Abstract
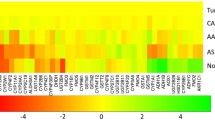
The complex role of drug metabolism-related enzymes and their possible influence in cancer development, treatment and outcome has not yet been completely elucidated. There is evidence that these enzymes can activate certain environmental procarcinogens to more toxic derivatives and thus a role has been proposed for them in carcinogenesis. The fact that they can also inactivate a number of chemotherapeutic drugs has raised the possibility of these enzymes influencing the sensitivity of tumour cells to anticancer agents. In this report, we analyse the expression of drug metabolism-related genes within a whole genome microarray study of 104 breast cancer and 17 normal breast specimens. Kaplan–Meier survival curves, Chi-squared, and Cox Regression analyses were used to identify associations between expression of gene transcripts and patients’ clinicopathological and survival data. Our results show that several of these genes are significantly expressed in both normal and tumour tissue; in many cases, expression is altered in the tumour specimens as compared to normal breast tissue. Moreover, expression of ARNT2 and GST A1 was correlated with prognosis. Kaplan–Meier analysis showed expression of ARNT2 mRNA to correlate significantly with favourable disease outcome for patients, in terms of both their disease-free survival (P = 0.0094) and overall survival (P = 0.0018) times from diagnosis, while detection of GST A1 mRNA correlated with shortened disease-free survival (P = 0.0131) and overall survival (P = 0.0028). Multivariate analysis indicated GST A1 expression to be an independent prognostic factor for overall survival (P = 0.045). Our results suggest a possible use of ARNT2 and GST A1 as prognostic breast cancer biomarkers.

Similar content being viewed by others
References
Perquin M, Oster T, Maul A, Froment N, Untereiner M, Bagrel D (2000) The glutathione-related detoxification pathway in the human breast: a highly coordinated system disrupted in the tumour tissues. Cancer Lett 1:7–16
Balendiran GK, Dabur R, Fraser D (2004) The role of glutathione in cancer. Cell Biochem Funct 6:343–352
Oyama T, Kagawa N, Kunugita N, Kitagawa K, Ogawa M, Yamaguchi T, Suzuki R, Kinaga T, Yashima Y, Ozaki S et al (2004) Expression of cytochrome P450 in tumor tissues and its association with cancer development. Front Biosci 1967–1976
Nebert DW, Dalton TP (2006) The role of cytochrome P450 enzymes in endogenous signalling pathways and environmental carcinogenesis. Nat Rev Cancer 12:947–960
Rodriguez-Antona C, Ingelman-Sundberg M (2006) Cytochrome P450 pharmacogenetics and cancer. Oncogene 11:1679–1691
Nelson DR, Koymans L, Kamataki T, Stegeman JJ, Feyereisen R, Waxman DJ, Waterman MR, Gotoh O, Coon MJ, Estabrook RW et al (1996) P450 superfamily: update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics 1:1–42
Yu LJ, Matias J, Scudiero DA, Hite KM, Monks A, Sausville EA, Waxman DJ (2001) P450 enzyme expression patterns in the NCI human tumor cell line panel. Drug Metab Dispos 3:304–312
Dhaini HR, Thomas DG, Giordano TJ, Johnson TD, Biermann JS, Leu K, Hollenberg PF, Baker LH (2003) Cytochrome P450 CYP3A4/5 expression as a biomarker of outcome in osteosarcoma. J Clin Oncol 13:2481–2485
Nebert DW, Vasiliou V (2004) Analysis of the glutathione S-transferase (GST) gene family. Hum Genomics 6:460–464
Jiang JG, Chen CL, Card JW, Yang S, Chen JX, Fu XN, Ning YG, Xiao X, Zeldin DC, Wang DW (2005) Cytochrome P450 2J2 promotes the neoplastic phenotype of carcinoma cells and is up-regulated in human tumors. Cancer Res 11:4707–4715
Adler V, Yin Z, Fuchs SY, Benezra M, Rosario L, Tew KD, Pincus MR, Sardana M, Henderson CJ, Wolf CR et al (1999) Regulation of JNK signaling by GSTp. EMBO J 5:1321–1334
Tew KD, Ronai Z (1999) GST function in drug and stress response. Drug Resist Updat 3:143–147
Murray GI, Taylor MC, McFadyen MC, McKay JA, Greenlee WF, Burke MD, Melvin WT (1997) Tumor-specific expression of cytochrome P450 CYP1B1. Cancer Res 14:3026–3031
Iscan M, Klaavuniemi T, Coban T, Kapucuoglu N, Pelkonen O, Raunio H (2001) The expression of cytochrome P450 enzymes in human breast tumours and normal breast tissue. Breast Cancer Res Treat 1:47–54
Muskhelishvili L, Thompson PA, Kusewitt DF, Wang C, Kadlubar FF (2001) In situ hybridization and immunohistochemical analysis of cytochrome P450 1B1 expression in human normal tissues. J Histochem Cytochem 229–236
Kapucuoglu N, Coban T, Raunio H, Pelkonen O, Edwards RJ, Boobis AR, Iscan M (2003) Expression of CYP3A4 in human breast tumour and non-tumour tissues. Cancer Lett 1:17–23
Page DL, Andersen AT (1987) Diagnostic histopathology. Churchill Livingstone, London, Melbourne and New York
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathol 403–410
Sobin LH, Wittekind C (1997). TNM classification of malignant tumours. John Wiley and Sons, New York
O’Driscoll L, McMorrow J, Doolan P, McKiernan E, Mehta JP, Ryan E, Gammell P, Joyce H, O’Donovan N, Walsh N et al (2006) Investigation of the molecular profile of basal cell carcinoma using whole genome microarrays. Mol Cancer 74
Karlgren M, Gomez A, Stark K, Svard J, Rodriguez-Antona C, Oliw E, Bernal ML, Ramon y Cajal S, Johansson I, Ingelman-Sundberg M (2006) Tumor-specific expression of the novel cytochrome P450 enzyme, CYP2W1. Biochem Biophys Res Commun 2:451–458
El-Rayes BF, Ali S, Heilbrun LK, Lababidi S, Bouwman D, Visscher D, Philip PA (2003) Cytochrome p450 and glutathione transferase expression in human breast cancer. Clin Cancer Res 5:1705–1709
Liloglou T, Walters M, Maloney P, Youngson J, Field JK (2002) A T2517C polymorphism in the GSTM4 gene is associated with risk of developing lung cancer. Lung Cancer 2:143–146
Brigelius-Flohe R, Muller C, Menard J, Florian S, Schmehl K, Wingler K (2001) Functions of GI-GPx: lessons from selenium-dependent expression and intracellular localization. Biofactors 1–4:101–106
Chu FF, Esworthy RS, Lee L, Wilczynski S (1999) Retinoic acid induces Gpx2 gene expression in MCF-7 human breast cancer cells. J Nutr 10:1846–1854
Woenckhaus M, Klein-Hitpass L, Grepmeier U, Merk J, Pfeifer M, Wild P, Bettstetter M, Wuensch P, Blaszyk H, Hartmann A et al (2006) Smoking and cancer-related gene expression in bronchial epithelium and non-small-cell lung cancers. J Pathol 2:192–204
van't Veer LJ, Dai H, van de Vijver MJ, He YD, Hart AA, Mao M, Peterse HL, van der Kooy K, Marton MJ, Witteveen AT et al (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 6871:530–536
Maltepe E, Keith B, Arsham AM, Brorson JR, Simon MC (2000) The role of ARNT2 in tumor angiogenesis and the neural response to hypoxia. Biochem Biophys Res Commun 1:231–238
Freeburg PB, Abrahamson DR (2004) Divergent expression patterns for hypoxia-inducible factor-1beta and aryl hydrocarbon receptor nuclear transporter-2 in developing kidney. J Am Soc Nephrol 10:2569–2578
Hosoya T, Oda Y, Takahashi S, Morita M, Kawauchi S, Ema M, Yamamoto M, Fujii-Kuriyama Y (2001) Defective development of secretory neurones in the hypothalamus of Arnt2-knockout mice. Genes Cells 4:361–374
Hsu HJ, Wang WD, Hu CH (2001) Ectopic expression of negative ARNT2 factor disrupts fish development. Biochem Biophys Res Commun 2:487–492
Schlezinger JJ, Liu D, Farago M, Seldin DC, Belguise K, Sonenshein GE, Sherr DH (2006) A role for the aryl hydrocarbon receptor in mammary gland tumorigenesis. Biol Chem 9:1175–1187
Tanner B, Hengstler JG, Dietrich B, Henrich M, Steinberg P, Weikel W, Meinert R, Kaina B, Oesch F, Knapstein PG (1997) Glutathione, glutathione S-transferase alpha and pi, and aldehyde dehydrogenase content in relationship to drug resistance in ovarian cancer. Gynecol Oncol 1:54–62
Peters WH, Roelofs HM, van Putten WL, Jansen JB, Klijn JG, Foekens JA (1993) Response to adjuvant chemotherapy in primary breast cancer: no correlation with expression of glutathione S-transferases. Br J Cancer 1:86–92
Alpert LC, Schecter RL, Berry DA, Melnychuk D, Peters WP, Caruso JA, Townsend AJ, Batist G (1997) Relation of glutathione S-transferase alpha and mu isoforms to response to therapy in human breast cancer. Clin Cancer Res 5:661–667
Wrigley EC, McGown AT, Buckley H, Hall A, Crowther D (1996) Glutathione-S-transferase activity and isoenzyme levels measured by two methods in ovarian cancer, and their value as markers of disease outcome. Br J Cancer 6:763–769
Sweeney C, Ambrosone CB, Joseph L, Stone A, Hutchins LF, Kadlubar FF, Coles BF (2003) Association between a glutathione S-transferase A1 promoter polymorphism and survival after breast cancer treatment. Int J Cancer 6:810–814
Acknowledgments
This work was supported by funding from Ireland’s Higher Educational Authority Programme for Research in Third Level Institutions (PRTLI) Cycle 3, equipment funding from Science Foundation Ireland, Dublin City University’s Albert College Fellowship, Dublin City University’s Research Fellowship, and the Health Research Board.
Author information
Authors and Affiliations
Corresponding author
Additional information
V. Martinez and S. Kennedy contributed equally.
Rights and permissions
About this article
Cite this article
Martinez, V., Kennedy, S., Doolan, P. et al. Drug metabolism-related genes as potential biomarkers: analysis of expression in normal and tumour breast tissue. Breast Cancer Res Treat 110, 521–530 (2008). https://doi.org/10.1007/s10549-007-9739-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9739-9