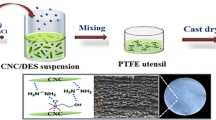
Abstract
An environment-friendly method for obtaining cellulose nanocrystals (CNC) using deep eutectic solvents (DES) was developed. Formation of highly crystalline CNC with average particle dimensions 20 × 100 × 700 nm was confirmed with SEM and AFM. Molecular dynamics simulations demonstrated that the hydrogen bond interactions of the cellulose hydroxyl groups with the urea C=O group and with the chloride ions were the key factors of the destruction of MCC particles in the process of solvation. The type of cellulose crystal structure (Iβ) and the high degree of crystallinity (about 80% according to Segal method) were preserved during treatment with DES. The ability of the prepared CNC to act as a reinforcing filler was tested by introduction of them into the chitosan-based films plasticized with DES. It was found that addition of 2 wt% of CNC led to an increase in the strength of the films from 11.4 up to 20.4 MPa with a simultaneous increase in the elongation at break.
Graphic abstract






Similar content being viewed by others
References
Abraham MJ, Murtola T, Schulz R et al (2015) GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2:19–25. https://doi.org/10.1016/J.SOFTX.2015.06.001
Amde M, Liu J-F, Pang L (2015) Environmental application, fate, effects, and concerns of ionic liquids: a review. Environ Sci Technol 49:12611–12627. https://doi.org/10.1021/acs.est.5b03123
Amin KNM, Annamalai PK, Morrow IC, Martin D (2015) Production of cellulose nanocrystals via a scalable mechanical method. RSC Adv 5:57133–57140. https://doi.org/10.1039/C5RA06862B
Armelin E, Pérez-Madrigal MM, Alemán C, Diaz DD (2016) Current status and challenges of biohydrogels for applications as supercapacitors and secondary batteries. J Mater Chem A 4:8952–8968. https://doi.org/10.1039/C6TA01846G
Bai W, Holbery J, Li K (2009) A technique for production of nanocrystalline cellulose with a narrow size distribution. Cellulose 16:455–465. https://doi.org/10.1007/s10570-009-9277-1
Bajkacz S, Adamek J (2017) Evaluation of new natural deep eutectic solvents for the extraction of isoflavones from soy products. Talanta 168:329–335. https://doi.org/10.1016/j.talanta.2017.02.065
Cao L, Yuan D, Fu X, Chen Y (2018) Green method to reinforce natural rubber with tunicate cellulose nanocrystals via one-pot reaction. Cellulose 25:4551–4563. https://doi.org/10.1007/s10570-018-1877-1
Chi K, Catchmark JM (2018) Improved eco-friendly barrier materials based on crystalline nanocellulose/chitosan/carboxymethyl cellulose polyelectrolyte complexes. Food Hydrocoll 80:195–205. https://doi.org/10.1016/j.foodhyd.2018.02.003
Decaen P, Rolland-Sabaté A, Guilois S et al (2017) Choline chloride vs choline ionic liquids for starch thermoplasticization. Carbohydr Polym 177:424–432. https://doi.org/10.1016/j.carbpol.2017.09.012
Deng F, Li M-C, Ge X et al (2017) Cellulose Nanocrystals/poly(methyl methacrylate) nanocomposite films: effect of preparation method and loading on the optical, thermal, mechanical, and gas barrier properties. Polym Compos 38:E137–E146. https://doi.org/10.1002/pc.23875
dos Santos FA, Iulianelli GCV, Tavares MIB (2017) Effect of microcrystalline and nanocrystals cellulose fillers in materials based on PLA matrix. Polym Test 61:280–288. https://doi.org/10.1016/j.polymertesting.2017.05.028
Essmann U, Perera L, Berkowitz ML et al (1995) A smooth particle mesh Ewald method. J Chem Phys 103:8577–8593. https://doi.org/10.1063/1.470117
Feng Q, Hou D, Zhao Y et al (2014) Electrospun regenerated cellulose nano fibrous membranes surface- grafted with polymer chains/brushes via the atom transfer radical polymerization method for catalase immobilization. ACS Appl Mater Interfaces 6:20958–20967. https://doi.org/10.1021/am505722g
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. https://doi.org/10.1007/s10570-013-0030-4
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Galvis-Sánchez AC, Sousa AMM, Hilliou L et al (2016) Thermo-compression molding of chitosan with a deep eutectic mixture for biofilms development. Green Chem 18:1571–1580. https://doi.org/10.1039/C5GC02231B
Hirota M, Tamura N, Saito T, Isogai A (2010) Water dispersion of cellulose II nanocrystals prepared by TEMPO-mediated oxidation of mercerized cellulose at pH 4.8. Cellulose 17:279–288. https://doi.org/10.1007/s10570-009-9381-2
Hoover WG (1985) Canonical dynamics: equilibrium phase-space distributions. Phys Rev A 31:1695–1697. https://doi.org/10.1103/PhysRevA.31.1695
Kabanov VA (ed) (1977) Enciklopedija polimerov, vol 3. Sovetskaya Encyclopedia, Moscow
Kony D, Damm W, Stoll S, Van Gunsteren WF (2002) An improved OPLS-AA force field for carbohydrates. J Comput Chem 23:1416–1429. https://doi.org/10.1002/jcc.10139
Kostritskii AY, Tolmachev DA, Lukasheva NV, Gurtovenko AA (2017) Molecular-level insight into the interaction of phospholipid bilayers with cellulose. Langmuir 33:12793–12803. https://doi.org/10.1021/acs.langmuir.7b02297
Lee CM, Kubicki JD, Fan B et al (2015) Hydrogen-bonding network and OH stretch vibration of cellulose: comparison of computational modeling with polarized IR and SFG spectra. J Phys Chem B 119:15138–15149. https://doi.org/10.1021/acs.jpcb.5b08015
Leung ACW, Hrapovic S, Lam E et al (2011) Characteristics and properties of carboxylated cellulose nanocrystals prepared from a novel one-step procedure. Small 7:302–305. https://doi.org/10.1002/smll.201001715
Li J, Wei X, Wang Q et al (2012) Homogeneous isolation of nanocellulose from sugarcane bagasse by high pressure homogenization. Carbohydr Polym 90:1609–1613. https://doi.org/10.1016/j.carbpol.2012.07.038
Li X, Feng Y, Chu G et al (2018) Directly and quantitatively studying the interfacial interaction between SiO 2 and elastomer by using peak force AFM. Compos Commun 7:36–41. https://doi.org/10.1016/j.coco.2017.12.006
Ling Z, Edwards JV, Guo Z et al (2019) Structural variations of cotton cellulose nanocrystals from deep eutectic solvent treatment: micro and nano scale. Cellulose 26:861–876. https://doi.org/10.1007/s10570-018-2092-9
Liu Y, Guo B, Xia Q et al (2017) Efficient cleavage of strong hydrogen bonds in cotton by deep eutectic solvents and facile fabrication of cellulose nanocrystals in high yields. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.7b00954
Lukasheva NV, Tolmachev DA (2016) Cellulose nanofibrils and mechanism of their mineralization in biomimetic synthesis of hydroxyapatite/native bacterial cellulose nanocomposites: molecular dynamics simulations. Langmuir 32:125–134. https://doi.org/10.1021/acs.langmuir.5b03953
Lynam JG, Kumar N, Wong MJ (2017) Deep eutectic solvents’ ability to solubilize lignin, cellulose, and hemicellulose; thermal stability; and density. Bioresour Technol. https://doi.org/10.1016/j.biortech.2017.04.079
Man Z, Muhammad N, Sarwono A et al (2011) Preparation of cellulose nanocrystals using an ionic liquid. J Polym Environ 19:726–731. https://doi.org/10.1007/s10924-011-0323-3
Mao H, Wei C, Gong Y et al (2019) Mechanical and water-resistant properties of eco-friendly chitosan membrane reinforced with cellulose nanocrystals. Polym (Basel) 11:166. https://doi.org/10.3390/polym11010166
Maréchal Y, Chanzy H (2000) The hydrogen bond network in Iβ cellulose as observed by infrared spectrometry. J Mol Struct 523:183–196. https://doi.org/10.1016/S0022-2860(99)00389-0
Marín-Silva DA, Rivero S, Pinotti A (2019) Chitosan-based nanocomposite matrices: development and characterization. Int J Biol Macromol 123:189–200. https://doi.org/10.1016/j.ijbiomac.2018.11.035
Miao J, Yu Y, Jiang Z, Zhang L (2016) One-pot preparation of hydrophobic cellulose nanocrystals in an ionic liquid. Cellulose 23:1209–1219. https://doi.org/10.1007/s10570-016-0864-7
Mondal S (2017) Preparation, properties and applications of nanocellulosic materials. Carbohydr Polym 163:301–316. https://doi.org/10.1016/j.carbpol.2016.12.050
Mostofian B, Smith JC, Cheng X (2014) Simulation of a cellulose fiber in ionic liquid suggests a synergistic approach to dissolution. Cellulose 21:983–997. https://doi.org/10.1007/s10570-013-0018-0
Mukesh C, Mondal D, Sharma M, Prasad K (2014) Choline chloride-thiourea, a deep eutectic solvent for the production of chitin nanofibers. Carbohydr Polym 103:466–471. https://doi.org/10.1016/j.carbpol.2013.12.082
Nosé S (1984) A molecular dynamics method for simulations in the canonical ensemble. Mol Phys 52:255–268. https://doi.org/10.1080/00268978400101201
Parrinello M, Rahman A (1981) Polymorphic transitions in single crystals: a new molecular dynamics method. J Appl Phys 52:7182–7190. https://doi.org/10.1063/1.328693
Ren H, Chen C, Wang Q et al (2016) The properties of choline chloride-based deep eutectic solvents and their performance in the dissolution of cellulose. BioResources 11:5435–5451. https://doi.org/10.15376/biores.11.2.5435-5451
Rovera C, Ghaani M, Santo N et al (2018) Enzymatic hydrolysis in the green production of bacterial cellulose nanocrystals. ACS Sustain Chem Eng 6:7725–7734. https://doi.org/10.1021/acssuschemeng.8b00600
Samarov AA, Smirnov MA, Sokolova MP, Toikka AM (2018a) Liquid–liquid equilibrium data for the system N-Octane + Toluene + DES at 293.15 and 313.15 K and atmospheric pressure. Theor Found Chem Eng 52:262–267. https://doi.org/10.1134/S0040579518020148
Samarov AA, Smirnov MA, Toikka AM, Prikhodko IV (2018b) Study of deep eutectic solvent on the base choline chloride as entrainer for the separation alcohol-ester systems. J Chem Eng Data 63:1877–1884. https://doi.org/10.1021/acs.jced.7b00912
Sambasivarao SV, Acevedo O (2009) Development of OPLS-AA force field parameters for 68 unique ionic liquids. J Chem Theory Comput 5:1038–1050. https://doi.org/10.1021/ct900009a
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-Ray diffractometer. Text Res J 29:786–794. https://doi.org/10.1177/004051755902901003
Sirviö JA, Visanko M, Liimatainen H (2015) Deep eutectic solvent system based on choline chloride-urea as a pre-treatment for nanofibrillation of wood cellulose. Green Chem 17:3401–3406. https://doi.org/10.1039/C5GC00398A
Sirviö JA, Visanko M, Liimatainen H (2016) Acidic Deep eutectic solvents as hydrolytic media for cellulose nanocrystal production. Biomacromolecules 17:3025–3032. https://doi.org/10.1021/acs.biomac.6b00910
Smirnov MA, Vorobiov VK, Sokolova MP et al (2018) Electrochemical properties of supercapacitor electrodes based on polypyrrole and enzymatically prepared cellulose nanofibers. Polym Sci Ser C 60:228–239. https://doi.org/10.1134/S1811238218020194
Smith EL, Abbott AP, Ryder KS (2014) Deep eutectic solvents (DESs) and their applications. Chem Rev 114:11060–11082. https://doi.org/10.1021/cr300162p
Sokolova MP, Smirnov MA, Samarov AA et al (2018) Plasticizing of chitosan films with deep eutectic mixture of malonic acid and choline chloride. Carbohydr Polym 197:548–557. https://doi.org/10.1016/j.carbpol.2018.06.037
Sun H, Li Y, Wu X, Li G (2013) Theoretical study on the structures and properties of mixtures of urea and choline chloride. J Mol Model 19:2433–2441. https://doi.org/10.1007/s00894-013-1791-2
Suopajärvi T, Sirviö JA, Liimatainen H (2017) Nanofibrillation of deep eutectic solvent-treated paper and board cellulose pulps. Carbohydr Polym 169:167–175. https://doi.org/10.1016/j.carbpol.2017.04.009
Surov OV, Voronova MI, Afineevskii AV, Zakharov AG (2018a) Polyethylene oxide films reinforced by cellulose nanocrystals: microstructure-properties relationship. Carbohydr Polym 181:489–498. https://doi.org/10.1016/j.carbpol.2017.10.075
Surov OV, Voronova MI, Rubleva NV et al (2018b) A novel effective approach of nanocrystalline cellulose production: oxidation–hydrolysis strategy. Cellulose 25:5035–5048. https://doi.org/10.1007/s10570-018-1910-4
Tang J, Berry RM, Tam KC (2016) Stimuli-responsive cellulose nanocrystals for surfactant-free oil harvesting. Biomacromolecules 17:1748–1756. https://doi.org/10.1021/acs.biomac.6b00144
Tenhunen T-M, Lewandowska AE, Orelma H et al (2018) Understanding the interactions of cellulose fibres and deep eutectic solvent of choline chloride and urea. Cellulose 25:137–150. https://doi.org/10.1007/s10570-017-1587-0
Troter DZ, Todorović ZB, Đokić-Stojanović DR et al (2016) Application of ionic liquids and deep eutectic solvents in biodiesel production: a review. Renew Sustain Energy Rev 61:473–500
Wang Y, Wang X, Xie Y, Zhang K (2018) Functional nanomaterials through esterification of cellulose: a review of chemistry and application. Cellulose 25:3703–3731. https://doi.org/10.1007/s10570-018-1830-3
Weerasinghe S, Smith PE (2003) A Kirkwood–Buff derived force field for sodium chloride in water. J Phys Chem B 119:3891–3898
Wu W, Huang F, Pan S et al (2015) Thermo-responsive and fluorescent cellulose nanocrystals grafted with polymer brushes. J Mater Chem A Mater energy Sustain 3:1995–2005. https://doi.org/10.1039/C4TA04761C
Ying Z, Wu D, Wang Z et al (2018) Rheological and mechanical properties of polylactide nanocomposites reinforced with the cellulose nanofibers with various surface treatments. Cellulose 25:3955–3971. https://doi.org/10.1007/s10570-018-1862-8
Zainal-Abidin MH, Hayyan M, Hayyan A, Jayakumar NS (2017) New horizons in the extraction of bioactive compounds using deep eutectic solvents: a review. Anal Chim Acta 979:1–23. https://doi.org/10.1016/j.aca.2017.05.012
Zhang Q, Vigier KDO, Royer S, Jerome F (2012) Deep eutectic solvents: syntheses, properties and applications. Chem Soc Rev Chem Soc Rev 41:7108–7146. https://doi.org/10.1039/c2cs35178a
Zhou Y, Saito T, Bergström L, Isogai A (2018) Acid-free preparation of cellulose nanocrystals by TEMPO oxidation and subsequent cavitation. Biomacromolecules 19:633–639. https://doi.org/10.1021/acs.biomac.7b01730
Zhu P, Gu Z, Hong S, Lian H (2017) One-pot production of chitin with high purity from lobster shells using choline chloride–malonic acid deep eutectic solvent. Carbohydr Polym 177:217–223. https://doi.org/10.1016/j.carbpol.2017.09.001
Acknowledgments
Preparation and study of cellulose nanocrystals and molecular dynamic simulations were conducted with a financial support of Russian Ministry of Education and Science within State Contract 14.W03.31.0014 (megagrant). The experimental work was facilitated by the equipment of the Resource Centre for X-ray Diffraction Studies, Centre for Microscopy and Microanalysis, Thermogravimetric and Calorimetric Research Centre, Centre for Optical and Laser Materials Research and Interdisciplinary Resource Centre for Nanotechnology at Saint Petersburg State University and by the Engineering Center of the Saint-Petersburg State Technological Institute (Technical University). Calculations were performed with the use of the resources of the Institute of Macromolecular Compounds and the resources of the Center of collective use “Complex of modelling and data research mega-class facilities” NRC “Kurchatov Institute”. Unique identifier RFMEFI62114X0006.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smirnov, M.A., Sokolova, M.P., Tolmachev, D.A. et al. Green method for preparation of cellulose nanocrystals using deep eutectic solvent. Cellulose 27, 4305–4317 (2020). https://doi.org/10.1007/s10570-020-03100-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03100-1