Abstract
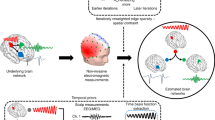
Magnetoencephalography (MEG) is clinically used to localize interictal spikes in discrete brain areas of epilepsy patients through the equivalent current dipole (ECD) method, but does not account for the temporal dynamics of spike activity. Recent studies found that interictal spike propagation beyond the temporal lobe may be associated with worse postsurgical outcomes, but studies using whole-brain data such as in MEG remain limited. In this pilot study, we developed a tool that visualizes the spatiotemporal dynamics of interictal MEG spikes normalized to spike-free sleep activity to assess their onset and propagation patterns in patients with temporal lobe epilepsy (TLE). We extracted interictal source data containing focal epileptiform activity in awake and asleep states from seven patients whose MEG ECD clusters localized to the temporal lobe and normalized the data against spike-free sleep recordings. We calculated the normalized activity over time per cortical label, confirmed maximal activity at onset, and mapped the activity over a 10 ms interval onto each patient’s brain using a custom-built Multi-Modal Visualization Tool. The onset of activity in all patients appeared near the clinically determined epileptogenic zone. By 10 ms, four of the patients had propagated source activity restricted to within the temporal lobe, and three had propagated source activity spread to extratemporal regions. Using this tool, we show that noninvasively identifying the onset and propagation of interictal spike activity in MEG can be achieved, which may help provide further insight into epileptic networks and guide surgical planning and interventions in patients with TLE.



Similar content being viewed by others
Data Availability
De-identified data not published within this manuscript will be shared with any qualified investigator upon reasonable written request.
References
Alarcon G, Garcia Seoane JJ, Binnie CD et al (1997) Origin and propagation of interictal discharges in the acute electrocorticogram. Implications for pathophysiology and surgical treatment of temporal lobe epilepsy. Brain 120(Pt 12):2259–2282. https://doi.org/10.1093/brain/120.12.2259
Asadi-Pooya AA, Stewart GR, Abrams DJ, Sharan A (2017) Prevalence and incidence of drug-resistant mesial temporal lobe epilepsy in the United States. World Neurosurg 99:662–666
Azeem A, von Ellenrieder N, Hall J et al (2021) Interictal spike networks predict surgical outcome in patients with drug-resistant focal epilepsy. Ann Clin Transl Neurol 8:1212–1223. https://doi.org/10.1002/acn3.51337
Barba C, Rheims S, Minotti L et al (2016) Temporal plus epilepsy is a major determinant of temporal lobe surgery failures. Brain 139:444–451. https://doi.org/10.1093/brain/awv372
Chang EF, Englot DJ, Vadera S (2015) Minimally invasive surgical approaches for temporal lobe epilepsy. Epilepsy Behav 47:24–33
Conrad EC, Tomlinson SB, Wong JN et al (2020) Spatial distribution of interictal spikes fluctuates over time and localizes seizure onset. Brain 143:554–569. https://doi.org/10.1093/brain/awz386
Dale AM, Liu AK, Fischl BR et al (2000) Dynamic statistical parametric mapping: combining fMRI and MEG for high-resolution imaging of cortical activity. Neuron 26:55–67. https://doi.org/10.1016/s0896-6273(00)81138-1
Derry CP, Duncan S (2013) Sleep and epilepsy. Epilepsy Behav 26:394–404
Ebersole JS (1997) Magnetoencephalography/magnetic source imaging in the assessment of patients with epilepsy. Epilepsia 38(Suppl 4):S1-5. https://doi.org/10.1111/j.1528-1157.1997.tb04533.x
Felsenstein O, Peled N, Hahn E et al (2019) Multi-modal neuroimaging analysis and visualization tool (MMVT). arXiv preprint arXiv:191210079
Fischer MJM, Scheler G, Stefan H (2005) Utilization of magnetoencephalography results to obtain favourable outcomes in epilepsy surgery. Brain 128:153–157. https://doi.org/10.1093/brain/awh333
Gumenyuk V, Roth T, Moran JE et al (2009) Cortical locations of maximal spindle activity: magnetoencephalography (MEG) study. J Sleep Res 18:245–253. https://doi.org/10.1111/j.1365-2869.2008.00717.x
Hara K, Lin FH, Camposano S et al (2007) Magnetoencephalographic mapping of interictal spike propagation: a technical and clinical report. AJNR Am J Neuroradiol 28:1486–1488
Harroud A, Bouthillier A, Weil AG, Nguyen DK (2012) Temporal lobe epilepsy surgery failures: a review. Epilepsy Res Treat 2012:201651. https://doi.org/10.1155/2012/201651
Jehi L (2018) The epileptogenic zone: concept and definition. Epilepsy Curr 18:12–16. https://doi.org/10.5698/1535-7597.18.1.12
Klein A, Tourville J (2012) 101 labeled brain images and a consistent human cortical labeling protocol. Front Neurosci 6:171. https://doi.org/10.3389/fnins.2012.00171
Knowlton RC, Shih J (2004) Magnetoencephalography in epilepsy. Epilepsia 45(Suppl 4):61–71
Kuznetsova A, Ossadtchi A (2021) Local propagation of MEG interictal spikes: source reconstruction with traveling wave priors. In: 2021 Third International Conference Neurotechnologies and Neurointerfaces (CNN). pp 53–56
Lambert I, Roehri N, Giusiano B et al (2018) Brain regions and epileptogenicity influence epileptic interictal spike production and propagation during NREM sleep in comparison with wakefulness. Epilepsia 59:235–243. https://doi.org/10.1111/epi.13958
Laohathai C, Ebersole JS, Mosher JC et al (2021) Practical fundamentals of clinical MEG interpretation in epilepsy. Front Neurol 12:722986. https://doi.org/10.3389/fneur.2021.722986
Matarrese MAG, Loppini A, Fabbri L et al (2023) Spike propagation mapping reveals effective connectivity and predicts surgical outcome in epilepsy. Brain. https://doi.org/10.1093/brain/awad118
Owen TW, Schroeder GM, Janiukstyte V et al (2023) MEG abnormalities and mechanisms of surgical failure in neocortical epilepsy. Epilepsia 64:692–704. https://doi.org/10.1111/epi.17503
Piastra MC, Nüßing A, Vorwerk J et al (2021) A comprehensive study on electroencephalography and magnetoencephalography sensitivity to cortical and subcortical sources. Hum Brain Mapp 42:978–992. https://doi.org/10.1002/hbm.25272
Pizzo F, Roehri N, Medina Villalon S et al (2019) Deep brain activities can be detected with magnetoencephalography. Nat Commun. https://doi.org/10.1038/s41467-019-08665-5
Ramantani G, Boor R, Paetau R et al (2006) MEG versus EEG: influence of background activity on interictal spike detection. J Clin Neurophysiol 23:498–508
Tanaka M, Du Q (1990) Front matter. In: Tanaka M, Du Q (eds) Boundary element methods. Pergamon, Oxford, p iii
Tanaka N, Hämäläinen MS, Ahlfors SP et al (2010) Propagation of epileptic spikes reconstructed from spatiotemporal magnetoencephalographic and electroencephalographic source analysis. Neuroimage 50:217–222. https://doi.org/10.1016/j.neuroimage.2009.12.033
Tanaka N, Peters JM, Prohl AK et al (2014) Clinical value of magnetoencephalographic spike propagation represented by spatiotemporal source analysis: correlation with surgical outcome. Epilepsy Res 108:280–288
Taulu S, Simola J (2006) Spatiotemporal signal space separation method for rejecting nearby interference in MEG measurements. PhysMedBiol 51:1759–1768
Tomlinson SB, Bermudez C, Conley C et al (2016) Spatiotemporal mapping of interictal spike propagation: a novel methodology applied to pediatric intracranial EEG recordings. Front Neurol 7:229. https://doi.org/10.3389/fneur.2016.00229
Wang ZI, Jin K, Kakisaka Y et al (2012) Imag(in)ing seizure propagation: MEG-guided interpretation of epileptic activity from a deep source. Hum Brain Mapp 33:2797–2801. https://doi.org/10.1002/hbm.21401
Zumsteg D, Friedman A, Wieser HG, Wennberg RA (2006) Propagation of interictal discharges in temporal lobe epilepsy: correlation of spatiotemporal mapping with intracranial foramen ovale electrode recordings. Clin Neurophysiol 117:2615–2626. https://doi.org/10.1016/j.clinph.2006.07.319
Acknowledgements
The authors thank Callisto Cordray for her technical assistance in the acquisition of recordings as well as for organizing and navigating the MMVT program files.
Funding
OT received salary and research support from the NIH P20GM130447 Cognitive Neuroscience and Development of Aging (CONDA) Award. VG received research funding from the NIH P20GM130447 Cognitive Neuroscience and Development of Aging (CoNDA) Award.
Author information
Authors and Affiliations
Contributions
DZ, VG, and NP wrote the main manuscript text, prepared the figures, and reviewed and revised. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
N.P. and S.S. are co-founders and shareholders of FIND Surgical Sciences Inc, a startup implementing a clinical decision support platform for epilepsy. All other authors report no competing interests.
Ethical Approval
The Institutional Review Board of the University of Nebraska Medical Center provided ethical approval, including waiver of informed consent, for the retrospective collection of electronic health record, imaging, and neurophysiologic data for all of the patients in this study (IRB #0714-21-EP).
Additional information
Handling Editor: Bin He
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, D.J., Gumenyuk, V., Taraschenko, O. et al. Visualization of the Spatiotemporal Propagation of Interictal Spikes in Temporal Lobe Epilepsy: A MEG Pilot Study. Brain Topogr 37, 116–125 (2024). https://doi.org/10.1007/s10548-023-01017-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-023-01017-z