Abstract
Purpose
Glucose intolerance (GI), defined as either prediabetes or diabetes, promotes cardiovascular events in patients with myocardial infarction (MI). Using the pooled clinical data from patients with MI and GI in the completed ABC and PPAR trials, we aimed to identify their clinical risk factors for cardiovascular events.
Methods
Using the limitless-arity multiple testing procedure, an artificial intelligence (AI)-based data mining method, we analyzed 415,328 combinations of < 4 clinical parameters.
Results
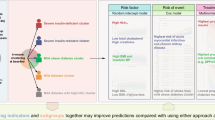
We identified 242 combinations that predicted the occurrence of hospitalization for (1) percutaneous coronary intervention for stable angina, (2) non-fatal MI, (3) worsening of heart failure (HF), and (4) all causes, and we analyzed combinations in 1476 patients. Among these parameters, the use of proton pump inhibitors (PPIs) or plasma glucose levels > 200 mg/dl after 2 h of a 75 g oral glucose tolerance test were linked to the coronary events of (1, 2). Plasma BNP levels > 200 pg/dl were linked to coronary and cardiac events of (1, 2, 3). Diuretics use, advanced age, and lack of anti-dyslipidemia drugs were linked to cardiovascular events of (1, 3). All of these factors were linked to (4). Importantly, each finding was verified by independently drawn Kaplan–Meier curves, indicating that the determined factors accurately affected cardiovascular events.
Conclusions
In most previous MI patients with GI, progression of GI, PPI use, or high plasma BNP levels were linked to the occurrence of coronary stenosis or recurrent MI. We emphasize that use of AI may comprehensively uncover the hidden risk factors for cardiovascular events.




Similar content being viewed by others
References
Braunwald E, Bristow MR. Congestive heart failure: fifty years of progress. Circulation. 2000;102(20 Suppl 4):Iv14–23.
Ambrosy AP, Fonarow GC, Butler J, Chioncel O, Greene SJ, Vaduganathan M, et al. The global health and economic burden of hospitalizations for heart failure: lessons learned from hospitalized heart failure registries. J Am Coll Cardiol. 2014;63(12):1123–33. https://doi.org/10.1016/j.jacc.2013.11.053.
Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339(4):229–34. https://doi.org/10.1056/nejm199807233390404.
Asakura M, Kim J, Asanuma H, Hamasaki T, Tsukahara K, Higashino Y, et al. Does Treatment of impaired glucose tolerance improve cardiovascular outcomes in patients with previous myocardial infarction? Cardiovasc Drugs Ther. 2017;31(4):401–11. https://doi.org/10.1007/s10557-017-6740-3.
Asakura M, Kim J, Asanuma H, Nakama Y, Tsukahara K, Higashino Y, et al. Cardiovascular outcomes in patients with previous myocardial infarction and mild diabetes mellitus following treatment with pioglitazone: reports of a randomised trial from the Japan Working Group for the Assessment Whether Pioglitazone Protects DM Patients Against Re-Infarction (PPAR Study). EClinicalMedicine. 2018;4-5:10–24. https://doi.org/10.1016/j.eclinm.2018.09.006.
Terada A, Okada-Hatakeyama M, Tsuda K, Sese J. Statistical significance of combinatorial regulations. Proc Natl Acad Sci U S A. 2013;110(32):12996–3001. https://doi.org/10.1073/pnas.1302233110.
Fukuda H, Shindo K, Sakamoto M, Ide T, Kinugawa S, Fukushima A, et al. Elucidation of the strongest predictors of cardiovascular events in patients with heart failure. EBioMedicine. 2018;33:185–95. https://doi.org/10.1016/j.ebiom.2018.06.001.
Hamet P, Tremblay J. Artificial intelligence in medicine. Metab Clin Exp. 2017;69s:S36–s40. https://doi.org/10.1016/j.metabol.2017.01.011.
Shindo K, Fukuda H, Hitsumoto T, Ito S, Kim J, Washio T, et al. Plasma BNP levels and diuretics use as predictors of cardiovascular events in patients with myocardial infarction and impaired glucose tolerance. Cardiovasc Drugs Ther. 2020;34(1):79–88. https://doi.org/10.1007/s10557-019-06922-9.
Dzau VJ, Antman EM, Black HR, Hayes DL, Manson JE, Plutzky J, et al. The cardiovascular disease continuum validated: clinical evidence of improved patient outcomes: part II: Clinical trial evidence (acute coronary syndromes through renal disease) and future directions. Circulation. 2006;114(25):2871–91. https://doi.org/10.1161/circulationaha.106.655761.
Yamasaki Y, Kawamori R, Matsushima H, Nishizawa H, Kodama M, Kubota M, et al. Asymptomatic hyperglycaemia is associated with increased intimal plus medial thickness of the carotid artery. Diabetologia. 1995;38(5):585–91. https://doi.org/10.1007/bf00400728.
Norhammar A, Tenerz A, Nilsson G, Hamsten A, Efendic S, Ryden L, et al. Glucose metabolism in patients with acute myocardial infarction and no previous diagnosis of diabetes mellitus: a prospective study. Lancet (London, England). 2002;359(9324):2140–4. https://doi.org/10.1016/s0140-6736(02)09089-x.
White HD, Chew DP. Acute myocardial infarction. Lancet (London, England). 2008;372(9638):570–84. https://doi.org/10.1016/s0140-6736(08)61237-4.
Fihn SD, Gardin JM, Abrams J, Berra K, Blankenship JC, Dallas AP, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012;60(24):e44–e164. https://doi.org/10.1016/j.jacc.2012.07.013.
Libby P, Aikawa M. Stabilization of atherosclerotic plaques: new mechanisms and clinical targets. Nat Med. 2002;8(11):1257–62. https://doi.org/10.1038/nm1102-1257.
Asakura M, Kim J, Asanuma H, Kitakaze M. Anti-diabetic drugs for secondary prevention of cardiovascular disease in mild diabetic and IGT patients: ABC study and PPAR study. Nihon Rinsho Jpn J Clin Med. 2010;68(5):887–91.
Singh R, Barden A, Mori T, Beilin L. Advanced glycation end-products: a review. Diabetologia. 2001;44(2):129–46.
Fujita M, Okuda H, Tsukamoto O, Asano Y, Hirata YL, Kim J, et al. Blockade of angiotensin II receptors reduces the expression of receptors for advanced glycation end products in human endothelial cells. Arterioscler Thromb Vasc Biol. 2006;26(10):e138–42. https://doi.org/10.1161/01.Atv.0000239569.99126.37.
Heitzer T, Schlinzig T, Krohn K, Meinertz T, Münzel T. Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation. 2001;104(22):2673–8.
Ghebremariam YT, LePendu P, Lee JC, Erlanson DA, Slaviero A, Shah NH, et al. Unexpected effect of proton pump inhibitors: elevation of the cardiovascular risk factor asymmetric dimethylarginine. Circulation. 2013;128(8):845–53. https://doi.org/10.1161/circulationaha.113.003602.
Goodman SG, Clare R, Pieper KS, Nicolau JC, Storey RF, Cantor WJ, et al. Association of proton pump inhibitor use on cardiovascular outcomes with clopidogrel and ticagrelor: insights from the platelet inhibition and patient outcomes trial. Circulation. 2012;125(8):978–86. https://doi.org/10.1161/circulationaha.111.032912.
Nakamura Y, Saitoh S, Takagi S, Ohnishi H, Chiba Y, Kato N, et al. Impact of abnormal glucose tolerance, hypertension and other risk factors on coronary artery disease. Circ J : official journal of the Japanese Circulation Society. 2007;71(1):20–5. https://doi.org/10.1253/circj.71.20.
Ogawa Y, Itoh H, Nakao K. Molecular biology and biochemistry of natriuretic peptide family. Clin Exp Pharmacol Physiol. 1995;22(1):49–53.
Omland T, Aakvaag A, Bonarjee VV, Caidahl K, Lie RT, Nilsen DW, et al. Plasma brain natriuretic peptide as an indicator of left ventricular systolic function and long-term survival after acute myocardial infarction. Comparison with plasma atrial natriuretic peptide and N-terminal proatrial natriuretic peptide. Circulation. 1996;93(11):1963–9.
Mega JL, Morrow DA, De Lemos JA, Sabatine MS, Murphy SA, Rifai N, et al. B-type natriuretic peptide at presentation and prognosis in patients with ST-segment elevation myocardial infarction: an ENTIRE-TIMI-23 substudy. J Am Coll Cardiol. 2004;44(2):335–9. https://doi.org/10.1016/j.jacc.2004.04.033.
Podgorelec V, Kokol P, Stiglic B, Rozman I. Decision trees: an overview and their use in medicine. J Med Syst. 2002;26(5):445–63.
Kim J, Washio T, Yamagishi M, Yasumura Y, Nakatani S, Hashimura K, et al. A novel data mining approach to the identification of effective drugs or combinations for targeted endpoints--application to chronic heart failure as a new form of evidence-based medicine. Cardiovasc Drugs Ther. 2004;18(6):483–9. https://doi.org/10.1007/s10557-004-6226-y.
Funding
Grants-in-Aid from the Ministry of Health, Labour and Welfare of Japan; Grants-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan; and Grants-in-Aid from the Japan Agency for Medical Research and Development.
Author information
Authors and Affiliations
Contributions
Study concept and design: Masafumi Kitakaze
Data collection, Kazuhiro Shindo, Hiroki Fukuda, Taturo Hitsumoto, Shin Ito, Jiyoong Kim
Data analysis: Yohei Miyashita, Takashi Washio
Figures and Tables: Hiroki Fukuda, Kazuhiro Shindo
Writing: Masafumi Kitakaze.
Corresponding author
Ethics declarations
Conflict of Interest
No relationship to industry for K.S., H.F, Y.M., J.K., S.I, and T.W. MK reports grants from the Japanese government, grants from Japan Heart Foundation, grants from Japan Agency for Medical Research and Development, personal fees from Daiichi-Sankyo, personal fees from Pfizer, grants and personal fees from Ono, personal fees from Bayer, grants and personal fees from Novartis, grants and personal fees from Boehringer, grants and personal fees from Tanabe-Mitsubishi, personal fees from Japan Medical Data Center, grants and personal fees from Takeda, and grants and personal fees from Astra Zeneca, outside the submitted work.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shindo, K., Fukuda, H., Hitsumoto, T. et al. Artificial Intelligence Uncovered Clinical Factors for Cardiovascular Events in Myocardial Infarction Patients with Glucose Intolerance . Cardiovasc Drugs Ther 34, 535–545 (2020). https://doi.org/10.1007/s10557-020-06987-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-06987-x