Abstract
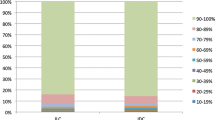
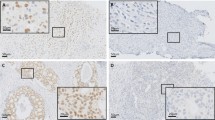
The great majority of invasive lobular carcinoma (ILC) is estrogen-dependent luminal A type carcinoma but the details of estrogen actions and its intratumoral metabolism have not been well studied compared to invasive ductal carcinoma (IDC). We first immunolocalized estrogen-related enzymes including estrogen sulfotransferase (EST), estrogen sulfatase (STS), 17β-hydroxysteroid dehydrogenase (HSD) 1/2, and aromatase. We then evaluated the tissue concentrations of estrogens in ILC and IDC and subsequently estrogen-responsive gene profiles in these tumors in order to explore the possible differences and/or similarity of intratumoral estrogen environment of these two breast cancer subtypes. The status of STS and 17βHSD1 was significantly lower in ILCs than IDCs (p = 0.022 and p < 0.0001), but that of EST and 17βHSD2 vice versa (p < 0.0001 and p = 0.0106). In ILCs, tissue concentrations of estrone and estradiol were lower than those in IDCs (p = 0.0709 and 0.069). In addition, the great majority of estrogen response genes tended to be lower in ILCs. Among those genes above, FOXP1 was significantly higher in ILCs than in IDCs (p = 0.002). FOXP1 expression was reported to be significantly higher in relapse-free IDC patients treated with tamoxifen. Therefore, tamoxifen may be considered an option of endocrine therapy for luminal A type ILC patients. This is the first study to demonstrate the detailed and comprehensive status of intratumoral production and metabolism of estrogens and the status of estrogen response genes in luminal A-like ILC with comparison to those in luminal A-like IDCs.




Similar content being viewed by others
References
Fisher ER, Gregorio RM, Fisher B et al (1975) The pathology of invasive breast cancer. A syllabus derived from findings of the National Surgical Adjuvant Breast Project (protocol no. 4). Cancer 36(1):1–85
Arpino G, Bardou VJ, Clark GM, Elledge RM (2004) Infiltrating lobular carcinoma of the breast: tumor characteristics and clinical outcome. Breast Cancer Res 6(3):R149–R156
Sastre-Garau X, Jouve M, Asselain B et al (1996) Infiltrating lobular carcinoma of the breast. Clinicopathologic analysis of 975 cases with reference to data on conservative therapy and metastatic patterns. Cancer 77(1):113–120
Li CI, Uribe DJ, Daling JR (2005) Clinical characteristics of different histologic types of breast cancer. Br J Cancer 93(9):1046–1052
Pestalozzi BC, Zahrieh D, Mallon E et al (2008) Distinct clinical and prognostic features of infiltrating lobular carcinoma of the breast: combined results of 15 International Breast Cancer Study Group clinical trials. J Clin Oncol 26(18):3006–3014
Coradini D, Pellizzaro C, Veneroni S, Ventura L, Daidone MG (2002) Infiltrating ductal and lobular breast carcinomas are characterised by different interrelationships among markers related to angiogenesis and hormone dependence. Br J Cancer 87(10):1105–1111
Cocquyt VF, Blondeel PN, Depypere HT et al (2003) Different responses to preoperative chemotherapy for invasive lobular and invasive ductal breast carcinoma. Eur J Surg Oncol 29(4):361–367
Mersin H, Yildirim E, Gulben K, Berberoglu U (2003) Is invasive lobular carcinoma different from invasive ductal carcinoma? Eur J Surg Oncol 29(4):390–395
Suzuki T, Miki Y, Moriya T et al (2007) In situ production of sex steroids in human breast carcinoma. Med Mol Morphol 40(3):121–127
Suzuki T, Nakata T, Miki Y et al (2003) Estrogen sulfotransferase and steroid sulfatase in human breast carcinoma. Cancer Res 63(11):2762–2770
Pasqualini JR, Chetrite GS (1999) Estrone sulfatase versus estrone sulfotransferase in human breast cancer: potential clinical applications. J Steroid Biochem Mol Biol 69(1–6):287–292
Suzuki T, Miki Y, Nakamura Y et al (2005) Sex steroid-producing enzymes in human breast cancer. Endocr Relat Cancer 12(4):701–720
Yoda T, McNamara KM, Miki Y et al (2014) Intratumoral androgen metabolism and actions in invasive lobular carcinoma of the breast. Cancer Sci 105(11):1503–1509
Sasano H, Suzuki T, Nakata T, Moriya T (2006) New development in intracrinology of breast carcinoma. Breast Cancer 13(2):129–136
Miksits M, Wlcek K, Svoboda M et al (2010) Expression of sulfotransferases and sulfatases in human breast cancer: impact on resveratrol metabolism. Cancer Lett 289(2):237–245
Ahmed M, Esposito E (2015) Report from the 37th San Antonio Breast Cancer Symposium, 9–13th December 2014, Texas, USA. Ecancermedicalscience 9:508
Metzger OG-HA, Mallon E, Viale G et al (2015) Relative effectiveness of letrozole compared with tamoxifen for patients with lobular carcinoma in the BIG 1-98 trial. J Clin Oncol 33(25):2772–2779
Ciriello G, Gatza ML, Beck AH et al (2015) Comprehensive molecular portraits of invasive lobular breast cancer. Cell 163(2):506–519
Goldhirsch A, Winer EP, Coates AS et al (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2013. Ann Oncol 24(9):2206–2223
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19(5):403–410
Nakagawa S, Miki Y, Miyashita M et al (2016) Tumor microenvironment in invasive lobular carcinoma: possible therapeutic targets. Breast Cancer Res Treat 155(1):65–75
Hammond ME, Hayes DF, Dowsett M et al (2010) American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28(16):2784–2795
Wolff AC, Hammond ME, Schwartz JN et al (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25(1):118–145
Miki Y, Suzuki T, Tazawa C et al (2007) Aromatase localization in human breast cancer tissues: possible interactions between intratumoral stromal and parenchymal cells. Cancer Res 67(8):3945–3954
Sasano H, Miki Y, Nagasaki S, Suzuki T (2009) In situ estrogen production and its regulation in human breast carcinoma: from endocrinology to intracrinology. Pathol Int 59(11):777–789
Shibuya R, Suzuki T, Miki Y et al (2008) Intratumoral concentration of sex steroids and expression of sex steroid-producing enzymes in ductal carcinoma in situ of human breast. Endocr Relat Cancer 15(1):113–124
Ijichi N, Shigekawa T, Ikeda K et al (2012) Association of double-positive FOXA1 and FOXP1 immunoreactivities with favorable prognosis of tamoxifen-treated breast cancer patients. Horm Cancer 3(4):147–159
Shigekawa T, Ijichi N, Ikeda K et al (2011) FOXP1, an estrogen-inducible transcription factor, modulates cell proliferation in breast cancer cells and 5-year recurrence-free survival of patients with tamoxifen-treated breast cancer. Horm Cancer 2(5):286–297
Masuda M, Miki Y, Hata S et al (2012) An induction of microRNA, miR-7 through estrogen treatment in breast carcinoma. J Transl Med 10(Suppl 1):S2
Picard N, Caron V, Bilodeau S et al (2012) Identification of estrogen receptor β as a SUMO-1 target reveals a novel phosphorylated sumoylation motif and regulation by glycogen synthase kinase 3β. Mol Cell Biol 2(14):2709–2721
Urano T, Saito T, Tsukui T et al (2002) Efp targets 14-3-3 sigma for proteolysis and promotes breast tumour growth. Nature 417(6891):871–875
Mo R, Zhu YT, Tony YJ, Zhang Z, Rao SM (2007) GAS6 is an estrogen-inducible gene in mammary epithelial cells. Biochem Biophys Res Commun. 353(1):189–194
Seksenyan A, Kadavallore A, Walts AE et al (2015) TOX3 is expressed in mammary ER+ epithelial cells and regulates ER target genes in luminal breast cancer. BMC Cancer 15(1):22
Suzuki T, Urano T, Tsukui T et al (2005) Estrogen-responsive finger protein as a new potential biomarker for breast cancer. Clin Cancer Res 11(17):6148–6154
McCormack O, Chung WY, Fitzpatrick P et al (2008) Growth arrest-specific gene 6 expression in human breast cancer. Br J Cancer 98(6):1141–1146
Nagasaki S, Suzuki T, Miki Y et al (2009) 17Beta-hydroxysteroid dehydrogenase type 12 in human breast carcinoma: a prognostic factor via potential regulation of fatty acid synthesis. Cancer Res 69(4):1392–1399
Takagi K, Miki Y, Nagasaki S (2010) Increased intratumoral androgens in human breast carcinoma following aromatase inhibitor exemestane treatment. Endocr Relat Cancer 17(2):415–430
Bourdeau V, Deschênes J, Métivier R et al (2004) Genome-wide identification of high-affinity estrogen response elements in human and mouse. Mol Endocrinol 18(6):1411–1427
Turashvili G, Bouchal J, Baumforth K et al (2007) Novel markers for differentiation of lobular and ductal invasive breast carcinomas by laser microdissection and microarray analysis. BMC Cancer 7:55
Lin X, Li J, Yin G et al (2013) Integrative analyses of gene expression and DNA methylation profiles in breast cancer cell line models of tamoxifen-resistance indicate a potential role of cells with stem-like properties. Breast Cancer Res 15(6):R119
Jung SY, Jeong J, Shin SH et al (2010) The invasive lobular carcinoma as a prototype luminal A breast cancer: a retrospective cohort study. BMC Cancer 10:664
Arpino G, Bardou VJ, Clark GM, Elledge RM (2004) Infiltrating lobular carcinoma of the breast: tumor characteristics and clinical outcome. Breast Cancer Res 6(3):R149–R156
Mersin H, Yildirim E, Gülben K, Berberoğlu U (2003) Is invasive lobular carcinoma different from invasive ductal carcinoma? Eur J Surg Oncol 29(4):390–395
Berx G, Cleton-Jansen AM, Strumane K et al (1996) E-cadherin is inactivated in a majority of invasive human lobular breast cancers by truncation mutations throughout its extracellular domain. Oncogene 13(9):1919–1925
Rakha EA, Patel A, Powe DG et al (2010) Clinical and biological significance of E-cadherin protein expression in invasive lobular carcinoma of the breast. Am J Surg Pathol 34(10):1472–1479
Fernández B, Paish EC, Green AR et al (2011) Lymph-node metastases in invasive lobular carcinoma are different from those in ductal carcinoma of the breast. J Clin Pathol 64(11):995–1000
Nilsson UW, Garvin S, Dabrosin C (2007) MMP-2 and MMP-9 activity is regulated by estradiol and tamoxifen in cultured human breast cancer cells. Breast Cancer Res Treat 102(3):253–261
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts of interest in any of the author.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10549_2016_3739_MOESM1_ESM.pptx
Supplemental Fig. 1. Relationship between intratumoral estrone concentration and intracrine-related enzymes in ILC and IDC. Expressions of intracrine-related enzymes were evaluated by immunohistochemistry. 17βHSD1 was not detected in IDC. Supplemental Fig. 2. Relationship between intratumoral estradiol concentration and intracrine-related enzymes in ILC and IDC. Expressions of intracrine-related enzymes were evaluated by immunohistochemistry. 17βHSD1 was not detected in IDC. Supplementary material 1 (PPTX 83 kb)
Rights and permissions
About this article
Cite this article
Takagi, M., Miki, Y., Miyashita, M. et al. Intratumoral estrogen production and actions in luminal A type invasive lobular and ductal carcinomas. Breast Cancer Res Treat 156, 45–55 (2016). https://doi.org/10.1007/s10549-016-3739-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-016-3739-6