Abstract
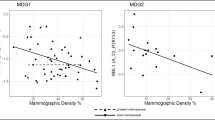
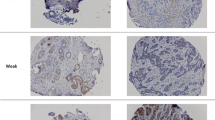
High mammographic density is associated with a increased risk of breast cancer. We hypothesized that specific pathways exist that are associated with increased mammographic density, and may therefore be used to identify potential targets for chemoprevention. Histologically confirmed normal breast tissue was collected from women undergoing breast surgery who had available demographic data and mammograms for review. Women with low versus high mammographic breast density were compared. Differentially expressed genes using Affymetrix HG U133Plus2 chips were identified in dense versus non-dense tissue. Immunohistochemical analysis (IHC) of estrogen receptor, progesterone receptor, Ki67, and COX2 expression was performed. About 66 women were identified, 28 (42%) had high, and 38 (58%) had low mammographic density. About 73 genes had differential expression between normal breast tissue with high and low mammographic density (P < 0.001, fold change ≥1.5with a low false discovery rate (<10%). Network and canonical pathway analysis indicated decreased TGFβ signaling (TGFBR2, SOS, SMAD3, CD44 and TNFRSF11B) in dense breast tissue relative to non-dense breast. By IHC, only COX2 expression in the stroma was statistically significant on multivariate analysis. TGFβ ligands are currently the only growth factors known to prevent mammary epithelial cell proliferation. TGFβ signaling has been reported to be inhibited by COX-2, and these molecules are highly differentially expressed in individuals at high risk of developing breast cancer. These results strongly suggest that COX2 inhibition should be investigated for breast cancer prevention despite possible increase in cardiovascular risk.



Similar content being viewed by others
References
Atkinson C, Warren R, Bingham SA, Day NE (1999) Mammographic patterns as a predictive biomarker of breast cancer risk: effect of tamoxifen. Cancer Epidemiol Biomarkers Prev 8(10):863–866
Boyd NF, Byng JW, Jong RA, Fishell EK, Little LE, Miller AB et al (1995) Quantitative classification of mammographic densities and breast cancer risk: results from the Canadian National Breast Screening Study. J Natl Cancer Inst 87(9):670–675. doi:10.1093/jnci/87.9.670
Boyd NF, Dite GS, Stone J, Gunasekara A, English DR, McCredie MR et al (2002) Heritability of mammographic density, a risk factor for breast cancer. N Engl J Med 347(12):886–894. doi:10.1056/NEJMoa013390
Boyd NF, Guo H, Martin LJ, Sun L, Stone J, Fishell E et al (2007) Mammographic density and the risk and detection of breast cancer. N Engl J Med 356(3):227–236. doi:10.1056/NEJMoa062790
Boyd NF, Lockwood GA, Martin LJ, Byng JW, Yaffe MJ, Tritchler DL (2001) Mammographic density as a marker of susceptibility to breast cancer: a hypothesis. IARC Sci Publ 154:163–169
Boyd NF, Lockwood GA, Martin LJ, Knight JA, Jong RA, Fishell E et al (1999) Mammographic densities and risk of breast cancer among subjects with a family history of this disease. J Natl Cancer Inst 91(16):1404–1408. doi:10.1093/jnci/91.16.1404
Boyd NF, Martin LJ, Li Q, Sun L, Chiarelli AM, Hislop G et al (2006) Mammographic density as a surrogate marker for the effects of hormone therapy on risk of breast cancer. Cancer Epidemiol Biomarkers Prev 15(5):961–966. doi:10.1158/1055-9965.EPI-05-0762
Byng JW, Yaffe MJ, Jong RA, Shumak RS, Lockwood GA, Tritchler DL et al (1998) Analysis of mammographic density and breast cancer risk from digitized mammograms. Radiographics 18(6):1587–1598
Harvey JA, Bovbjerg VE (2004) Quantitative assessment of mammographic breast density: relationship with breast cancer risk. Radiology 230(1):29–41. doi:10.1148/radiol.2301020870
Warwick J, Pinney E, Warren RM, Duffy SW, Howell A, Wilson M et al (2003) Breast density and breast cancer risk factors in a high-risk population. Breast 12(1):10–16. doi:10.1016/S0960-9776(02)00212-6
Chen J, Pee D, Ayyagari R, Graubard B, Schairer C, Byrne C et al (2006) Projecting absolute invasive breast cancer risk in white women with a model that includes mammographic density. J Natl Cancer Inst 98(17):1215–1226
Cuzick J, Warwick J, Pinney E, Warren RM, Duffy SW (2004) Tamoxifen and breast density in women at increased risk of breast cancer. J Natl Cancer Inst 96(8):621–628
Fisher B, Costantino JP, Wickerham DL, Redmond CK, Kavanah M, Cronin WM et al (1998) Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst 90(18):1371–1388. doi:10.1093/jnci/90.18.1371
Freedman M, San Martin J, O’Gorman J, Eckert S, Lippman ME, Lo SC et al (2001) Digitized mammography: a clinical trial of postmenopausal women randomly assigned to receive raloxifene, estrogen, or placebo. J Natl Cancer Inst 93(1):51–56. doi:10.1093/jnci/93.1.51
Tiersten A, Ng YY, Pile-Spellman E, Nelsen C, Noguera-Irizarry W, Brafman R et al (2004) Relationship between mammographic breast density and tamoxifen in women with breast cancer. Breast J 10(4):313–317. doi:10.1111/j.1075-122X.2004.21332.x
Goss PE, Strasser-Weippl K (2004) Prevention strategies with aromatase inhibitors. Clin Cancer Res 10(1 Pt 2):372S–379S. doi:10.1158/1078-0432.CCR-031210
Chlebowski RT, McTiernan A (2003) Biological significance of interventions that change breast density. J Natl Cancer Inst 95(1):4–5
van Gils CH, Hendriks JH, Holland R, Karssemeijer N, Otten JD, Straatman H et al (1999) Changes in mammographic breast density and concomitant changes in breast cancer risk. Eur J Cancer Prev 8(6):509–515. doi:10.1097/00008469-199912000-00006
van Gils CH, Otten JD, Hendriks JH, Holland R, Straatman H, Verbeek AL (1999) High mammographic breast density and its implications for the early detection of breast cancer. J Med Screen 6(4):200–204
BI-RADS (2003)BI-RADS. American College of Radiology, Reston, VA
Chang JC, Wooten EC, Tsimelzon A, Hilsenbeck SG, Gutierrez MC, Elledge R et al (2003) Gene expression profiling for the prediction of therapeutic response to docetaxel in patients with breast cancer. Lancet 362(9381):362–369. doi:10.1016/S0140-6736(03)14023-8
Pavlidis P, Qin J, Arango V, Mann JJ, Sibille E (2004) Using the gene ontology for microarray data mining: a comparison of methods and application to age effects in human prefrontal cortex. Neurochem Res 29(6):1213–1222. doi:10.1023/B:NERE.0000023608.29741.45
Forrester E, Chytil A, Bierie B, Aakre M, Gorska AE, Sharif-Afshar AR et al (2005) Effect of conditional knockout of the type II TGF-beta receptor gene in mammary epithelia on mammary gland development and polyomavirus middle T antigen induced tumor formation and metastasis. Cancer Res 65(6):2296–2302. doi:10.1158/0008-5472.CAN-04-3272
Thirunavukkarasu K, Miles RR, Halladay DL, Yang X, Galvin RJ, Chandrasekhar S et al (2001) Stimulation of osteoprotegerin (OPG) gene expression by transforming growth factor-beta (TGF-beta). Mapping of the OPG promoter region that mediates TGF-beta effects. J Biol Chem 276(39):36241–36250. doi:10.1074/jbc.M104319200
Shipitsin M, Campbell LL, Argani P, Weremowicz S, Bloushtain-Qimron N, Yao J et al (2007) Molecular definition of breast tumor heterogeneity. Cancer Cell 11(3):259–273. doi:10.1016/j.ccr.2007.01.013
Yu Q, Stamenkovic I (2004) Transforming growth factor-beta facilitates breast carcinoma metastasis by promoting tumor cell survival. Clin Exp Metastasis 21(3):235–242. doi:10.1023/B:CLIN.0000037705.25256.d3
Studer RK, Chu CR (2005) p38 MAPK and COX2 inhibition modulate human chondrocyte response to TGF-beta. J Orthop Res 23(2):454–461. doi:10.1016/j.orthres.2004.08.012
Uttamsingh S, Bao X, Nguyen KT, Bhanot M, Gong J, Chan JL et al (2008) Synergistic effect between EGF and TGF-beta1 in inducing oncogenic properties of intestinal epithelial cells. Oncogene 27(18):2626–2634. doi:10.1038/sj.onc.1210915
Liu CH, Chang SH, Narko K, Trifan OC, Wu MT, Smith E et al (2001) Overexpression of cyclooxygenase-2 is sufficient to induce tumorigenesis in transgenic mice. J Biol Chem 276(21):18563–18569. doi:10.1074/jbc.M010787200
Subbaramaiah K, Hudis C, Chang SH, Hla T, Dannenberg AJ (2008) EP2 and EP4 receptors regulate aromatase expression in human adipocytes and breast cancer cells. Evidence of a BRCA1 and p300 exchange. J Biol Chem 283(6):3433–3444. doi:10.1074/jbc.M705409200
Chang MC, Wu HL, Lee JJ, Lee PH, Chang HH, Hahn LJ et al (2004) The induction of prostaglandin E2 production, interleukin-6 production, cell cycle arrest, and cytotoxicity in primary oral keratinocytes and KB cancer cells by areca nut ingredients is differentially regulated by MEK/ERK activation. J Biol Chem 279(49):50676–50683. doi:10.1074/jbc.M404465200
Chang SH, Liu CH, Conway R, Han DK, Nithipatikom K, Trifan OC et al (2004) Role of prostaglandin E2-dependent angiogenic switch in cyclooxygenase 2-induced breast cancer progression. Proc Natl Acad Sci USA 101(2):591–596. doi:10.1073/pnas.2535911100
Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer J et al (2000) COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer 89(12):2637–2645. doi:10.1002/1097-0142(20001215)89:12≤2637::AID-CNCR17≥3.0.CO;2-B
Schreinemachers DM, Everson RB (1994) Aspirin use and lung, colon, and breast cancer incidence in a prospective study. Epidemiology 5(2):138–146. doi:10.1097/00001648-199403000-00003
Huls G, Koornstra JJ, Kleibeuker JH (2003) Non-steroidal anti-inflammatory drugs and molecular carcinogenesis of colorectal carcinomas. Lancet 362(9379):230–232. doi:10.1016/S0140-6736(03)13915-3
Steinbach G, Lynch PM, Phillips RK, Wallace MH, Hawk E, Gordon GB et al (2000) The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N Engl J Med 342(26):1946–1952. doi:10.1056/NEJM200006293422603
Senior K (2005) COX-2 inhibitors: cancer prevention or cardiovascular risk? Lancet Oncol 6(2):68. doi:10.1016/S1470-2045(05)01720-1
Dannenberg AJ, Altorki NK, Boyle JO, Dang C, Howe LR, Weksler BB et al (2001) Cyclo-oxygenase 2: a pharmacological target for the prevention of cancer. Lancet Oncol 2(9):544–551. doi:10.1016/S1470-2045(01)00488-0
Acknowledgments
This study was funded by P50CA116199 grant from the National Cancer Institute (GNH, with a career development award to WTY). This study was supported in part by the Breast Cancer Research Foundation (JCC), the Emma Jacobs Clinical Breast Cancer Fund (JCC), the NCI Breast Cancer SPORE P50 CA50183 (JCC, MTL),1 R01 CA112305-01 from the National Cancer Institute (JCC), P30 CA125123 from the National Institute of Health (JCC).
Author information
Authors and Affiliations
Corresponding author
Additional information
Wei Tse Yang and Michael T. Lewis are the co-first authors.
Aysegul Sahin and Jenny C. Chang are the co-senior authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, W.T., Lewis, M.T., Hess, K. et al. Decreased TGFβ signaling and increased COX2 expression in high risk women with increased mammographic breast density. Breast Cancer Res Treat 119, 305–314 (2010). https://doi.org/10.1007/s10549-009-0350-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0350-0