Summary
Purpose
To evaluate the hypothesis that telomere DNA content (TC) in breast tumor tissue correlates with TNM staging and prognosis.
Experimental design
Slot blot assay was used to quantitate TC in 70 disease-free normal tissues from multiple organ sites, and two independent sets of breast tumors containing a total of 140 samples. Non-parametric Rank–Sums tests, logistic regression and Cox proportional hazards models were used to evaluate the relationships between TC and tumor size, nodal involvement, TNM stage, 5-year survival and disease-free interval.
Results
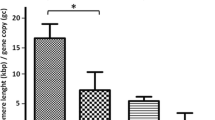
TC in 95% of normal tissues was 75–143% of that in the placental DNA standard, whereas only 50% of tumors had TC values in this range. TC was associated with tumor size (p=0.02), nodal involvement (p<0.0001), TNM stage (p=0.004), 5-year overall survival (p=0.0001) and 5-year disease-free survival (p=0.0004). A multivariable Cox model was developed using age at diagnosis, TNM stage and TC as independent predictors of breast cancer-free survival. Relative to the high TC group (>123% of standard), low TC (<101% of standard) conferred an adjusted relative hazard of 4.43 (95% CI 1.4–13.6, p=0.009). Receiver operating characteristic curves using thresholds defined by the TC distribution in normal tissues predicted 5-year breast cancer-free survival with 50% sensitivity and 95% specificity, and predicted death due to breast cancer with 75% sensitivity and 70% specificity.
Conclusions
TC in breast cancer tissue is an independent predictor of clinical outcome and survival interval, and may discriminate by stage.
Similar content being viewed by others
References
Goldhirsch A, Glick JH, Gelber RD, Senn HJ Meeting highlights: International Consensus Panel on the Treatment of Primary Breast Cancer J Natl Cancer Inst 90: 1601–1608, 1998
McGuire WL Breast cancer prognostic factors: evaluation guidelines J Natl Cancer Inst 83: 154–155, 1991
Polychemotherapy for early breast cancer: an overview of the randomised trials Early Breast Cancer Trialists’ Collaborative Group Lancet 352: 930–942, 1998
Eifel P, Axelson JA, Costa J, Crowley J, Curran WJ Jr, Deshler A, Fulton S, Hendricks CB, Kemeny M, Kornblith AB, Louis TA, Markman M, Mayer R, Roter D National Institutes of Health Consensus Development Conference Statement: adjuvant therapy for breast cancer, November 1–3, 2000 J Natl Cancer Inst 93: 979–989, 2001
Glick JH, Gelber RD, Goldhirsch A, Senn HJ Meeting highlights: adjuvant therapy for primary breast cancer J Natl Cancer Inst 84: 1479–1485, 1992
Hanahan D, Weinberg RA. The hallmarks of cancer Cell, 100: 57–70, 2000
Lo AW, Sabatier L, Fouladi B, Pottier G, Ricoul M, Murnane JP DNA amplification by breakage/fusion/bridge cycles initiated by spontaneous telomere loss in a human cancer cell line Neoplasia 4: 531–538, 2002
O’Hagan R, Chang S, Maser R, Mohan R, Artandi S, Chin L, DePinho R Telomere dysfunction provokes regional amplification and deletion in cancer genomes Cancer Cell 2: 149–155, 2002
Counter CM, Avilion AA, LeFeuvre CE, Stewart NG, Greider CW, Harley CB, Bacchetti S Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity EMBO J 11: 1921–1929, 1992
de Lange T, Shiue L, Myers RM, Cox DR, Naylor SL, Killery AM, Varmus HE Structure and variability of human chromosome ends Mol Cell Biol 10: 518–527, 1990
Saltman D, Morgan R, Cleary ML, de Lange T Telomeric structure in cells with chromosome end associations Chromosoma 102: 121–128, 1993
Hande MP, Samper E, Lansdorp P, Blasco MA Telomere length dynamics and chromosomal instability in cells derived from telomerase null mice J Cell Biol 144: 589–601, 1999
Allsopp RC, Chang E, Kashefiaazam M, Rogaev EI, Piatyszek MA, Shay JW, Harley CB Telomere shortening is associated with cell division in vitro and in vivo Exp Cell Res 220: 194–200, 1995
Karlseder J, Smogorzewska A, de Lange T Senescence induced by altered telomere state, not telomere loss Science 295: 2446–2449, 2002
Smogorzewska A, Van Steensel B, Bianchi A, Oelmann S, Schaefer MR, Schnapp G, de Lange T Control of human telomere length by TRF1 and TRF2 Mol Cell Biol. 20: 1659–1668, 2000
Vulliamy T, Marrone A, Dokal I, Mason PJ Association between aplastic anaemia and mutations in telomerase RNA Lancet 359: 2168–2170, 2002
Gisselsson D, Jonson T, Petersen A, Strombeck B, Dal Cin P, Hoglund M, Mitelman F, et al. Telomere dysfunction triggers extensive DNA fragmentation and evolution of complex chromsome abnormalities in human malignant tumors Proc Natl Acad Sci USA 98:12683–12688, 2001
Vaziri H Critical telomere shortening regulated by the ataxia-telangiectasia gene acts as a DNA damage signal leading to activation of p53 protein and limited life-span of human diploid fibroblasts. A review Biochemistry (Moscow) 62:1306–1310, 1997
Griffith JK, Bryant JE, Fordyce CA, Gilliland FD, Joste NE, Moyzis RK Reduced telomere DNA content is correlated with genomic instability and metastasis in invasive human breast carcinoma Breast Cancer Res Treat 54: 59–64, 1999
Fordyce CA, Heaphy CM, Joste NE, Smith AY, Hunt WC, Griffith JK Association between cancer-free survival and telomere DNA content in prostate tumors J Urol 173: 610–614, 2005
Donaldson L, Fordyce C, Gilliland F, Smith A, Feddersen R, Joste N, Moyzis R, Griffith JK Association between outcome and telomere DNA content in prostate cancer J Urol 162: 1788–1792, 1999
Grabowski P, Hultdin M, Karlsson K, Tobin G, Aleskog A, Thunberg U, Laurell A, Sundstrom C, Rosenquist R, Roos G Telomere length as a prognostic parameter in chronic lymphocytic leukemia with special reference to VH gene mutation status Blood 105: 4807–4812, 2005
Kubuki Y, Suzuki M, Sasaki H, Toyama T, Yamashita K, Maeda K, Ido A, Matsuoka H, Okayama A, Nakanishi T, Tsubouchi H Telomerase activity and telomere length as prognostic factors of adult T-cell leukemia Leuk Lymphoma 46:393–399, 2005
Drummond M, Lennard A, Brummendorf T, Holyoake T Telomere shortening correlates with prognostic score at diagnosis and proceeds rapidly during progression of chronic myeloid leukemia Leuk Lymphoma 45:1775–1781, 2004
Bryant JE, Hutchings KG, Moyzis RK, Griffith JK Measurement of telomeric DNA content in human tissues Biotechniques 23: 476–478, 1997
Fordyce CA, Heaphy CM, Griffith JK Chemiluminescent measurement of telomere DNA content in biopsies Biotechniques 33: 144–148, 2002
Hiyama E, Hiyama K, Yokoyama T, Ichikawa T, Matsuura Y Length of telomeric repeats in neuroblastoma: correlation with prognosis and other biological characteristics Jpn J Cancer Res 83: 159–164, 1992
Hakin-Smith V, Jellinek DA, Levy D, Carroll T, Teo M, Timperley WR, McKay MJ, Reddel RR, Royds JA Alternative lengthening of telomeres and survival in patients with glioblastoma multiforme Lancet 361: 836–838, 2003
Baumgartner KB, Baumgartner R, Ballard-Barbash R, Hunt C, Crumley D, Gilliland F, McTiernen A, Bernstein L Association of body composition and weight history with breast cancer prognostic markers in Hispanic and Non-Hispanic White women Am J Epidemiology 160: 1087–1097, 2004
Singletary SE, Allred C, Ashley P, Bassett LW, Berry D, Bland KI, Borgen PI, Clark G, Edge SB, Hayes DF, Hughes LL, Hutter RV, Morrow M, Page DL, Recht A, Theriault RL, Thor A, Weaver DL, Wieand HS, Greene FL Revision of the American Joint Committee on Cancer staging system for breast cancer J Clin Oncol 20: 3628–3636, 2002
Heaphy CM, Bisoffi M, Fordyce CA, Haaland, C Joste, NE, Griffith, JK: Telomere DNA Content and allelic imbalance in histologically normal tissue adjacent to breast tumors: Implications for prognosis. Int J Cancer DOI: 10.1002/ijc.21815
Acknowledgments
This work was supported by research grants DAMD17-01-1-0572 and W81XWH-05-1-0226 to JKG from the DOD BCRP. CAF and CMH were supported by predoctoral training awards, DAMD 17-00-1-0370 and W81XWH-05-1-0273 from the DOD BCRP. JLW and CMH also were supported by an NIH MBRS Award, R25 GM60201, an NIH MARC Award, T34 GM08751, and DOD BCRP Undergraduate Breast Cancer Summer Research Training Program Award, DAMD17-02-1-0513-01. RNB and KBB and data from the HEAL Study were supported by SEER/NCI N01-CN-65034-29. We are indebted to Dr Melanie Royce for critically␣reviewing the manuscript and her several helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Colleen A. Fordyce and Christopher M. Heaphy contributed equally to this study
Rights and permissions
About this article
Cite this article
Fordyce, C.A., Heaphy, C.M., Bisoffi, M. et al. Telomere content correlates with stage and prognosis in breast cancer. Breast Cancer Res Treat 99, 193–202 (2006). https://doi.org/10.1007/s10549-006-9204-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9204-1