Abstract
Background Incomplete stent apposition (ISA) at follow-up has been reported to be more common after drug-eluting stent (DES) implantation than after bare-metal stent (BMS) implantation. The aim of this study was to use intravascular ultrasound (IVUS) to evaluate the coronary characteristics after drug-eluting stent implantation in patients with ISA at follow-up. Methods From the IVUS database of our institute, a total of 89 patients with 125 native lesions who underwent DES implantation into de novo lesions with IVUS imaging at 6-month follow-up were identified, and 15 (16.9%) patients had documented ISA at follow-up by IVUS. The ISA group was compared with a matched control group of patients (n = 30) who had no evidence of ISA at follow-up. Results Of the 15 documented ISA at follow-up after DES implantation, two located at the edge (within 5 mm from stent margin) while 13 in the body of the stent. The maximum area and arc of ISA measured 5.3 ± 2.2 mm2 and 163 ± 67°, respectively. In patients with ISA, the maximum EEM area of stent segment with ISA was significantly larger than the adjacent stent segment without ISA (24.1 ± 3.3 vs. 20.1 ± 3.1 mm2, P = 0.002), while stent area, plaque plus media (P&M) area and intrastent lumen area were comparable (P > 0.05). Compared to the matched control cohort without ISA at follow-up, the maximum EEM area was also significantly larger (24.1 ± 3.3 vs. 18.8 ± 4.2 mm2, P < 0.001), while the areas of reference EEM and lumen, stent, P&M behind the stent, intimal hyperplasia and intrastent lumen were all comparable between the two groups (P > 0.05). Conclusion ISA at follow-up after DES implantation for de novo coronary lesions was associated with a larger EEM area.
Similar content being viewed by others
Introduction
A large amount of randomized studies have demonstrated that drug-eluting stent (DES) dramatically reduced in-stent restenosis and target-vessel revascularization by effectively inhibiting neointimal hyperplasia [1–9]. However, incomplete stent apposition (ISA) at follow-up, an unusual intravascular ultrasound (IVUS) finding, was reported to be more common after DES implantation than after bare-metal stent (BMS) implantation [9–13]. Previous studies have demonstrated ISA after BMS implantation was associated with positive coronary remodeling [14–16]. Nevertheless, the effects of DESs on the vessel wall have not been fully investigated in humans using intravascular ultrasound (IVUS). In the present study, we sought to use IVUS to evaluate the coronary characteristics after drug-eluting stent implantation in patients with versus without ISA at follow-up.
Methods
Patient selection
From the Zhongshan Hospital core IVUS database, a total of 89 patients with 125 native lesions who underwent DES implantation into de novo lesions with IVUS imaging at 6-month follow-up were identified, and 15 (16.9%) patients had documented ISA at follow-up by IVUS. The ISA group was compared with a matched control group of patients (n = 30) who had no evidence of ISA at follow-up (the matched group was selected to be two times the size of the ISA group). Matching criteria included: (1) identical target coronary artery; (2) identical diabetes status; (3) identical DES type; and (4) similar reference external elastic membrane (EEM) cross-sectional area. All patients underwent de novo DES implantation (without adjunct brachytherapy) into a native coronary artery not in the setting of acute myocardial infarction.
Before catheterization, all patients were pre-medicated with 300 mg of aspirin, which was continued with a dose of 100 mg/day for a life-long time. Antithrombotic regimens, including intravenous heparin, low molecular weight heparin and glycoprotein IIb/IIIa inhibitors, were administered at each operator’s discretion. A loading dose of 300 mg of clopidogrel was also administered before intervention, and clopidogrel 75 mg/day was recommended for at least 6 months after stenting.
IVUS imaging and analysis
IVUS imaging were performed after intracoronary administration of 0.2 mg of nitroglycerin with a motorized transducer pullback system and a commercially available scanner (SCIMED) that consisted of a rotating 30- or 40-MHz transducer within a 3.2 F or 3.0 F imaging sheath. The imaging catheter was advanced 10 mm distal to the stent, and the transducer was withdrawn at 0.5 mm/s back to the guiding catheter. All studies were recorded on 0.5-inch high-resolution s-VHS videotape for subsequent analysis.
Qualitative analysis involved review of all IVUS tapes for the presence of ISA at follow-up. ISA was defined as separation of at least 1 stent strut from the intimal surface of the arterial wall, not overlapping a side branch, with evidence of blood flow (speckling) behind the strut.
Using computerized planimetry, we performed quantitative IVUS analysis at (1) ISA sections, (2) stented segments with complete late apposition, and (3) reference segments. Quantitative measurements included EEM, lumen, and stent area at stented and reference segments; and EEM, stent, plaque plus media (P&M), intrastent lumen, intimal hyperplasia, and ISA area at ISA sections [17]. ISA area was measured every 1 mm within ISA sections.
Statistical analysis
Statistical analysis was performed with the SPSS 11.0 software program (SPSS Inc). Continuous variables are presented as mean ± SD and compared with unpaired Student t-test or Mann–Whitney U-test. A P value < 0.05 was considered statistically significant.
Results
ISA was documented in 15 patients with 15 lesions: 12 (80%) with sirolimus-eluting stents and 3 (20%) with paclitaxel-eluting stents. The baseline clinical and angiographic characteristics are shown in Table 1. All baseline clinical, angiographic and postprocedural characteristics were comparable between ISA patients and matched control cohort. And no dissection or thrombus was observed by angiogram after stenting. The coronary angiogram of patients with ISA at follow-up showed different extent of irregularity in the segment of stent, with contrast media filling outside the stent strut appearing as “Zig-Zag” like silhouette, which had not been observed in the post-procedural angiogram. Typical angiographic images of ISA at follow-up are shown in Fig. 1. All patients with ISA and matched control patients are asymptomatic at 6-month follow-up.
Angiographic and IVUS pictures of LAD in a patient with incomplete stent apposition at 6-month follow-up after sirolimus-eluting stent implantation. The stent segment is shown at higher magnification (A) and longitudinal IVUS image showed significant incomplete stent apposition (B). The maximum EEM area of the stent segment with ISA (E) was significantly larger than that of the proximal (D) and distal adjacent stent segment without ISA (F), and also larger than that of the proximal (C) and distal reference (G)
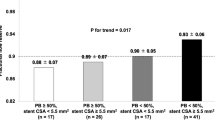
In the patients with ISA at follow-up after DES implantation, IVUS finding comparing stent segments with ISA and the adjacent stent segments without ISA are shown in Table 2. And the typical IVUS images of ISA at follow-up are also shown in Fig. 1. The location of ISA was at the edge (within 5 mm from stent margin) in 2 (13%) lesions and within the body of the stent in 13 (87%) lesions. The maximum area and arc of ISA measured 5.3 ± 2.2 mm2 and 163 ± 67°, respectively. Compared with the stent segment without ISA, the maximum EEM area and ratio of EEM area between stented segment and reference were significantly larger in stent segment with ISA (24.1 ± 3.3 vs. 20.1 ± 3.1 mm2, P = 0.002; 1.6 ± 0.2 vs. 1.3 ± 0.2, P = 0.001, respectively) while stent area, P&M area and intrastent lumen area were comparable in the different segments (P > 0.05).
IVUS findings comparing patients with ISA at follow-up and the matched control cohort are shown in Table 3. The distal and proximal reference area of EEM and lumen, stent area were very similar between ISA and the matched control group (P > 0.05). However, the maximum EEM area and ratio of EEM area between stented segment and reference in ISA group were significantly larger than that in the matched control group (24.1 ± 3.3 vs. 18.8 ± 4.2 mm2, P < 0.001; 1.6 ± 0.2 vs. 1.3 ± 0.2, P < 0.001, respectively). There was no significant difference in P&M area behind the stent between the two groups, as well as the areas of intimal hyperplasia and intrastent lumen.
Discussion
The major findings of the present study are the following: (1) In patients with ISA 6 months post DES implantation, the maximum EEM area in stent segment with ISA was significantly larger than that without ISA. (2) Compared to the matched control group without ISA, the EEM area of ISA segment was significant larger in patients with ISA at follow-up. (3) There was no difference in P&M area behind the stent and inhibition of in-stent neointimal proliferation between the two groups with and without ISA. In other words, ISA 6 months after DES implantation was associated with a larger EEM area but without difference in plaque mass and in-stent neointimal proliferation.
Postulated mechanisms of ISA at follow-up after DES implantation include: (1) incomplete apposition that is not recognized at the time of implantation and only detected at follow-up; (2) a decrease in plaque with or without any change in EEM; (3) chronic stent recoil in the absence of any change in vessel wall dimensions; and (4) an increase in EEM that either occurs in the absence of an increase in plaque or that is greater than the increase in plaque. Previous studies has reported that unrecognized ISA after implantation accounted a small part of total ISA at follow-up [14] and half of them resolved at 6-months [18]. Moreover, some serial IVUS studies have virtually excluded the presence of chronic stent recoil and the decrease in plaque after DES implantation [10, 11, 19]. In the current analysis, the separation of the intima from the stent correlated with the enlargement of EEM areas. Therefore, the mechanism of ISA at follow-up after DES implantation may be caused by an increase of EEM occurring in the setting of no increase in plaque mass. This finding is similar to previous studies of ISA after BMS implantation [14–16].
Using serial volumetric IVUS, Degertekin et al. [19] reported that DES didn’t affect vessel volume and plaque behind the stent at follow-up. It was somewhat different from our finding in the present analysis. However, ISA is a focal arterial phenomenon, and vessel enlargement occurring within the region of ISA. The regional changes may not been well detected when changes in total EEM volumes were assessed. It may not be appropriate to assess mechanisms of ISA (a focal phenomenon) by measuring changes in total arterial or plaque volume, even if these are the “standard” analyses performed in IVUS laboratories.
The pathophysiologic mechanism of vessel enlargement and following ISA after DES implantation remains uncertain. It could be proposed to be associated with specific hypersensitivity to the metallic stent, polymer or drug (sirolimus or paclitaxel). Because of the lack of information regarding the respective vascular responses with each component of drug-eluting stent in the use of these devices during percutaneous coronary intervention, the definite mechanism of vessel enlargement associated with ISA after DES implantation should be further investigated.
Previous studies have suggested that ISA after DES implantation was not associated with any adverse event, compared with non-ISA groups [9–12, 20, 21]. However, considering the increasing reports of late stent thrombosis of DES in real-world patients, particularly after the discontinuation of double antiplatelet therapy, [22–31] special attention should be paid on the patients with ISA at follow-up after DES implantation. It needs to be emphasized and aggressively studied to evaluate whether ISA would be the potential mechanism of late in-stent thrombosis. Prolonged dual antiplatelet drug therapy with aspirin and clopidogrel may be important, though so far no study could tell us how long it should be. Since no lumen compromise exists and patients are clinically asymptomatic, the significance of angiography and IVUS follow-up after DES implantation should be emphasized.
Study limitations
This was a retrospective, observational analysis from a single center and the number of patients with ISA was small. This was not a serial IVUS study, and the relationship between the vascular remodeling at baseline procedure and that at follow-up was not evaluated. Plaque composition data, which might affect the expansion of stents and the redistribution of plaque, was not available in this study. IVUS virtual histology (VH) prior to stenting in prospective studies with routine IVUS follow-up will answer the question whether plaque composition plays a significant role in this field. In the present study, an angiographic “zig-zag” like silhouette might have triggered unscheduled IVUS examination at follow-up and lead to an unavoidable selection bias and the result might be an over-representation of cases with both ISA and angiographic lumen irregularities.
References
Sousa JE, Costa MA, Abizaid AC et al (2001) Sustained suppression of neointimal proliferation by sirolimus-eluting stents: one-year angiographic and intravascular ultrasound follow-up. Circulation 104:2007–2011
Serruys PW, Degertekin M, Tanabe K et al (2002) Intravascular ultrasound findings in the multicenter, randomized, double-blind RAVEL (RAndomized study with the sirolimus-eluting VElocity balloon expandable stent in the treatment of patients with de novo native coronary artery Lesions) trial. Circulation 106:798–803
Moses JW, Leon MB, Popma JJ et al (2003) Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med 349:1315–1323
Sousa JE, Costa MA, Sousa AG et al (2003) Two-year angiographic and intravascular ultrasound follow-up after implantation of sirolimus-eluting stents in human coronary arteries. Circulation 107:381–383
Colombo A, Drzewiecki J, Banning A et al (2003) Randomized study to assess the effectiveness of slow- and moderate-release polymer-based paclitaxel-eluting stents for coronary artery lesions. Circulation 108:788–794
Tanabe K, Serruys PW, Degertekin M et al (2004) Chronic arterial responses to polymer-controlled paclitaxel-eluting stents: comparison with bare metal stents by serial intravascular ultrasound analyses: data from the randomized TAXUS-II trial. Circulation 109:196–200
Weissman NJ, Koglin J, Cox DA et al (2005) Polymer-based paclitaxel-eluting stents reduce in-stent neointimal tissue proliferation: a serial volumetric intravascular ultrasound analysis from the TAXUS-IV trial. J Am Coll Cardiol 45:1201–1205
Zhang F, Ge J, Qian J et al (2007) Sirolimus-eluting stents in real-world patients with ST-segment elevation acute myocardial infarction. Int Heart J 48: 303–311
Aoki J, Colombo A, Dudek D et al (2005) Peristent remodeling and neointimal suppression two years after polymer-based, paclitaxel-eluting stent implantation. Insights from serial intravascular ultrasound analysis in the TAXUS-II study. Circulation 112:3876–3883
Ako J, Morino Y, Honda Y et al (2005) Late incomplete stent apposition after sirolimus-eluting stent implantation a serial intravascular ultrasound analysis. J Am Coll Cardiol 46(6):1002–1005
Hong MK, Mintz GS, Lee CW et al (2006) Late stent malapposition after drug-eluting stent implantation: an intravascular ultrasound analysis with long-term follow-up. Circulation 113(3):414–419 Jan 24
Mintz GS, Weissman NJ, Dawkins K et al (2006) Frequency, predictors, and clinical outcomes of late-acquired incomplete stent apposition in patients treated with Taxus stents a volumetric intravascular ultrasound meta-analysis from the TAXUS IV, V, and VI trials (abstr). J Am Coll Cardiol 47(Suppl B):26B
Zhang F, Qian JY, Ge JB et al (2006) Late stent malapposition and marked positive vessel remodeling after sirolimus-eluting coronary stent implantation. Chin Med J 119(4):349–350
Shah VM, Mintz GS, Apple S et al (2002) Background incidence of late malapposition after bare-metal stent implantation. Circulation 106:1753–1755
Nakamura M, Kataoka T, Honda Y et al (2003) Late incomplete stent apposition and focal vessel expansion after bare metal stenting. Am J Cardiol 92:1217–1219
Hong MK, Mintz GS, Lee CW et al (2004) Incidence, mechanism, predictors, and long-term prognosis of late stent malapposition after bare-metal stent implantation. Circulation 109:881–886
Mintz GS, Nissen SE, Anderson WD et al (2001) American College of Cardiology clinical expert consensus document on standards for acquisition, measurement and reporting of Intravascular Ultrasound Studies (IVUS). A report of the American College of Cardiology task force on clinical expert consensus documents. J Am Coll Cardiol 37:1478–1492
Tanabe K, Serruys PW, Degertekin M et al (2005) Incomplete stent apposition after implantation of paclitaxel-eluting stents or bare metal stents: insights from the randomized TAXUS II trial. Circulation. 111(7):900–905
Degertekin M, Regar E, Tanabe K et al (2003) Evaluation of coronary remodeling after sirolimus-eluting stent implantation by serial three-dimensional intravascular ultrasound. Am J Cardiol 91(9):1046–1050
Degertekin M, Serruys PW, Tanabe K et al (2003) Long-term follow-up of incomplete stent apposition in patients who received sirolimus-eluting stent for de novo coronary lesions: an intravascular ultrasound analysis. Circulation 108:2747–2750
Zhang F, Qian JY, Ge JB (2007) Rapid development of late stent malappositon and coronary aneurysm following implantation of a paclitaxel-eluting coronary stent. Chin Med J 120:614–616
McFadden EP, Stabile E, Regar E et al (2004) Late thrombosis in drug-eluting coronary stents after discontinuation of antiplatelet therapy, Lancet 364:1519–1521
Virmani R, Guagliumi G, Farb A et al (2004) Localized hypersensitivity and late coronary thrombosis secondary to a sirolimus-eluting stent: should we be cautious? Circulation 109:701–705
Ong AT, Hoye A, Aoki J et al (2005) Thirty day incidence and six-month clinical outcome of thrombotic stent occlusion after bare metal, sirolimus, or paclitaxel stent implantation. J Am Coll Cardiol 45:947–953
Iakovou I, Schmidt T, Bonizzoni E et al (2005) Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 293:2126–2130
Acar G, Dogan A, Altinbas A et al (2006) Recurrent acute stent thrombosis associated with protein C and S deficiencies. Int J Cardiovasc Imaging 22:333–337
Breuckmann F, Nassenstein K, Boese D et al (2006) Successful nitinol stent implantation in a large coronary aneurysm: post-interventional patency assessment by magnetic resonance imaging. Int J Cardiovasc Imaging 22:501–505
Takano M, Mizuno K (2006) Late coronary thrombosis in a sirolimus-eluting stent due to the lack of neointimal coverage. Eur Heart J 27:1133
Henderson D, Gunalingam B (2006) Very late stent thrombosis of a sirolimus-eluting stent. Catheter Cardiovasc Interv 68:406–408
Makaryus AN, Lefkowitz L, Lee AD (2007) Coronary artery stent fracture. Int J Cardiovasc Imaging 23:305–309
Kuchulakanti PK, Chu WW, Torguson R et al (2006) Correlates and long-term outcomes of angiographically proven stent thrombosis with sirolimus- and paclitaxel-eluting stents. Circulation 113:1108–1113
Acknowledgments
This study was supported by the young scientist fund from Shanghai municipal health bureau (2006Y15).
Author information
Authors and Affiliations
Corresponding author
Additional information
Juying Qian, Feng Zhang contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Qian, J., Zhang, F., Wu, H. et al. Comparison of intravascular ultrasonic imaging with versus without incomplete stent apposition at follow-up after drug-eluting stent implantation. Int J Cardiovasc Imaging 24, 133–139 (2008). https://doi.org/10.1007/s10554-007-9249-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-007-9249-1