exp Neurol 173(1):31–45
Liu C, Que H, Shu C et al (2006) Effect of hepatocyte growth factor on the promotion of in vitro spinal cord neurite regeneration. J Clin Rehab 10(29):173–176
Naim R, Naumann A, Barnes J et al (2008) Transforming growth factor-beta1-antisense modulates the expression of hepatocyte growth factor/scatter factor in keloid fibroblast cell culture. Aesthet Plast Surg 32(2):346–352
Saunders MR, Fry EJ, Lane M (2002) Spinal cord injuries: making the re-connection. Biologist 49(3):107–112
Shimamura M, Sato N, Waguri S et al (2006) Gene transfer of hepatocyte growth factor gene improves learning and memory in the chronic stage of cerebral infarction. Hypertension 47(4):742–751
Shimazaki K, Yoshida K, Hirose Y et al (2003) Cytokines regulate c-Met expression in cultured astrocytes. Brain Res 962(1–2):105–110
Yu AC, Lee YL, Eng LF (1993) Astrogliosis in culture: I. The model and the effect of antisense oligonucleotides on glial fibrillary acidic protein synthesis. J Neurosci Res 34(3):295–303
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, C., Wu, Zz., Shu, Cl. et al. Experimental Investigation of HGF Inhibiting Glial Scar In Vitro. Cell Mol Neurobiol 31, 259–268 (2011). https://doi.org/10.1007/s10571-010-9616-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-010-9616-7