Abstract
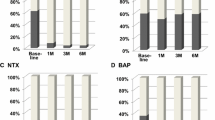
It is speculated that adjuvant use of bisphosphonate reduces recurrence in breast cancer patients through suppression of bone resorption. To determine the prognostic impact of bone resorption markers, we investigated serum levels of the pyridinoline crosslinked carboxyterminal telopeptide of type I collagen (1CTP) and N-terminal crosslinking telopeptides of type I collagen (NTX). 1CTP and NTX were measured at baseline (before operation or neoadjuvant therapies) and afterward in 469 patients operated on breast cancer. The optimal cutoff value of 1CTP for relapse-free survival (RFS) was set at 3.6 ng/ml with an area under the receiver operating characteristics curve of 0.641 [95 % confidence interval (CI) = 0.560–0.721; p = 0.0011]. However, we were unable to determine a significant cutoff value for NTX. RFS was significantly worse for 1CTP-high patients with than for those with low levels of 1CTP (p = 0.0002). Multivariate analysis with tumor size, lymph node metastasis, and nuclear grade showed that 1CTP was a significant independent prognostic factor (hazard ratio = 2.04, 95 % CI = 1.13–3.68; p = 0.018). Worse prognosis for the subset with high 1CTP levels applied only to postmenopausal patients (p = 0.0002). RFS of 130 patients whose 1CTP changed from low at baseline to high at 6 months postoperatively showed RFS almost as poor as that for patients with high 1CTP throughout. These findings suggest that 1CTP may be useful not only for identifying patients with unfavorable prognosis, but also for selecting patients who may benefit from administration of bone-modifying agents in an adjuvant setting.





Similar content being viewed by others
References
Rogers MJ, Gordon S, Benford HL, Coxon FP, Luckman SP, Monkkonen J, Frith JC (2000) Cellular and molecular mechanisms of action of bisphosophonates. Cancer 88:2961–2978
Bekker PJ, Holloway DL, Rasmussen AS, Murphy R, Martin SW, Leese PT, Holmes GB, Dunstan CR, DePaoli AM (2005) A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. 2004. J Bone Miner Res 20:2275–2282
Lipton A (2011) Bones, breasts and bisphosphonates; rationale for the use of zoledronic acid in advanced and early breast cancer. Breast Cancer 3:1–7. doi:10.2147/BCTT.S16774
Coleman R, de Boer R, Eidtmann H, Llombart A, Davidson N, Neven P, von Minckwitz G, Sleeboom HP, Forbes J, Barrios C, Frassoldati A, Campbell I et al (2013) Zoledronic acid (zoledronate) for postmenopausal women with early breast cancer receiving adjuvant letrozole (ZO-FAST study): final 60-month results. Ann Oncol 24:398–405. doi:10.1093/annonc/mds277
Brufsky AM, Harker WG, Beck JT, Bosserman L, Vogel C, Seidler C, Jin L, Warsi G, Argonza-Aviles E, Hohneker J, Ericson SG, Perez EA (2012) Final 5-year results of Z-FAST trial: adjuvant zoledronic acid maintains bone mass in postmenopausal breast cancer patients receiving letrozole. Cancer 118:1192–1201. doi:10.1002/cncr.26313
Gnant M, Mlineritsch B, Stoeger H, Luschin-Ebengreuth G, Knauer M, Moik M, Jakesz R, Seifert M, Taucher S, Bjelic-Radisic V, Balic M, Eidtmann H et al (2015) Zoledronic acid combined with adjuvant endocrine therapy of tamoxifen versus anastrozole plus ovarian function suppression in premenopausal early breast cancer: final analysis of the Austrian Breast and Colorectal Cancer Study Group Trial 12. Ann Oncol 26:313–320. doi:10.1093/annonc/mdu544
Coleman R, Cameron D, Dodwell D, Bell R, Wilson C, Rathbone E, Keane M, Gil M, Burkinshaw R, Grieve R, Barrett-Lee P, Ritchie D et al (2014) Adjuvant zoledronic acid in patients with early breast cancer: final efficacy analysis of the AZURE (BIG 01/04) randomised open-label phase 3 trial. Lancet Oncol 15:997–1006. doi:10.1016/S1470-2045(14)70302-X
He M, Fan W, Zhang X (2013) Adjuvant zoledronic acid therapy for patients with early stage breast cancer: an updated systematic review and meta-analysis. J Hematol Oncol 6:80. doi:10.1186/1756-8722-6-80
Coleman RE, Marshall H, Cameron D, Dodwell D, Burkinshaw R, Keane M, Gil M, Houston SJ, Grieve RJ, Barrett-Lee PJ, Ritchie D, Pugh J et al (2011) Breast-cancer adjuvant therapy with zoledronic acid. N Engl J Med 365:1396–1405. doi:10.1056/NEJMoa1105195
Mundy GR (1997) Mechanisms of bone metastasis. Cancer 80:1546–1556
Goldhirsch A, Glick JH, Gelber RD, Coates AS, Thürlimann B, Senn HJ, Panel Members (2005) Meeting highlights: international expert consensus on the primary therapy of early breast cancer 2005. Ann Oncol 16:1569–1583
Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thürlimann B, Senn HJ (2007) Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. 10th St Gallen conference. Ann Oncol 18:1133–1144
Goldhirsch A, Ingle JN, Gelber RD, Coates AS, Thürlimann B, Senn HJ, Panel Members (2009) Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Ann Oncol 20:1319–1329. doi:10.1093/annonc/mdp322
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thürlimann B, Senn HJ, Panel Members (2011) Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer. Ann Oncol 22:1736–1747. doi:10.1093/annonc/mdr304
Keskikuru R, Bloigu R, Risteli J, Kataja V, Jukkola A (2002) Elevated preoperative serum ICTP is a prognostic factor for overall and disease-free survival in breast cancer. Oncol Rep 9:1323–1327
Riethmüller G, Klein CA (2001) Early cancer cell dissemination and late metastatic relapse: clinical reflections and biological approaches to the dormancy problem in patients. Semin Cancer Biol 11:307–311
Dunn LK, Mohammad KS, Fournier PG, McKenna CR, Davis HW, Niewolna M, Peng XH, Chirgwin JM, Guise TA (2009) Hypoxia and TGF-beta drive breast cancer bone metastases through parallel signaling pathways in tumor cells and the bone microenvironment. PLoS ONE 4:e6896. doi:10.1371/journal.pone.0006896
Neudert M, Fischer C, Krempien B, Bauss F, Seibel MJ (2003) Site-specific human breast cancer (MDA-MB-231) metastases in nude rats: model characterisation and in vivo effects of ibandronate on tumour growth. Int J Cancer 107:468–477
Gnant M, Mlineritsch B, Luschin-Ebengreuth G, Kainberger F, Kässmann H, Piswanger-Sölkner JC, Seifert M, Ploner F, Menzel C, Dubsky P, Fitzal F, Bjelic-Radisic V, Steger G, Greil R, Marth C, Kubista E, Samonigg H, Wohlmuth P, Mittlböck M, Jakesz R, Austrian Breast and Colorectal Cancer Study Group (ABCSG) (2008) Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 5-year follow-up of the ABCSG-12 bone-mineral density substudy. Lancet Oncol 9:840–849. doi:10.1016/S1470-2045(08)70204-3
Eastell R, Adams J, Clack G, Howell A, Cuzick J, Mackey J, Beckmann MW, Coleman RE (2011) Long-term effects of anastrozole on bone mineral density: 7-year results from the ATAC trial. Ann Oncol 22:857–862. doi:10.1093/annonc/mdq541
Vehmanen LK, Elomaa I, Blomqvist CP, Saarto T (2014) The effect of ovarian dysfunction on bone mineral density in breast cancer patients 10 years after adjuvant chemotherapy. Acta Oncol 53:75–79. doi:10.3109/0284186X.2013.792992
Cheleuitte D, Mizuno S, Glowacki J (1998) In vitro secretion of cytokines by human bone marrow: effects of age and estrogen status. J Clin Endocrinol Metab 83:2043–2051
Bismar H, Diel I, Ziegler R, Pfeilschifter J (1995) Increased cytokine secretion by human bone marrow cells after menopause or discontinuation of estrogen replacement. J Clin Endocrinol Metab 80:3351–3355
Pfeilschifter J, Diel I, Scheppach B, Bretz A, Krempien R, Erdmann J, Schmid G, Reske N, Bismar H, Seck T, Krempien B, Ziegler R (1998) Concentration of transforming growth factor beta in human bone tissue: relationship to age, menopause, bone turnover, and bone volume. J Bone Miner Res 13:716–730
Lipton A, Chapman JA, Demers L, Shepherd LE, Han L, Wilson CF, Pritchard KI, Leitzel KE, Ali SM, Pollak M (2011) Elevated bone turnover predicts for bone metastasis in postmenopausal breast cancer: results of NCIC CTG MA.14. J Clin Oncol 29:3605–3610. doi:10.1200/JCO.2010.31.5069
Pectasides D, Farmakis D, Nikolaou M, Kanakis I, Kostopoulou V, Papaconstantinou I, Karamanos NK, Economopoulos T, Raptis SA (2005) Diagnostic value of bone remodeling markers in the diagnosis of bone metastases in patients with breast cancer. J Pharm Biomed Anal 37:171–176
Kiuchi K, Ishikawa T, Hamaguchi Y, Momiyama N, Hasegawa S, Ishiyama A, Kono T, Doi T, Chishima T, Shimada H (2002) Crosslinked collagen C- and N-telopeptides for an early diagnosis of bone metastasis from breast cancer. Oncol Rep 9:595–598
Greenspan SL, Dresner-Pollak R, Parker RA, London D, Ferguson L (1997) Diurnal variation of bone mineral turnover in elderly men and women. Calcif Tissue Int 60:419–423
Ju HS, Leung S, Brown B, Stringer MA, Leigh S, Scherrer C, Shepard K, Jenkins D, Knudsen J, Cannon R (1997) Comparison of analytical performance and biological variability of three bone resorption assays. Clin Chem 43:1570–1576
Acknowledgments
The present study was supported by the grant of Hyogo College of Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors of this manuscript declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10549_2015_3638_MOESM1_ESM.pptx
Supplementary material 1 (PPTX 74 kb). (Supplemental figure) Receiver operating characteristics (ROC) curves for 1CTP and NTX for relapse-free survival. The cutoff values were set at 3.6 ng/ml for 1CTP and 14.05 nmol BCE/L for NTX with the areas under the curves (AUC), that is, 0.641 (95 % confidence interval (CI) = 0.560-0.721, p = 0.0011) for 1CTP and 0.549 (95 % CI = 0.461-0.637, p = 0.27) for NTX
Rights and permissions
About this article
Cite this article
Imamura, M., Nishimukai, A., Higuchi, T. et al. High levels at baseline of serum pyridinoline crosslinked carboxyterminal telopeptide of type I collagen are associated with worse prognosis for breast cancer patients. Breast Cancer Res Treat 154, 521–531 (2015). https://doi.org/10.1007/s10549-015-3638-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3638-2