Abstract
Impaired aortic distensibility index (ADI) is associated with cardiovascular risk factors. This study evaluates the relation of ADI measured by computed tomographic angiography (CTA) with the severity of coronary atherosclerosis in subjects with suspected coronary artery disease (CAD). Two hundred and twenty-nine subjects,age 63 ± 9 years, 42% female, underwent coronary artery calcium (CAC) scanning and CTA, and their ADI and Framingham risk score (FRS) were measured. End-systolic and end-diastolic (ED) cross-sectional-area(CSA) of ascending-aorta (AAo) was measured 15-mm above the left-main coronary ostium. ADI was defined as: [(Δlumen-CSA)/(lumen-CSA in ED × systemic-pulse-pressure) × 103]. ADI measured by 2D-trans-thoracic echocardiography (TTE) was compared with CTA-measured ADI in 26 subjects without CAC. CAC was defined as 0, 1–100, 101–400 and 400+. CAD was defined as luminal stenosis 0, 1–49% and 50%+. There was an excellent correlation between CTA- and TTE-measured ADI (r 2 = 0.94, P = 0.0001). ADI decreased from CAC 0 to CAC 400+; similarly from FRS 1–9% to FRS 20% + (P < 0.05). After adjustment for risk factors, the relative risk for each standard deviation decrease in ADI was 1.66 for CAC 1–100, 2.26 for CAC 101–400 and 2.32 for CAC 400+ as compared to CAC 0; similarly, 2.36 for non-obstructive CAD and 2.67 for obstructive CAD as compared to normal coronaries. The area under the ROC-curve to predict significant CAD was 0.68 for FRS, 0.75 for ADI, 0.81 for CAC and 0.86 for the combination (P < 0.05). Impaired aortic distensibility strongly correlates with the severity of coronary atherosclerosis. Addition of ADI to CAC and traditional risk factors provides incremental value to predict at-risk individuals.
Similar content being viewed by others
Introduction
Vascular dysfunction has been shown to be an independent predictor of adverse cardiovascular events [1–3]. Impaired vascular function is associated with coronary artery disease [4], hypertension [5], diabetes [6, 7], and hypercholesterolemia [8–10].
Arterial distensibility, a measure of vascular function, can serve as a marker of coronary heart disease risk in humans [11, 12], In addition animal studies suggest that reduced arterial distensibility is an early sign of atherosclerotic change [13]. Previous studies revealed an inverse relationship between aortic distensibility and cardiovascular risk factors and may predict outcome [14–16].
The aorta maintains low left ventricular after-load, promotes optimal subendocardial coronary blood flow [17], and transforms pulsatile into more luminar blood flow. Impaired aortic distensibility leads to higher left ventricular systolic pressures, diminished subendocardial blood supply [17] and may ultimately contribute to left ventricular dysfunction [18, 19].
Coronary artery calcium (CAC) is a marker of overall atherosclerotic burden and correlates well with the total plaque burden, the presence of obstructive coronary artery disease (CAD) and future cardiovascular events in both genders across ethnicities [20–22]. Systemic vascular dysfunction measured through reactive hyperemia procedure is strongly correlated with increasing Framingham risk score (FRS) and CAC independent of age, sex, and traditional cardiac risk factors and is superior to FRS for the prediction of significant CAC [23].
This study evaluates whether impaired aortic distensibility index (ADI) is associated with the severity of coronary atherosclerosis in at-risk individuals.
Methods
Subjects and study design
Two hundred and twenty-nine consecutive subjects referred for coronary artery calcium (CAC) scanning and CTA for suspected coronary artery disease (CAD) were enrolled. Patients with established cardiovascular disease, stroke, diabetic retinopathy, Raynaud’s syndrome, infection, cancer, immunosuppression, systemic inflammation, advanced renal or liver disease, creatinine >1.4 mg/dl, and triglyceride >400 mg/dl were excluded. In a sub-study, 26 patients with normal coronaries and CAC 0 underwent 2D trans-thoracic echocardiography (TTE). The ascending aorta diameter and cross-sectional area measured by CTA and TTE were compared. Risk factors were determined, and FRS was calculated to assess the risk of developing total coronary disease events (angina, myocardial infarction, or cardiovascular death) over the next 10 years [24].
CAC scanning
The coronary arteries were imaged with 30–40 contiguous 3 mm slices during mid-diastole using ECG-triggering during a 15 s breath hold. CAC was considered present in a coronary artery when a density of >130 Hounsfield units (HU) was detected in ≥3 contiguous pixels (>1 mm2) overlying that coronary artery and quantified using the previously described Agatston scoring method [25].
Cardiac CTA
Beta blockers were administered for pulses greater than 65 bpm. CTA was performed according to standard clinical technique with retrospective ECG gating and ECG pulsing applied. Mean heart rate during the scan was 57 ± 2 bpm.
Data acquisition
A 64- Multi-detector Computed Tomography (MDCT) scanner (Lightspeed VCT, General Electric Healthcare Technologies, Milwaukee, WI) was used for all subjects. Imaging was started 1 inch above the left main ostium and continued to 1 inch below the bottom of the heart. The following imaging and reconstruction parameters were applied: data acquisition collimation 0.625 mm × 64 = 4 cm; 120 kVp; 220–670 mAs; pitch 0.18–0.24 (depending on heart rate); rotation time 0.35 s; slice width 0.625 mm; matrix 512 × 512 and pixel size 0.39 mm2. ECG-triggered dose modulation was applied in each case with 400–600 mA in 60–80% R–R interval and 250–350 mA for the rest of the cardiac cycle (81 to 59% of the next cycle).
Cardiac data were reconstructed from 5 to 95% of R–R interval and with 10% intervals. Coronary vessels were reviewed (AW Volume Share™, GE Medical Systems, Milwaukee, WI) and volume renderings and curved multi-planar reformations were done. CAC was defined as CAC 0, CAC 1–100, CAC 101–400 and CAC 400+. CAD was defined as normal coronaries, non-obstructive and obstructive CAD (luminal stenosis 1–49 and 50%, respectively). Two skilled cardiologists blinded to the clinical data measured the CAC and interpreted CTA separately.
Measurements
Cardiac CTA
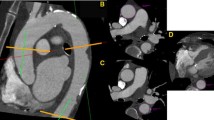
The principle of aortic distensibility measurement is illustrated in Fig. 1. The window level and width of 300 and 800 HU was used. True CSA and diameter of ascending aorta (AAo) was measured both manually and with the automated software (Workstation AW 4.4, GE Medical Systems, Milwaukee, WI) using the aligned and adjusted planes. The CSA and diameter of AAo were measurements 15 mm above the ostium of left main artery [26]. AAo diameter and CSA were measured from 5 to 95% of the R–R interval in 10% increments. Maximum AAo diameter and CSA was observed at end-systolic phase, 35% of R–R interval in individuals with a heart rate ≤60 bpm. AAo diameter and CSA decreased linearly from end-systole to end-diastole phases from 35 to 95% of R–R interval; which the minimum AAo diameter and CSA was observed at the end-diastolic (95% of R–R interval) phase.
Changes in the volume of ascending aorta (AAo) at the end systolic and end diastolic phase, 35% and 95% of R–R interval respectively, 15 mm above LM ostium using axial and coronal views. Using axial image, AAo image 15 mm above LM ostium was assessed (C), then perpendicular diameter and volume of AAo was measured using coronal and oblique views (B) at end systole and diastole, respectively
Consensus was reached with a third expert measurement in case of disagreements between the manual and automated measures (2 cases).
ADI was defined as: [ADI = (∆ lumen AAo CSA)/(end-diastole AAo CSA × systemic pulse pressure) × 103] [27, 28]. Δlumen AAo CSA is defined as: end-systolic AAo CSA − end-diastolic AAo CSA. Brachial artery blood pressure was measured before and after CTA acquisition, and the mean value was calculated.
Trans-thoracic echocardiography
In a sub-study, 26 subjects with normal coronaries and CAC 0 underwent TTE within a week after cardiac CTA. AAo diameter was measured 15 mm above sinotubular junction in the M-mode tracings, guided by the two-dimensional long parasternal axis [29] using Hewlett-Packard Sonos 5,500 echocardiographic system (Hewlett-Packard Corporation, Palo Alto, CA). Simultaneous recording of the ECG during TTE permitted evaluation of changes in AAo diameter during various phases of cardiac cycle, and reliable comparison with CTA measured AAo diameter. AAo cross section area (CSA) was calculated from 35%, end-systolic-, and 95%, end-diastolic-phase, of echocardiogram measured diameter with the assumption that the CSA is circular (area = (diameter/2)2 × (π)). TTE and CTA measured ADI was driven from calculated and measured AAo CSA, respectively.
Statistical analysis
Mean ± SD and proportions were used to summarize the characteristics of the study group. Continuous variables were compared by ANOVA, and categorical variables were compared by the Kruskal–Wallis multivariate analysis. Inter-observer and inter-modality (CTA vs. TTE) variability of AAo diameter measurement were calculated using intra-class correlation coefficient (ICC), and Bland & Altman plots [30]. Relative risk regression analyses were employed to assess the association between the severity of CAC and ADI, also the association between ADI and CAD before and after adjustment for conventional cardiovascular risk factors, as well as for Framingham risk score. ROC curves were constructed and the area under the ROC curve (AUC) was calculated to predict the ability of each model to detect a) significant CAC (CAC 100+), and B) significant CAD (obstructive CAD), respectively. All statistical analyses were performed with PASW version 18.0 (SPSS Institute Inc, Chicago, IL, http://www.spss.com). The level of significance was set at P < 0.05 (two-tailed). The study protocol was approved by the IRB Committee Board of the Los Angeles Biomedical Research Institute at Harbor UCLA, Torrance, California and all patients provided written informed consent.
Results
Aortic distensibility index (ADI)
CTA measured ADI was compared to the TTE measured ADI. There was excellent correlation between CTA and TTE measured ADI (r 2 = 0.94, ICC = 0.99, P = 0.0001). However, CTA measured ADI was 3.4% higher than TTE. (Figure 2a) There was excellent inter-observer agreement in measuring ADI using TTE and CTA. The inter-observer variability was higher with TTE than CTA (Fig. 2b).
Patient characteristics and aortic artery distensibility
Patients were characterized based upon the severity of CAC and placed into one of 4 groups (CAC 0, 1–100, 101–400 and 400+) (Table 1). There were no significant differences in gender, smoking status, diabetes mellitus, hypertension, hypercholesterolemia, lipid profile, and family history of premature CAD between the cohorts. The mean age was higher in patients with significant CAC as compared to normal cohort. The Framingham risk score increased substantially with increasing CAC. In a sub-study in subjects with CAC zero, ADI decreased with the metabolic status from normal cohorts to diabetes mellitus (P < 0.05).
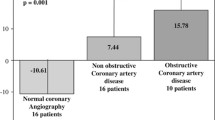
There was strong inverse correlation between ADI and CAC (r 2 = −0.69, P = 0.0001). ADI decreased proportionally from CAC 0 to CAC 1–100 to CAC 101–400 to CAC 400+ in both genders (Fig. 3a and b). Furthermore, severity of CAC increased incrementally in each FRS category as ADI decreased; similarly, in each ADI category as FRS increased. The highest CAC was noted with the combination of the lowest tertile of ADI and FRS 20%+; The lowest ADI was observed in subjects with highest CAC and FRS (Fig. 3b). ADI decreased with the severity of metabolic status in each CAC category in which diabetics with CAC 400+ had the least ADI. (Figure 3c) After adjustment for age, gender, conventional cardiac risk factors, and FRS by relative risk regression analyses, the risk of each standard deviation decrease in ADI was 1.66 for CAC 1–100, 2.26 for CAC 101–400 and 2.32 for CAC 400+ as compared to CAC 0. (Table 2) Multivariate regression analysis showed that ADI was an independent predictor of the severity of CAC.
Increasing severity of CAD was associated with increase in CAC and FRS, and decrease in ADI (Table 1). After adjustment for age, gender, conventional cardiac risk factors, and CAC, ADI was independent predictor of significant CAD. (Table 3).
ROC curves were constructed to assess the ability of cardiovascular risk factors and ADI to predict: a) significant CAC, and b) significant CAD. The AUC for proposed models is summarized in Fig. 4. Addition of ADI to FRS increased the area under the ROC curve to predict significant CAC significantly from 0.63 to 0.84 (P < 0.05) and provided the largest contributor to significant CAC prediction. Similarly, addition of ADI to CAC and traditional risk factors increased the area under the ROC curve to predict significant CAD from 0.68 to 0.86.
Discussion
The current study demonstrates that: 1. Excellent correlation exists between TTE and CTA measured ADI, 2- ADI decreased with the severity of metabolic status, 3. CTA measured ADI decreased proportionally with the severity of CAC independent of cardiovascular risk factors, and 4. Addition of ADI to FRS improved the ability to detect significant CAC from 0.63 to 0.84.
There is considerable heterogeneity in the magnitude of vascular dysfunction in individuals with similar risk factor profiles [31, 32]. In this regard, vascular dysfunction may be seen as an important “integrative factor” of the inherent atherosclerotic risk of an individual which takes into account the cumulative effects of various risk and protective factors [31]. In addition to improving risk assessment for the prediction of cardiovascular outcomes, another important application of vascular function assessment is to evaluate response to therapies [33–37].
Arterial distensibility serves as a marker of the structure and function of the artery (i.e. vascular function) and has been shown to diminish with advancing age and with increasing cardiovascular risk factors [38]. Recent studies have demonstrated that oxidative modification of LDL particles, and vascular wall remodeling induced by hyperglycemia may play a pathophysiological role in reducing arterial distensibility and revealed that aortic distensibility decreased with increasing cardiovascular risk factors [6, 8–10, 18, 19, 39].
Previous studies provided evidence that reduced arterial distensibility predicts future cardiovascular events [10, 15, 40]. Haluska et al. studied the association of arterial distensibility and long-term clinical outcome of 719 patients. They reported that impaired arterial distensibility independently correlated with poor outcome in patients with varying degrees of cardiovascular risk, and arterial distensibility can identify patients at risk of future fatal and non-fatal events [41]. Finally, recent studies have shown that functional changes in arteries precede the development of structural changes and also reverse more quickly in response to therapies [37, 42–44].
Clinical guidelines recommend risk management based on the Framingham score [45, 46] followed by goal-directed therapy when necessary [47–49] in patients without known prior CAD. Recent studies have showed that FRS has significant limitation in identifying high risk subjects without known CAD [50, 51]. Current findings revealed that CTA provides an accurate measure for vascular function rather than just anatomical studies; Furthermore, in this study aortic distensibility decreased substantially with the severity of coronary atherosclerosis in both genders. The excellent correlations between impaired aortic distensibility measured by CTA and FRS, and, in particular, the superior independent and incremental value of aortic distensibility for predicting significant coronary atherosclerosis, highlight the role of vascular function assessment as a part of cardiovascular risk assessment to improve identification of high risk individuals and optimizing patient management.
Limitations
Beta blockers which are vasoactive medications were administered for heart rates greater than 65 bpm. However, recent studies demonstrated that concomitant therapy with non-nitrate vasoactive drugs has no significant effect on vascular function assessment [52]. The effect of data acquisition and analysis during a breath-hold is another potential source of error. Nonetheless, the CTA measurements correlated well with the TTE measurements, suggesting that potential errors introduced by non-simultaneous blood pressures and breath-hold are minimal.
Conclusion
Aortic distensibility measured by CTA correlates well with corresponding values measured by TTE. Impaired aortic distensibility is associated with the severity of coronary atherosclerosis independent of age, gender, and cardiovascular risk factors. Furthermore, addition of ADI to traditional risk factors and CAC provided the largest contribution to detect significant coronary atherosclerosis, and highlights the diagnostic utility of ADI in the early detection and monitoring of patients with atherosclerotic cardiovascular disease by a single noninvasive diagnostic study.
References
Fuster V, Badimon L, Badimon JJ, Chesebro JH (1992) The pathogenesis of coronary artery disease and the acute coronary syndromes (1). N Engl J Med 326(4):242–250
Griendling KK, Ushio-Fukai M, Lassegue B, Alexander RW (1997) Angiotensin II signaling in vascular smooth muscle. New concepts. Hypertension 29(1 Pt 2):366–373
Vane JR, Anggard EE, Botting RM (1990) Regulatory functions of the vascular endothelium. N Engl J Med 323(1):27–36
Bogren HG, Mohiaddin RH, Klipstein RK, Firmin DN, Underwood RS, Rees SR, Longmore DB (1989) The function of the aorta in ischemic heart disease: a magnetic resonance and angiographic study of aortic compliance and blood flow patterns. Am Heart J 118(2):234–247
Laurent S, Caviezel B, Beck L, Girerd X, Billaud E, Boutouyrie P, Hoeks A, Safar M (1994) Carotid artery distensibility and distending pressure in hypertensive humans. Hypertension 23(6 Pt 2):878–883
Eren M, Gorgulu S, Uslu N, Celik S, Dagdeviren B, Tezel T (2004) Relation between aortic stiffness and left ventricular diastolic function in patients with hypertension, diabetes, or both. Heart 90(1):37–43
Lehmann ED, Gosling RG, Sonksen PH (1992) Arterial wall compliance in diabetes. Diabet Med 9(2):114–119
Dart AM, Lacombe F, Yeoh JK, Cameron JD, Jennings GL, Laufer E, Esmore DS (1991) Aortic distensibility in patients with isolated hypercholesterolaemia, coronary artery disease, or cardiac transplant. Lancet 338(8762):270–273
Lehmann ED, Watts GF, Gosling RG (1992) Aortic distensibility and hypercholesterolaemia. Lancet 340(8828):1171–1172
Oliver JJ, Webb DJ (2003) Noninvasive assessment of arterial stiffness and risk of atherosclerotic events. Arterioscler Thromb Vasc Biol 23(4):554–566
Bank AJ, Wang H, Holte JE, Mullen K, Shammas R, Kubo SH (1996) Contribution of collagen, elastin, and smooth muscle to in vivo human brachial artery wall stress and elastic modulus. Circulation 94(12):3263–3270
Sudhir K, Mullen WL, Hausmann D, Fitzgerald PJ, Chou TM, Yock PG, Chatterjee K (1995) Contribution of endothelium-derived nitric oxide to coronary arterial distensibility: an in vivo two-dimensional intravascular ultrasound study. Am Heart J 129(4):726–732
Hironaka K, Yano M, Kohno M, Tanigawa T, Obayashi M, Konishi M, Umemoto S, Matsuzaki M (1997) In vivo aortic wall characteristics at the early stage of atherosclerosis in rabbits. Am J Physiol 273(3 Pt 2):H1142–H1147
Tentolouris N, Liatis S, Moyssakis I, Tsapogas P, Psallas M, Diakoumopoulou E, Voteas V, Katsilambros N (2003) Aortic distensibility is reduced in subjects with type 2 diabetes and cardiac autonomic neuropathy. Eur J Clin Invest 33(12):1075–1083
Cruickshank K, Riste L, Anderson SG, Wright JS, Dunn G, Gosling RG (2002) Aortic pulse-wave velocity and its relationship to mortality in diabetes and glucose intolerance: an integrated index of vascular function? Circulation 106(16):2085–2090
Kingwell BA, Waddell TK, Medley TL, Cameron JD, Dart AM (2002) Large artery stiffness predicts ischemic threshold in patients with coronary artery disease. J Am Coll Cardiol 40(4):773–779
Belz GG (1995) Elastic properties and windkessel function of the human aorta. Cardiovasc Drugs Ther 9(1):73–83
Kelly RP, Tunin R, Kass DA (1992) Effect of reduced aortic compliance on cardiac efficiency and contractile function of in situ canine left ventricle. Circ Res 71(3):490–502
Ohtsuka S, Kakihana M, Watanabe H, Sugishita Y (1994) Chronically decreased aortic distensibility causes deterioration of coronary perfusion during increased left ventricular contraction. J Am Coll Cardiol 24(5):1406–1414
Detrano R, Guerci AD, Carr JJ, Bild DE, Burke G, Folsom AR, Liu K, Shea S, Szklo M, Bluemke DA, O’Leary DH, Tracy R, Watson K, Wong ND, Kronmal RA (2008) Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med 358(13):1336–1345
Brown ER, Kronmal RA, Bluemke DA, Guerci AD, Carr JJ, Goldin J, Detrano R (2008) Coronary calcium coverage score: determination, correlates, and predictive accuracy in the Multi-Ethnic Study of Atherosclerosis. Radiology 247(3):669–675
Budoff MJ, Nasir K, McClelland RL, Detrano R, Wong N, Blumenthal RS, Kondos G, Kronmal RA (2009) Coronary calcium predicts events better with absolute calcium scores than age-sex-race/ethnicity percentiles: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol 53(4):345–352
Ahmadi N, Hajsadeghi F, Gul K, Vane J, Usman N, Flores F, Nasir K, Hecht H, Naghavi M, Budoff M (2008) Relations between digital thermal monitoring of vascular function, the Framingham risk score, and coronary artery calcium score. J Cardiovasc Comput Tomogr 2(6):382–388
Wilson PW, D’Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB (1998) Prediction of coronary heart disease using risk factor categories. Circulation 97(18):1837–1847
Becker CR, Knez A, Jakobs TF, Aydemir S, Becker A, Schoepf UJ, Bruening R, Haberl R, Reiser MF (1999) Detection and quantification of coronary artery calcification with electron-beam and conventional CT. Eur Radiol 9(4):620–624
Mao S, Lu B, Takasu J, Oudiz RJ, Budoff MJ (2003) Measurement of the RT interval on ECG records during electron-beam CT. Acad Radiol 10(6):638–643
McLeod AL, Newby DE, Northridge DB, Fox KA, Uren NG (2003) Influence of differential vascular remodeling on the coronary vasomotor response. Cardiovasc Res 59(2):520–526
Yamagishi M, Umeno T, Hongo Y, Tsutsui H, Goto Y, Nakatani S, Miyatake K (1997) Intravascular ultrasonic evidence for importance of plaque distribution (eccentric vs circumferential) in determining distensibility of the left anterior descending artery. Am J Cardiol 79(12):1596–1600
Stefanadis C, Stratos C, Boudoulas H, Kourouklis C, Toutouzas P (1990) Distensibility of the ascending aorta: comparison of invasive and non-invasive techniques in healthy men and in men with coronary artery disease. Eur Heart J 11(11):990–996
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1(8476):307–310
Halcox JP, Schenke WH, Zalos G, Mincemoyer R, Prasad A, Waclawiw MA, Nour KR, Quyyumi AA (2002) Prognostic value of coronary vascular endothelial dysfunction. Circulation 106(6):653–658
Quyyumi AA (2003) Prognostic value of endothelial function. Am J Cardiol 91(12A):19H–24H
Binggeli C, Spieker LE, Corti R, Sudano I, Stojanovic V, Hayoz D, Luscher TF, Noll G (2003) Statins enhance postischemic hyperemia in the skin circulation of hypercholesterolemic patients: a monitoring test of endothelial dysfunction for clinical practice? J Am Coll Cardiol 42(1):71–77
Celermajer DS (2003) Statins, skin, and the search for a test of endothelial function. J Am Coll Cardiol 42(1):78–80
Charbonneau F, Anderson TJ, Title L, Jobin J, Poirier P, Huyhn T, Chan S, Walling A, Hutchison S, Tran T, Lonn E, Buithieu J, Genest J (2008) Modulation of arterial reactivity using amlodipine and atorvastatin measured by ultrasound examination (MARGAUX). Atherosclerosis 197(1):420–427
Stulc T, Kasalova Z, Prazny M, Vrablik M, Skrha J, Ceska R (2003) Microvascular reactivity in patients with hypercholesterolemia: effect of lipid lowering treatment. Physiol Res 52(4):439–445
Warnholtz A, Wild P, Ostad MA, Elsner V, Stieber F, Schinzel R, Walter U, Peetz D, Lackner K, Blankenberg S, Munzel T (2008) Effects of oral niacin on endothelial dysfunction in patients with coronary artery disease: results of the randomized, double-blind, placebo-controlled INEF study. Atherosclerosis
van Popele NM, Grobbee DE, Bots ML, Asmar R, Topouchian J, Reneman RS, Hoeks AP, van der Kuip DA, Hofman A, Witteman JC (2001) Association between arterial stiffness and atherosclerosis: the Rotterdam study. Stroke 32(2):454–460
Sun H, Zhong M, Miao Y, Ma X, Gong HP, Tan HW, Zhang Y, Zhang W (2009) Impaired elastic properties of the aorta in fat-fed, streptozotocin-treated rats. Vascular remodeling in diabetic arteries. Cardiology 114(2):107–113
Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L, Ducimetiere P, Benetos A (2001) Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 37(5):1236–1241
Haluska BA, Jeffries L, Carlier S, Marwick TH (2009) Measurement of arterial distensibility and compliance to assess prognosis. Atherosclerosis
Shechter M, Issachar A, Marai I, Koren-Morag N, Freinark D, Shahar Y, Shechter A, Feinberg MS (2009) Long-term association of brachial artery flow-mediated vasodilation and cardiovascular events in middle-aged subjects with no apparent heart disease. Int J Cardiol 134(1):52–58
Papathanassiou K, Naka KK, Kazakos N, Kanioglou C, Makriyiannis D, Pappas K, Katsouras CS, Liveris K, Kolettis T, Tsatsoulis A, Michalis LK (2009) Pioglitazone vs glimepiride: differential effects on vascular endothelial function in patients with type 2 diabetes. Atherosclerosis 205(1):221–226
Ostad MA, Eggeling S, Tschentscher P, Schwedhelm E, Boger R, Wenzel P, Meinertz T, Munzel T, Warnholtz A (2009) Flow-mediated dilation in patients with coronary artery disease is enhanced by high dose atorvastatin compared to combined low dose atorvastatin and ezetimibe: results of the CEZAR study. Atherosclerosis 205(1):227–232
Wood D, De Backer G, Faergeman O, Graham I, Mancia G, Pyorala K (1998) Prevention of coronary heart disease in clinical practice. Summary of recommendations of the second joint task force of European and other societies on coronary prevention. J Hypertens 16(10):1407–1414
Fedder DO, Koro CE, L’Italien GJ (2002) New National Cholesterol Education Program III guidelines for primary prevention lipid-lowering drug therapy: projected impact on the size, sex, and age distribution of the treatment-eligible population. Circulation 105(2):152–156
Third Report of the National Cholesterol Education Program (NCEP) (2002) Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation 106(25):3143–3421
Grundy SM, Cleeman JI, Merz CN, Brewer HB Jr, Clark LT, Hunninghake DB, Pasternak RC, Smith SC Jr, Stone NJ (2004) Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 110(2):227–239
Smith SC Jr, Greenland P, Grundy SM (2000) AHA conference proceedings. Prevention conference V: beyond secondary prevention: identifying the high-risk patient for primary prevention: executive summary. American heart association. Circulation 101(1):111–116
Khot UN, Khot MB, Bajzer CT, Sapp SK, Ohman EM, Brener SJ, Ellis SG, Lincoff AM, Topol EJ (2003) Prevalence of conventional risk factors in patients with coronary heart disease. JAMA 290(7):898–904
Scridon T, Novaro GM, Bush HS, Asher CR, Dandes E, Kabirdas D, Scridon C, Kuo BT, Whiteman M, Shen MY (2008) Reclassification of patients for aggressive cholesterol treatment: additive value of multislice coronary angiography to the National Cholesterol Education Program guidelines. Clin Cardiol 31(9):419–423
Gokce N, Holbrook M, Hunter LM, Palmisano J, Vigalok E, Keaney JF Jr, Vita JA (2002) Acute effects of vasoactive drug treatment on brachial artery reactivity. J Am Coll Cardiol 40(4):761–765
Disclosures
Matthew Budoff has a financial disclosure in relationship to this study, as he is on the speaker’s bureau for General Electric, whereas other authors have no conflicts to disclose.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ahmadi, N., Nabavi, V., Hajsadeghi, F. et al. Impaired aortic distensibility measured by computed tomography is associated with the severity of coronary artery disease. Int J Cardiovasc Imaging 27, 459–469 (2011). https://doi.org/10.1007/s10554-010-9680-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-010-9680-6