Abstract
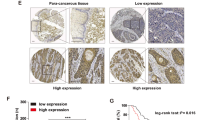
Increasing evidence suggests that targeting ubiquitin-specific peptidase 8 (USP8) serves as an attractive anti-cancer strategy. However, the role of USP8 inhibitor, DUB-IN-1, in esophageal squamous cell carcinoma (ESCC) cells still needs to be explored. Here, immunohistochemistry was employed to examine the expression of USP8 in ESCC tissues. Cell Counting Kit-8 (CCK-8) was used to evaluate cell proliferation ability, and propidium iodide (PI) was selected to test the effect of DUB-IN-1 on cell cycle. AnnexinV-FITC/PI staining and the activity of caspase 3 were detedcted to evaluate apoptosis. Transmission electron microscope, microtubule-associated protein 1 light-chain 3 (LC3) expression, and acridine orange (AO) staining were selected to check if there was autophagy. Comet assay and γ-H2AX immunofluorescence was used to monitor DNA damage. Rescue experiment was used to determine the key role of of p53 in cell cycle, apoptosis, and autophagy. Results revealed that the leve of USP8 was higher in ESCC tissues than that in tissues adjacent to carcinoma. DUB-IN-1, an USP8 inhibitor, caused DNA damage, led to G2/M phase block by p53-p21 axis, and triggered apoptosis by regulating the p53 target proteins including Bax, Noxa, and Puma. Besides, DUB-IN-1 could stimulate autophagy through p53-dependent adenosine 5′-monophosphate (AMP)–activated protein kinase (AMPK) activation. Taken together, this study revealed the cytotoxic effects and the mechanism of DUB-IN-1, which indicated that DUB-IN-1 may be a novel inhibitor targeting USP8 that can kill ESCC cells.
Graphical abstract
USP8 inhibitor, DUB-IN-1, treatment could inhibit esophageal squamous cell carcinoma cell growth and induce G2/M cell cycle arrest, apoptosis, and autophagy by DNA damage–induced p53 activation. DUB-IN-1 treatment led to G2/M cell cycle arrest by upregulating the protein level of p21 and triggered apoptosis by modulating the p53 target proteins including Bax, Noxa, and Puma. Meanwhile, DUB-IN-1 treatment stimulated protective autophagy through p53-dependent AMPK activation. Collectively, these findings suggested that DNA damage–triggered p53 activation, p53-Puma/Noxa/Bax, p53-p21, and p53-AMPK pathways were all involved in the effect of DUB-IN-1.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Code availability
Not applicable.
References
Abbas T, Dutta A. p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer. 2009;9:400–14. https://doi.org/10.1038/nrc2657.
Agathanggelou A, Smith E, Davies NJ, Kwok M, Zlatanou A, Oldreive CE, et al. USP7 inhibition alters homologous recombination repair and targets CLL cells independently of ATM/p53 functional status. Blood. 2017;130:156–66. https://doi.org/10.1182/blood-2016-12-758219.
AlwanHAvanLeeuwen JE. UBPY-mediated epidermal growth factor receptor (EGFR) de-ubiquitination promotes EGFR degradation. J Biol Chem. 2007;282:1658–69. https://doi.org/10.1074/jbc.M604711200.
Baykara M, Yaman M, Buyukberber S, Tufan G, Demirci U, Benekli M, et al. Clinical and prognostic importance of XIAP and USP8 in advanced stages of non-small cell lung cancer. J BUON. 2013;18:921–7.
Budanov AV, Karin M. p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell. 2008;134:451–60. https://doi.org/10.1016/j.cell.2008.06.028.
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP, et al. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science. 1998;282:1497–501. https://doi.org/10.1126/science.282.5393.1497.
Burmakin M, Shi Y, Hedstrom E, Kogner P, Selivanova G. Dual targeting of wild-type and mutant p53 by small molecule RITA results in the inhibition of N-Myc and key survival oncogenes and kills neuroblastoma cells in vivo and in vitro. Clin Cancer Res. 2013;19:5092–103. https://doi.org/10.1158/1078-0432.CCR-12-2211.
Byun S, Lee SY, Lee J, Jeong CH, Farrand L, Lim S, et al. USP8 is a novel target for overcoming gefitinib resistance in lung cancer. Clin Cancer Res. 2013;19:3894–904. https://doi.org/10.1158/1078-0432.CCR-12-3696.
Colombo M, Vallese S, Peretto I, Jacq X, Rain JC, Colland F, et al. Synthesis and biological evaluation of 9-oxo-9H-indeno[12-b]pyrazine-23-dicarbonitrile analogues as potential inhibitors of deubiquitinating enzymes. ChemMedChem. 2010;5:552–8. https://doi.org/10.1002/cmdc.200900409.
Cui J, Xing L, Li Z, Wu S, Wang J, Liu J, et al. Ochratoxin A induces G(2) phase arrest in human gastric epithelium GES-1 cells in vitro. Toxicol Lett. 2010;193:152–8. https://doi.org/10.1016/j.toxlet.2009.12.019.
Duan B, Wang C, LiuZYang X. USP8 is a novel therapeutic target in melanoma through regulating receptor tyrosine kinase levels. Cancer Manag Res. 2021;13:4181–9. https://doi.org/10.2147/CMAR.S300195.
Duan L, Perez RE, Davaadelger B, Dedkova EN, Blatter LA, Maki CG. p53-regulated autophagy is controlled by glycolysis and determines cell fate. Oncotarget. 2015;6:23135–56. https://doi.org/10.18632/oncotarget.5218.
Feng Z, Zhang H, Levine AJ, Jin S. The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci U S A. 2005;102:8204–9. https://doi.org/10.1073/pnas.0502857102.
Fischle W, Tseng BS, Dormann HL, Ueberheide BM, Garcia BA, Shabanowitz J, et al. Regulation of HP1-chromatin binding by histone H3 methylation and phosphorylation. Nature. 2005;438:1116–22. https://doi.org/10.1038/nature04219.
Ge C, Che L, Ren J, Pandita RK, Lu J, Li K, et al. BRUCE regulates DNA double-strand break response by promoting USP8 deubiquitination of BRIT1. Proc Natl Acad Sci U S A. 2015;112:E1210-9. https://doi.org/10.1073/pnas.1418335112.
Halazonetis TD, Gorgoulis VG, Bartek J. An oncogene-induced DNA damage model for cancer development. Science. 2008;319:1352–5. https://doi.org/10.1126/science.1140735.
Han J, Tian Y, Yu L, Zhang Q, Xu X, Zhang Y, et al. Discovery of novel USP8 inhibitors via Ubiquitin-Rho-110 fluorometric assay based high throughput screening. Bioorg Chem. 2020;101:103962. https://doi.org/10.1016/j.bioorg.2020.103962.
He Y, Li D, Shan B, Liang D, Shi J, Chen W, et al. Incidence and mortality of esophagus cancer in China 2008–2012. Chin J Cancer Res. 2019;31:426–34. https://doi.org/10.21147/j.issn.1000-9604.2019.03.04.
Hirao A, Kong YY, Matsuoka S, Wakeham A, Ruland J, Yoshida H, et al. DNA damage-induced activation of p53 by the checkpoint kinase Chk2. Science. 2000;287:1824–7. https://doi.org/10.1126/science.287.5459.1824.
Hong YA, Lim JH, Kim MY, Kim Y, Park HS, Kim HW, et al. Extracellular superoxide dismutase attenuates renal oxidative stress through the activation of adenosine monophosphate-activated protein kinase in diabetic nephropathy. Antioxid Redox Signal. 2018;28:1543–61. https://doi.org/10.1089/ars.2017.7207.
Hu T, Zhang J, Sha B, Li M, Wang L, Zhang Y, et al. Targeting the overexpressed USP7 inhibits esophageal squamous cell carcinoma cell growth by inducing NOXA-mediated apoptosis. Mol Carcinog. 2019;58:42–54. https://doi.org/10.1002/mc.22905.
Islam MT, ChenFChen H. The oncogenic role of ubiquitin specific peptidase (USP8) and its signaling pathways targeting for cancer therapeutics. Arch Biochem Biophys. 2021;701:108811. https://doi.org/10.1016/j.abb.2021.108811.
Jeong CH. Inhibition of ubiquitin-specific peptidase 8 suppresses growth of gefitinib-resistant non-small cell lung cancer cells by inducing apoptosis. J Cancer Prev. 2015;20:57–63. https://doi.org/10.15430/JCP.2015.20.1.57.
Jeong M, Lee EW, Seong D, Seo J, Kim JH, Grootjans S, et al. USP8 suppresses death receptor-mediated apoptosis by enhancing FLIPL stability. Oncogene. 2017;36:458–70. https://doi.org/10.1038/onc.2016.215.
Ji J, Wu K, Wu M, Zhan Q. p53 functional activation is independent of its genotype in five esophageal squamous cell carcinoma cell lines. Front Med China. 2010;4:412–8. https://doi.org/10.1007/s11684-010-0260-x.
Jiang S, Park DW, Gao Y, Ravi S, Darley-Usmar V, Abraham E, et al. Participation of proteasome-ubiquitin protein degradation in autophagy and the activation of AMP-activated protein kinase. Cell Signal. 2015;27:1186–97. https://doi.org/10.1016/j.cellsig.2015.02.024.
Jing X, Chen Y, Chen Y, Shi G, Lv S, Cheng N, et al. Down-regulation of USP8 inhibits cholangiocarcinoma cell proliferation and invasion. Cancer Manag Res. 2020;12:2185–94. https://doi.org/10.2147/CMAR.S234586.
Juretschke T, Beli P. Causes and consequences of DNA damage-induced autophagy. Matrix Biol. 2021;100–101:39–53. https://doi.org/10.1016/j.matbio.2021.02.004.
Kageyama K, Asari Y, Sugimoto Y, NiiokaKDaimon M. Ubiquitin-specific protease 8 inhibitor suppresses adrenocorticotropic hormone production and corticotroph tumor cell proliferation. Endocr J. 2020;67:177–84. https://doi.org/10.1507/endocrj.EJ19-0239.
Karuman P, Gozani O, Odze RD, Zhou XC, Zhu H, Shaw R, et al. The Peutz-Jegher gene product LKB1 is a mediator of p53-dependent cell death. Mol Cell. 2001;7:1307–19. https://doi.org/10.1016/s1097-2765(01)00258-1.
Kim Y, Shiba-Ishii A, Nakagawa T, Husni RE, Sakashita S, Takeuchi T, et al. Ubiquitin-specific protease 8 is a novel prognostic marker in early-stage lung adenocarcinoma. Pathol Int. 2017;67:292–301. https://doi.org/10.1111/pin.12546.
Li Y, Chen Y. AMPK and Autophagy. Adv Exp Med Biol. 2019;1206:85–108. https://doi.org/10.1007/978-981-15-0602-4_4.
Liu K, Zheng M, Lu R, Du J, Zhao Q, Li Z, et al. The role of CDC25C in cell cycle regulation and clinical cancer therapy: a systematic review. Cancer Cell Int. 2020;20:213. https://doi.org/10.1186/s12935-020-01304-w.
McHugh A, Fernandes K, Chinner N, Ibrahim AFM, Garg AK, Boag G, et al. The identification of potential therapeutic targets for cutaneous squamous cell carcinoma. J Invest Dermatol. 2020;140(1154–65):e5. https://doi.org/10.1016/j.jid.2019.09.024.
Meijer IM, van Leeuwen JE. ERBB2 is a target for USP8-mediated deubiquitination. Cell Signal. 2011;23:458–67. https://doi.org/10.1016/j.cellsig.2010.10.023.
Naviglio S, Mattecucci C, Matoskova B, Nagase T, Nomura N, Di Fiore PP, et al. UBPY: a growth-regulated human ubiquitin isopeptidase. EMBO J. 1998;17:3241–50. https://doi.org/10.1093/emboj/17.12.3241.
O’Connor MJ. Targeting the DNA damage response in cancer. Mol Cell. 2015;60:547–60. https://doi.org/10.1016/j.molcel.2015.10.040.
Oh YM, Lee SB, Choi J, Suh HY, Shim S, Song YJ, et al. USP8 modulates ubiquitination of LRIG1 for Met degradation. Sci Rep. 2014;4:4980. https://doi.org/10.1038/srep04980.
Panner A, Crane CA, Weng C, Feletti A, Fang S, Parsa AT, et al. Ubiquitin-specific protease 8 links the PTEN-Akt-AIP4 pathway to the control of FLIPS stability and TRAIL sensitivity in glioblastoma multiforme. Cancer Res. 2010;70:5046–53. https://doi.org/10.1158/0008-5472.CAN-09-3979.
Peng H, Yang F, Hu Q, Sun J, Peng C, Zhao Y, et al. The ubiquitin-specific protease USP8 directly deubiquitinates SQSTM1/p62 to suppress its autophagic activity. Autophagy. 2020;16:698–708. https://doi.org/10.1080/15548627.2019.1635381.
Qiu H, Kong J, Cheng Y, Li G. The expression of ubiquitin-specific peptidase 8 and its prognostic role in patients with breast cancer. J Cell Biochem. 2018;119:10051–8. https://doi.org/10.1002/jcb.27337.
Robert C, Rassool FV. HDAC inhibitors: roles of DNA damage and repair. Adv Cancer Res. 2012;116:87–129. https://doi.org/10.1016/B978-0-12-394387-3.00003-3.
Rong Z, Zhu Z, Cai S, Zhang B. Knockdown of USP8 inhibits the growth of lung cancer cells. Cancer Manag Res. 2020;12:12415–22. https://doi.org/10.2147/IJN.S259191.
Shenker BJ, Walker LM, Zekavat A, Weiss RHBoesze-Battaglia K. The cell-cycle regulatory protein p21(CIP1/WAF1) is required for cytolethal distending toxin (Cdt)-induced apoptosis.Pathogens. 2020; 9. https://doi.org/10.3390/pathogens9010038.
Shi Q, Shen LY, Dong B, Fu H, Kang XZ, Yang YB, et al. The identification of the ATR inhibitor VE-822 as a therapeutic strategy for enhancing cisplatin chemosensitivity in esophageal squamous cell carcinoma. Cancer Lett. 2018;432:56–68. https://doi.org/10.1016/j.canlet.2018.06.010.
Shin S, Kim K, Kim HR, Ylaya K, Do SI, Hewitt SM, et al. Deubiquitylation and stabilization of Notch1 intracellular domain by ubiquitin-specific protease 8 enhance tumorigenesis in breast cancer. Cell Death Differ. 2020;27:1341–54. https://doi.org/10.1038/s41418-019-0419-1.
Siegel RL, Miller KD, Jemal A. Cancer statistics 2020. CA Cancer J Clin. 2020;70:7–30. https://doi.org/10.3322/caac.21590.
Sun J, Shen D, Gao Y, Zheng Y, Zhao L, Maa M, et al. Down-regulation of USP8 suppresses HER-3 positive gastric cancer cells proliferation. Onco Targets Ther. 2020;13:7973–84. https://doi.org/10.2147/OTT.S264108.
Sun J, Shen D, Zheng Y, Ren H, Liu H, Chen X, et al. USP8 inhibitor suppresses HER-2 positive gastric cancer cell proliferation and metastasis via the PI3K/AKT signaling pathway. Onco Targets Ther. 2020;13:9941–52. https://doi.org/10.2147/OTT.S271496.
Thanasoula M, Escandell JM, Suwaki N, Tarsounas M. ATM/ATR checkpoint activation downregulates CDC25C to prevent mitotic entry with uncapped telomeres. EMBO J. 2012;31:3398–410. https://doi.org/10.1038/emboj.2012.191.
Wang C, Wang H, Lieftink C, du Chatinier A, Gao D, Jin G, et al. CDK12 inhibition mediates DNA damage and is synergistic with sorafenib treatment in hepatocellular carcinoma. Gut. 2020;69:727–36. https://doi.org/10.1136/gutjnl-2019-318506.
Wang L, Li M, Sha B, Hu X, Sun Y, Zhu M, et al. Inhibition of deubiquitination by PR-619 induces apoptosis and autophagy via ubi-protein aggregation-activated ER stress in oesophageal squamous cell carcinoma. Cell Prolif. 2021;54:e12919. https://doi.org/10.1111/cpr.12919.
Yan M, Zhao C, Wei N, Wu X, Cui J, Xing Y. High expression of ubiquitin-specific protease 8 (USP8) is associated with poor prognosis in patients with cervical squamous cell carcinoma. Med Sci Monit. 2018;24:4934–43. https://doi.org/10.12659/MSM.909235.
Yu Z, Wang H, Zhang L, Tang A, Zhai Q, Wen J, et al. Both p53-PUMA/NOXA-Bax-mitochondrion and p53–p21cip1 pathways are involved in the CDglyTK-mediated tumor cell suppression. Biochem Biophys Res Commun. 2009;386:607–11. https://doi.org/10.1016/j.bbrc.2009.06.083.
Zhang C, Liu K, Yao K, Reddy K, Zhang Y, Fu Y, et al. HOI-02 induces apoptosis and G2-M arrest in esophageal cancer mediated by ROS. Cell Death Dis. 2015;6:e1912. https://doi.org/10.1038/cddis.2015.227.
Zhu Y, Xu J, Hu W, Wang F, Zhou Y, Gong W, et al. Inhibiting USP8 overcomes hepatocellular carcinoma resistance via suppressing receptor tyrosine kinases. Aging (Albany NY). 2021;13:14999–5012. https://doi.org/10.18632/aging.203061.
Zou J, Li S, Chen Z, Lu Z, Gao J, Zou J, et al. A novel oral camptothecin analog, gimatecan, exhibits superior antitumor efficacy than irinotecan toward esophageal squamous cell carcinoma in vitro and in vivo. Cell Death Dis. 2018;9:661. https://doi.org/10.1038/s41419-018-0700-0.
Acknowledgements
We appreciate the Transmission Electron Microscope (TEM) Center of Henan University of Chinese Medicine for providing the TEM services and pictures.
Funding
This work was supported by the National Natural Science Foundation Grant of China (Grant No. 81672421), Program for Innovation Research Team (in Science and Technology) in University of Henan Province (Grant No. 20IRTSTHN026), and Open Project Fund of Henan Key Laboratory of Precision Clinical Pharmacy (Grant No. HKLPCP-2020–06).
National Natural Science Foundation of China,81672421,Ping Chen,Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province,20IRTSTHN026,Pei Li,Open Project Fund of Henan Key Laboratory of Precision Clinical Pharmacy,HKLPCP-2020–06,Ping Chen
Author information
Authors and Affiliations
Contributions
Conceptualization: Tao Hu and Ping Chen; methodology: Beibei Sha, Yaxin Sun, Shan Zhao, Miaomiao Li, Wenjing Huang, and Zheng Li; data collection and analysis: Beibei Sha and Yaxin Sun; writing—original draft preparation: Beibei Sha, Yaxin Sun and Tao Hu; writing—review and editing: Jianxiang Shi and Xuefei Han; funding acquisition: Ping Chen and Pei Li; supervision: Tao Hu and Ping Chen. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Graphical headlights
1. USP8 inhibitor, DUB-IN-1, treatment induces DNA damage and activates p53.
2. DUB-IN-1 triggers apoptosis by p53-Puma/Noxa/Bax.
3. DUB-IN-1 leads to G2/M cell cycle arrest by p53-p21.
4. DUB-IN-1 stimulates protective autophagy through p53-dependent AMPK activation.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sha, B., Sun, Y., Zhao, S. et al. USP8 inhibitor–induced DNA damage activates cell cycle arrest, apoptosis, and autophagy in esophageal squamous cell carcinoma. Cell Biol Toxicol 39, 2011–2032 (2023). https://doi.org/10.1007/s10565-021-09686-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09686-x