Abstract
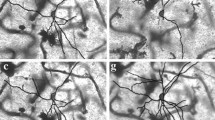
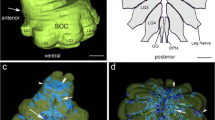
Intracellular recordings were obtained from brain slice preparation in neurons of the striatum of the turtle Trachemys scripta elegans, analogous to the mammalian striatum in its topographic organization, synaptic connectivity, cytoarchitecture, and neurochemistry. Here we show that these similarities extend to the electrophysiological properties of its neurons. Biocytin staining revealed that 85% of the recorded neurons were medium spiny neurons while 15% were aspiny neurons. Spiny neurons of the turtle resembled those found in the mammalian and avian striatum and express dopaminergic D1 and D2 class receptors. Because the striatum of the turtle receives a dense dopaminergic innervation from tegmental dopaminergic neurons we investigated the postsynaptic actions of selective dopamine receptor agonists in the excitability of spiny neurons. As in mammals and birds, activation of D1-receptors enhances, whereas activation of D2-receptors decreases the evoked discharge. Apparently, actions of dopamine agonists occur via the modulation of L-type (CaV1) Ca2+-conductances. Strong cellular evidence suggests that the role of dopamine in the modulation of motor networks is preserved along vertebrate evolution.




Similar content being viewed by others
References
Andersen A, Baestrup C, Randrup A (1975) Apomorphine-induced stereotyped biting in the tortoise in relation to dopaminergic mechanisms. Brain Behav Evol 11:365–373
Bargas J, Galarraga E, Aceves J (1989) An early outward conductance modulates the firing latency and frequency of neostriatal neurons of the rat brain. Exp Brain Res 75:146–156
Brauth SE, Reiner A, Kitt CA, Karten HJ (1983) The substance P-containing striato-tegmental path in reptiles: an immunohistochemical study. J Comp Neurol 219:305–327
Butler A, Hodos W (2005). Comparative vertebrate neuroanatomy, evolution and adaptation, 2nd edn. Wiley Interscience, p 484
Carrillo-Reid L, Tecuapetla F, Tapia D, Hernandez-Cruz A, Galarraga E, Drucker-Colin R, Bargas J (2008) Encoding network states by striatal cell assemblies. J Neurophysiol 99:1435–1450
Cepeda C, Colwell CS, Itri JN, Chandler SH, Levine MS (1998) Dopaminergic modulation of NMDA-induced whole cell currents in neostriatal neurons in slices: contribution of calcium conductances. J Neurophysiol 79:82–94
Delgado-Lezama R, Perrier JF, Hounsgaard J (1999) Oscillatory interaction between dorsal root excitability and dorsal root potentials in the spinal cord of the turtle. Neurosci 93:731–739
Ding L, Perkel DJ (2002) Dopamine modulates excitability of spiny neurons in the avian basal ganglia. J Neurosci 22:5210–5218
Donkelaar HJT (1998) Reptiles. In: Nieuwenhuys R, Donkeelar HJT, Nicholson C (eds) Central nervous system of vertebrates. Springer-Verlag, Berlin, pp 1315–1524
Farries MA, Perkel DJ (2000) Electrophysiological properties of avian basal ganglia neurons recorded in vitro. J Neurophysiol 84:2502–2513
Farries MA, Perkel DJ (2002) A telencephalic nucleus essential for song learning contains neurons with physiological characteristics of both Striatum and Globus Pallidus. J Neurosci 22:3776–3787
Farries MA, Meitzen J, Perkel DJ (2005) Electrophysiological properties of neurons in the basal ganglia of the domestic chick: conservation and divergence in the evolution of the avian basal ganglia. J Neurophysiol 94:454–467
Fowler M, Medina L, Reiner A (1999) Inmunohistochemical localization of NMDA- and AMPA-type glutamate receptor subunits in the basal ganglia of red eared turtles. Brain Behav Evol 54:276–289
Galarraga E, Pacheco-Cano MT, Flores-Hernández JV, Bargas J (1994) Subthreshold rectification in neostriatal spiny projection neurons. Exp Brain Res 100:239–249
Galarraga E, Hernández-López S, Reyes A, Barral J, Bargas J (1997) Dopamine facilitates EPSPs through an L-type Ca2+-conductance. Neuroreport 8:2183–2186
González A, Smeets WJAJ (1994) Catecholamine systems in the CNS of amphibians. In: Smeets WJAJ, Reiner A (eds) Phylogeny and development of catecholamine systems in the CNS of vertebrates. Cambridge University Press, Cambridge, pp 77–102
González-Burgos G, Kröner S, Krimer LS, Seamans JK, Urban NN, Henze DA, Lewis DA, Barrionuevo G (2002) Dopamine modulation of neuronal function in the monkey prefrontal cortex. Physiol Behav 77:537–543
Graybiel AM (1990) The basal ganglia and the initiation of the movement. Rev Neurol 146:570–574
Greenberg N, Font E, Switzer RC (1988) The reptilian striatum revisited: studies on Anolis lizards. In: Schwerdtfeger WK, Smeets WJAJ (eds) The forebrain of reptiles: current concepts of structure, function. Karger, Basel, pp 162–177
Grillner S (2006) Biological pattern generation: the cellular and computational logic of networks in motion. Neuron 52:751–766
Guzmán JN, Hernandez A, Galarraga E, Tapia D, Laville A, Vergara R, Aceves J, Bargas J (2003) Dopaminergic modulation of axon collaterals interconnecting spiny neurons of the rat striatum. J Neurosci 23:8931–8940
Henselmans JM, Hoogland PV, Stoof JC (1991) Differences in the regulation of acetylcholine release upon D2 dopamine and N-methyl-D-aspartate receptor activation between the striatal complex of reptiles and the neostriatum of rats. Brain Res 566:8–12
Henze DA, González-Burgos GR, Urban NN, Lewis DA, Barrionuevo G (2000) Dopamine increases excitability of pyramidal neurons in primate prefrontal cortex. J Neurophysiol 84:2799–2809
Hernández-López S, Bargas J, Surmeier DJ, Reyes A, Galarraga E (1997) D1 receptor activation enhances evoked discharge in neostriatal medium spiny neurons by modulating an L-type Ca2+-conductance. J Neurosci 17:3334–3342
Hernández-López S, Tkatch T, Pérez-Garci E, Galarraga E, Bargas J, Hamm H, Surmeier DJ (2000) D2 dopamine receptors in striatal medium spiny neurons reduce L-type Ca2+ currents and excitability through a novel PLCB1/IP3/calcineurin signaling cascade. J Neurosci 20:8987–8995
Hopf FW, Mailliard WS, Gonzalez GF, Diamond I, Bonci A (2005) Atypical protein kinase C is a novel mediator of dopamine-enhanced firing in nucleus accumbens neurons. J Neurosci 25:985–989
Kröner S, Rosenkranz JA, Grace A, Barrionuevo G (2005) Dopamine modulates excitability of basolateral amygdala neurons in vitro. J Neurophysiol 93:1598–1610
Lefebvre L, Reader SM, Sol D (2004) Brains, innovations and evolution in birds and primates. Brain Behav Evol 63:233–246
Marin O, Smeets W, González A (1998) Evolution of the basal ganglia in tetrapods: a new perspective based on recent studies in amphibians. TINS 21:487–494
Medina L, Reiner A (1995) Neurotransmitter organization and connectivity of the basal ganglia in vertebrates: implications for the evolution of basal ganglia. Brain Behav Evol 46:235–258
Medina L, Smeets WJAJ (1991) Comparative aspects of the basal ganglia-tectal pathways in reptiles. J Comp Neurol 308:614–629
Nisenbaum ES, Wilson CJ (1995) Potassium currents responsible for inward and outward rectification in rat neostriatal spiny projection neurons. J Neurosci 15:4449–4463
Northcutt RG (1981) Evolution of the telencephalon in non-mammals. Ann Rev Neurosci 4:301–350
Pacheco-Cano MT, Bargas J, Hernández-López S, Tapia D, Galarraga E (1996) Inhibitory action of dopamine involves a subthreshold Cs+-sensitive conductance. Exp Brain Res 110:205–211
Parent A (1973) Distribution of monoamine-containing nerve terminals in the brain of the painted turtle, Chrysemys picta. J Comp Neurol 148:153–165
Parent A (1976) Striatal afferent connections in the turtle (Chrysemys picta) as revealed by retrograde axonal transport of horseradish peroxidase. Brain Res 108:25–36
Pérez-Garci E, Bargas J, Galarraga E (2003) The role of Ca2+ channels in the repetitive firing of striatal projection neurons. Neuroreport 14:1253–1256
Perkel DJ, Farries MA (2000) Complementary ‘bottom-up’ and ‘top-down’ approaches to basal ganglia function. Curr Opin Neurobiol 10:725–731
Pineda JC, Galarraga E, Bargas J, Cristancho M, Aceves J (1992) Charybdotoxin and apamin sensitivity of the calcium-dependent repolarization and the afterhyperpolarization in neostriatal neurons. J Neurophysiol 68:287–294
Powers AS, Reiner A (1980) A stereotaxic atlas of the forebrain and midbrain of the pattern painted turtle (Chrysemys picta picta). J Hirnforsch 21:125–159
Ramanathan S, Tkatch T, Atherton JF, Wilson CJ, Bevan MD (2008) D2-like dopamine receptors modulate SKCa channel function in subthalamic nucleus neurons through inhibition of 2 channels. J Neurophysiol 99:442–459
Redgrave P, Presccott T, Gurney K (1999) The basal ganglia: a vertebrate solution to the selection problem? Neuroscience 89:1009–1023
Reiner A (1987) The distribution of proenkephali-derived peptides in the central nervous system of turtles. J Comp Neurol 259:65–91
Reiner A, Brauth SE, Kitt CA, Karten HJ (1980) Basal ganglionic pathways to the tectum: studies in reptiles. J Comp Neurol 193:565–589
Reiner A, Brauth SE, Karten HJ (1984) Evolution of the amniote basal ganglia. TINS 7:320–325
Reiner A, Medina L, Veenman CL (1998) Structural and functional evolution of the basal ganglia in vertebrates. Brain Res Rev 28:235–285
Reyes A, Galarraga E, Flores-Hernández J, Tapia D, Bargas J (1998) Passive properties of neostriatal neurons during potassium conductance blockade. Exp Brain Res 120:70–84
Richfield EK, Young AB, Penney JB (1987) Comparative distribution of dopamine D1 and D2 receptors in the basal ganglia of turtles, pigeons, rats, cats, and monkeys. J Comp Neurol 262:446–463
Rieke GL (1981) Movement disorders and lesions of pigeon brainstem analogues of basal ganglia. Physiol Behav 26:379–384
Sánchez-Mejorada E, Sánchez-Mondragon G, Pineda JC, González M, Barral J (2009) N-type of calcium channels mediates a GABAB presynaptic modulation of the corticostriatal synapses in turtle’s paleostriatum augmentatum. Synapse 63:855–862
Smeets W (1994) Catecholamine system in the CNS of reptiles: structure and functional correlations. In: Smeets W, Reiner A (eds) Phylogeny and development of cathecholamine systems in the CNS of vertebrates. Cambridge University Press, Cambridge, pp 103–133
Smeets W, Gonzalez A (2000) Catecholamine systems in the brain of vertebrates: new perspectives through a comparative approach. Brain Res Rev 33:308–379
Smeets W, Jonker AJ, Hoogland PV (1987) The distribution of dopamine in the forebrain and midbrain of a turtle, Pseudemys scripta elegans, reinvestigated using antibodies against dopamine. Brain Behav Evol 30:121–142
Smeets W, Marín O, González A (2000) Evolution of the basal ganglia: new perspectives through a comparative approach. J Anat 196:501–517
Smeets W, Lopez JM, Gonzalez A (2003) Immunohistochemical localization of DARPP-32 in the brain of the turtle, Pseudemys scripta elegans: further assessment of its relationship with dopaminergic systems in reptiles. J Chem Neuroanat 25:83–95
West AR, Grace AA (2002) Opposite influences of endogenous dopamine D1 and D2 receptor activation on activity states and electrophysiological properties of striatal neurons: studies combining in vivo intracellular recordings and reverse microdialysis. J Neurosci 22:294–304
Wilson CJ (2004) The Basal ganglia. In: Shepherd GM (ed) The synaptic organization of the brain. Oxford University Press, NY, pp 361–414
Yang CR, Seamans JK (1996) Dopamine D1 receptor actions in layers V–VI rat prefrontal cortex neurons in vitro: modulation of dendritic-somatic signal integration. J Neurosci 16:1922–1935
Acknowledgments
We thank Beatriz Rubio Morales and Felipe Correa Sanchez from the Herpetology Lab of FES-Iztacala-UNAM for animal care. We also thank JA Laville for technical support and advice. This study was supported by: DGAPA Grants (IN201507 to EG, IN201607 to J Bargas, IN204407 to J Barral), PAPCA-2009-2010-FES Iztacala grant (to J Barral), and CONACyT grant (49484 to J Bargas, 81518 to J Barral).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barral, J., Galarraga, E., Tapia, D. et al. Dopaminergic Modulation of Spiny Neurons in the Turtle Striatum. Cell Mol Neurobiol 30, 743–750 (2010). https://doi.org/10.1007/s10571-010-9499-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-010-9499-7