Abstract
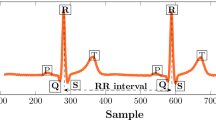
The aim of this study is to develop and describe a new ambulatory holter electrocardiogram (ECG) events detection-delineation algorithm with the major focus on the bounded false-alarm probability (FAP) segmentation of an information-optimized decision statistic. After implementation of appropriate preprocessing methods to the discrete wavelet transform (DWT) of the original ECG data, a uniform length sliding window is applied to the obtained signal and in each slid, six feature vectors namely as summation of the nonlinearly amplified Hilbert transform, summation of absolute first order differentiation, summation of absolute second order differentiation, curve length, area and variance of the excerpted segment are calculated to construct a newly proposed principal components analyzed geometric index (PCAGI) by application of a linear orthonormal projection. In the next step, the α-level Neyman-Pearson classifier (which is a FAP controlled tester) is implemented to detect and delineate QRS complexes. The presented method was applied to MIT-BIH Arrhythmia Database, QT Database, and T-Wave Alternans Database and as a result, the average values of sensitivity and positive predictivity Se = 99.96% and P+ = 99.96% are obtained for the detection of QRS complexes, with the average maximum delineation error of 5.7, 3.8 and 6.1 m for P-wave, QRS complex and T-wave, respectively. Also, the proposed method was applied to DAY general hospital high resolution holter data (more than 1,500,000 beats including Bundle Branch Blocks-BBB, Premature Ventricular Complex-PVC and Premature Atrial Complex-PAC) and average values of Se = 99.98% and P+ = 99.97% are obtained for QRS detection. In summary, marginal performance improvement of ECG events detection-delineation process in a widespread values of signal to noise ratio (SNR), reliable robustness against strong noise, artifacts and probable severe arrhythmia(s) of high resolution holter data and the processing speed 155,000 samples/s can be mentioned as important merits and capabilities of the proposed algorithm.













Similar content being viewed by others
Abbreviations
- PCAGI:
-
Principal components analyzed geometrical index
- GI:
-
Geometrical index
- HT:
-
Hilbert transform
- SVC:
-
Superior vena cava
- IVC:
-
Inferior vena cava
- AFW:
-
Atrial free wall
- TVA:
-
Tricuspid valve annulus
- FAP:
-
False alarm probability
- Pdf:
-
Probability density function
- LR:
-
Likelihood ratio
- ECG:
-
Electrocardiogram
- DWT:
-
Discrete wavelet transform
- SNR:
-
Signal to noise ratio
- QTDB:
-
QT database
- MITDB:
-
MIT-BIH arrhythmia database
- TWADB:
-
T-wave alternans database
- FP:
-
False positive
- FN:
-
False negative
- PVC:
-
Premature ventricular contraction
- AF:
-
Atrial fibrillation
- AFL:
-
Atrial flutter
- PAC:
-
Premature atrial contraction
- RCA:
-
Retrograde conduction into atrium
- FCP:
-
Full compensatory pause
- BBB:
-
Bundle branch block
- IAFDB:
-
Intracardiac atrial fibrillation database
- TP:
-
True positive
- P+:
-
Positive predictivity (%)
- Se:
-
Sensitivity (%)
- SMF:
-
Smoothing function
- FIR:
-
Finite-duration impulse response
- LE:
-
Location error
- CHECK#0:
-
Procedure of evaluating obtained results using MIT-BIH annotation files
- CHECK#1:
-
Procedure of evaluating obtained results consulting with a control cardiologist
- CHECK#2:
-
Procedure of evaluating obtained results consulting with a control cardiologist and also at least with 3 residents
References
Arzeno NM, Zhi-De Deng CSP. Analysis of first-derivative based QRS detection algorithms. IEEE Trans Biomed Eng. 2008;55(2):478–84.
Benitez D, Gaydecki PA, Zaidi A, Fitzpatrick AP. The use of the Hilbert transform in ECG signal analysis. Comput Biol Med. 2001;31:399–406.
Bishop CM. Pattern recognition and machine learning. Berlin: Springer; 2006.
Chiarugi F, Varanini M, Cantini F, Conforti F, Vrouchos G. Noninvasive ECG as a tool for predicting termination of Paroxysmal Atrial fibrillation. IEEE Trans Biomed Eng. 2007;54(8):1399–406.
Christov I, Simova I. Q-onset and T-end delineation: assessment of the performance of an automated method with the use of a reference database. Physiol Meas. 2007;28:213–21.
de Lannoy G, Frenay B, Verleysen M, Delbeke J. Supervised ECG delineation using the wavelet transform and hidden Markov models. IFMBE Proc. 2008;22:22–5.
Ghaffari A, Homaeinezhad MR, Atarod M, Ahmady Y, Rahmani R. Detecting and quantifying T-wave alternans using the correlation method and comparison with the FFT-based Method. In: 34th Annual conference of computers in cardiology (CinC). Italy: Bologna. 2008;14–7.
Ghaffari A, Homaeinezhad MR, Akraminia M, Atarod M, Daevaeiha M. Detecting and discriminating premature atrial and ventricular contractions: application to prediction of Paroxysmal Atrial fibrillation. In: 35th Annual conference of computers in cardiology (CinC), USA: Lake City-Utah. 2009a;13–6.
Ghaffari A, Homaeinezhad MR, Akraminia M, Atarod M, Daevaeiha M. Detecting and quantifying T-Wave alternans in patients with heart failure and Non-Ischemic Cardiomyopathy via modified spectral method. In: 35th Annual conference of computers in cardiology (CinC), USA: Lake City-Utah. 2009b;13–6.
Ghaffari A, Homaeinezhad MR, Akraminia M, Atarod M, Daevaeiha M. A robust wavelet-based multi-lead electrocardiogram delineation algorithm. Med Eng Phys. 2009c;31(10):1219–27.
Ghaffari A, Homaeinezhad MR, Akraminia M, Daevaeiha M. Finding events of electrocardiogram and arterial blood pressure signals via discrete wavelet transform with modified scales. Imeche proceedings, Part H: engineering in medicine (in-press) 2009d.
Ghaffari A, Homaeinezhad MR, Akraminia M, Atarod M. A methodology for prediction of acute hypotensive episodes in ICU via a risk scoring model including analysis of ST-Segment Variations. Cardiovascular engineering (in-press) 2010a.
Ghaffari A, Homaeinezhad MR, Khazraee M, Daevaeiha M. Segmentation of Holter ECG Waves via analysis of a discrete wavelet-derived multiple Skewness-Kurtosis based metric. Annals of biomedical engineering, Springer Publishing (in-press) 2010b.
Guyton AC. Text book of physiology, vol. I. Philadelphia: W.B. Saunders; 1996.
Hamilton PS, Tompkins W. Quantitative investigation of QRS detection rules using the MIT/BIH arrhythmia database. IEEE Trans Biomed Eng. 1986;33:1157–65.
Kannathal N, Lim CM, Acharya UR, Sadasivan PK. Cardiac state diagnosis using adaptive neuro fuzzy technique. Med Eng Phys. 2006;28:809–15.
Kay SM. Fundamentals of statistical signal processing: estimation theory. New Jersey: Prentice-Hall Inc; 1979.
Laguna P, Mark R, Goldenberger A, Moody GB. A database for evaluation of algorithms for measurement of QT and other waveform intervals in ECG. Proc Comput Cardiol. 1997;673–6.
Laguna P, Jane R, Caminal P. Automatic detection of wave boundaries in multi lead ECG signals: validation with the CSE database. Comput Biomed Res. 1994;27(1):45–60.
Li C, Zheng C, Tai C. Detection of ECG characteristic points using wavelet transforms. IEEE Trans Biomed Eng. 1995;42:21–8.
Lin CH, Du YC, Chen T. Adaptive wavelet network for multiple cardiac arrhythmias recognition. Expert Syst Appl. 2008;34:2601–11.
Martinez JP, Almeida R, Olmos S, Rocha AP, Laguna P. A wavelet-based ECG delineator: evaluation on standard databases. IEEE Trans Biomed Eng. 2004;51(4):570–81.
Minhas FAA, Arif M. Robust electrocardiogram (ECG) beat classification using discrete wavelet transform. Physiol Meas. 2008;29:555–70.
Mitra M, Mitra S. A software based approach for detection of QRS vector of ECG signal. IFMBE Proc. 2007;15:348–51.
Montgomery DC, Runger GC. Applied statistics and probability for engineers. 3rd ed. New Jersey: Wiley; 2003.
Moody GB. The physionet/computers in cardiology challenge 2008: T-wave alternans. Proc Comput Cardiol. 2008;35:505–8.
Moody GB, Mark RG. Development and evaluation of a 2-Lead ECG analysis program. Comput Cardiol. 1982;39–44.
Moody GB, Mark RG. The MIT-BIH Arrhythmia databaseon CD-Rom and software for it. Comput Cardiol. 1990;185–188.
Moody GB. WFDB applications guide, Tenth Edition, Harvard-MIT Division of Health Sciences and Technology, 2006. (http://www.physionet.org/physiotools/wag/).
Pan J, Tompkins WJ. A real-time QRS detection algorithm. IEEE Trans Biomed Eng. 1985;32:230–6.
Poor HV. An introduction to signal detection and estimation, vol. 1–3. 2nd ed. Berlin: Springer-Verlag; 1994.
Sayadi O, Shamsollahi MB. A model-based Bayesian framework for ECG beat segmentation. Physiol Meas. 2009;30:335–52.
Vila J, Gang Y, Presedo J, Fernandez-Delgado M, Malik M. A new approach for TU complex characterization. IEEE Trans Biomed Eng. 2000;47:764–72.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Discrete Wavelet Transform Using à Trous Method
Generally, it can be stated that the wavelet transform is a quasi-convolution of the hypothetical signal x(t) and the wavelet function ψ(t) with the dilation parameter a and translation parameter b, as follows
The parameter a can be used to adjust the wideness of the basis function and therefore the transform can be adjusted in temporal resolutions. Suppose that the function \( Y_{{a^{x} }} (b) \) is obtained based on a quasi-convolution of signal x(t) and function θ(t), as follows
If the derivative of \( Y_{{a^{x} }} (b) \) is calculated relative to b, then
On the other hand, if ψ(t) is the derivative of a smoothing function θ(t), i.e. ψ(t) = θ′(t), then
Accordingly, it can be concluded that wavelet transform at the scale a is proportional to the quasi-convolution derivative of the signal x(t) and the smoothing function θ(t). Therefore, if wavelet transform of the signal crosses of zero, it will be an indicative of local extremum(s) existence in the smoothed signal and the absolute maximum value of the wavelet transform in different scales represents a maximum slope in the filtered signal. Thus, useful information can be obtained using wavelet transform in different scales.
In Eq. 33, for dilation parameter “a” and translation parameter “b”, the values a = q k and \( b = q^{k} lT \) can be used in which q is the discretization parameter, l is a positive constant, k is the discrete scale power and T is the sampling period. By substituting the new values of the parameters “a” and “b” into Eq. 33, the following result is obtained
The scale index k determines the width of wavelet function, while the parameter l provides translation of the wavelet function.If the scale factor a and the translation parameter b are chosen as q = 2 i.e., a = 2 k and b = 2k l, the dyadic wavelet with the following basis function will be resulted (Martinez et al. 2004)
To implement the à trous wavelet transform algorithm, filters H(z) and G(z) should be used according to the block diagram represented in Fig. 14a (Martinez et al. 2004). According to this block diagram, each smoothing function (SMF) is obtained by sequential low-pass filtering (convolving with G(z) filters), while after high-pass filtering of a SMF (convolving with H(z) filters), the corresponding DWT at appropriate scale is generated. In order to decompose the input signal x(t) into different frequency passbands, according to the block diagram of Fig. 14b, sequential high-pass low-pass filtering including down-sampling should be implemented. The filter outputs x H (t) and x L (t) can be obtained by convolving the input signal x(t) with corresponding high-pass and low-pass FIRs and contributing the down-sampling as
On the other hand, to reconstruct the transformed signal, the obtained signals x H (t) and x L (t) should be first be up-sampled by following simple operation
If the FIR lengths of the H(z) and G(z) filters are represented by L H and L G , respectively, then the reconstructing high-pass and low-pass filters are obtained as
Then the reconstructed signal x R (t) is obtained by superposition of the up-sampled signals convolution with their appropriately flipped FIR filters as follow
For a prototype wavelet ψ(t) with the following quadratic spline Fourier transform,
the transfer functions H(z) and G(z) can be obtained from the following equation
and therefore,
It should be noted that for frequency contents of up to 50 Hz, the à trous algorithm can be used in different sampling frequencies. Therefore, one of the most prominent advantages of the à trous algorithm is the approximate independency of its results from sampling frequency. This is because of the main frequency contents of the ECG signal concentrate on the range less than 20 Hz (Martinez et al. 2004; Ghaffari et al. (2009c). After examination of various databases with different sampling frequencies (range between 136–10 kHz), it has been concluded that in low sampling frequencies (less than 750 Hz), scales 2λ (λ = 1,2,…,5) are usable while for sampling frequencies more than 1,000 Hz, scales 2λ (λ = 1,2,…,8) contain profitable information that can be used for the purpose of wave detection, delineation and classification.
In Fig. 15, several dyadic scales obtained via application of à trous DWT to an arbitrary holter record belonging to the DAY general hospital database are illustrated. As it can be seen, by increment of the dyadic dilation parameter 2λ, the high-frequency fluctuations of the transform induced by measurement noises are diminished. The pseudo code associated with Eqs. 33–45 can be written in Matlab format as follows 
Rights and permissions
About this article
Cite this article
Homaeinezhad, M.R., Ghaffari, A., Toosi, H.N. et al. Optimal Delineation of Ambulatory Holter ECG Events via False-Alarm Bounded Segmentation of a Wavelet-Based Principal Components Analyzed Decision Statistic. Cardiovasc Eng 10, 136–156 (2010). https://doi.org/10.1007/s10558-010-9103-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10558-010-9103-2