Abstract
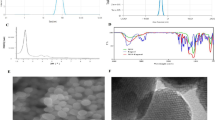
Gemcitabine (GEM) is the first-line treatment for pancreatic adenocarcinoma (PAC) yet chemoresistance is common. Nitric oxide (NO) is the predominant species responsible for the cytotoxic action of macrophages against cancer cells yet localized delivery is difficult given the short half-life. We sought to study the effect of locally delivered NO on GEM mediated PAC cytotoxicity and the potential role of SMAD4 in this effect. We hypothesized that NO would enhance the cytotoxicity of GEM in a SMAD4 dependent manner. NO-Silica nanoparticles (NO-Si) were synthesized via a co-condensation of tetraethoxysilane with aminoalkoxysilane under high-pressure nitrous oxide. NO release was measured using chemiluminescence. A SMAD4 negative PAC cell line (SMAD4-) was made using retroviral knockdown of Panc1 PAC cells. Panc1 and SMAD4- cells were treated with gemcitabine (100 nm (hi) to 30 μm (lo)), 30 mg NOSi particles, or both (NOSihi or NOSilo) and cell viability assessed. NoSi reduced cell viability by 25.99% in Panc1 and 24.38% in SMAD4-. When combined with gemcitabine, further reductions were seen in a dose dependent manner for both cell lines. We have demonstrated the in-vitro dose dependent cytotoxic effects of NOSi. When combined with GEM there is a synergistic effect resulting in improved cytotoxicity seen in both Panc1 and SMAD4- PAC cells with a differential pattern of cell death seen at high concentrations of NO. These findings suggest not only that NO is useful chemosensitizing agent but that SMAD4- may play a role in its synergism with GEM.





Similar content being viewed by others
References
R. Altieri, M. Fontanella, A. Agnoletti, P.P. Panciani, G. Spena, E. Crobeddu, et al., Role of nitric oxide in glioblastoma therapy: Another step to resolve the terrible puzzle? Transl Med UniSa 12, 54–59 (2015)
D. Ansari, A. Gustafsson, R. Andersson, Update on the management of pancreatic cancer: Surgery is not enough. World J. Gastroenterol. 21(11), 3157–3165 (2015). https://doi.org/10.3748/wjg.v21.i11.3157
N. Bardeesy, K.H. Cheng, J.H. Berger, G.C. Chu, J. Pahler, P. Olson, et al., Smad4 is dispensable for normal pancreas development yet critical in progression and tumor biology of pancreas cancer. Genes Dev. 20(22), 3130–3146 (2006). https://doi.org/10.1101/gad.1478706
K. Bian, F. Murad, What is next in nitric oxide research? From cardiovascular system to cancer biology. Nitric Oxide 43, 3–7 (2014). https://doi.org/10.1016/j.niox.2014.08.006
Y. Binenbaum, S. Na'ara, Z. Gil, Gemcitabine resistance in pancreatic ductal adenocarcinoma. Drug Resist. Updat. 23, 55–68 (2015). https://doi.org/10.1016/j.drup.2015.10.002
A. Blackford, O.K. Serrano, C.L. Wolfgang, G. Parmigiani, S. Jones, X. Zhang, et al., SMAD4 gene mutations are associated with poor prognosis in pancreatic cancer. Clin. Cancer Res. 15(14), 4674–4679 (2009). https://doi.org/10.1158/1078-0432.CCR-09-0227
M. Brunori, A. Giuffre, P. Sarti, G. Stubauer, M.T. Wilson, Nitric oxide and cellular respiration. Cell. Mol. Life Sci. 56(7–8), 549–557 (1999)
N.S. Bryan, M.B. Grisham, Methods to detect nitric oxide and its metabolites in biological samples. Free Radic. Biol. Med. 43(5), 645–657 (2007). https://doi.org/10.1016/j.freeradbiomed.2007.04.026
A.W. Carpenter, M.H. Schoenfisch, Nitric oxide release: Part II. Therapeutic applications. Chem. Soc. Rev. 41(10), 3742–3752 (2012). https://doi.org/10.1039/c2cs15273h
Y.W. Chen, P.J. Hsiao, C.C. Weng, K.K. Kuo, T.L. Kuo, D.C. Wu, et al., SMAD4 loss triggers the phenotypic changes of pancreatic ductal adenocarcinoma cells. BMC Cancer 14, 181 (2014). https://doi.org/10.1186/1471-2407-14-181
H. Cheng, L. Wang, M. Mollica, A.T. Re, S. Wu, L. Zuo, Nitric oxide in cancer metastasis. Cancer Lett. 353(1), 1–7 (2014). https://doi.org/10.1016/j.canlet.2014.07.014
A. Cid-Arregui, V. Juarez, Perspectives in the treatment of pancreatic adenocarcinoma. World J. Gastroenterol. 21(31), 9297–9316 (2015). https://doi.org/10.3748/wjg.v21.i31.9297
L. de Sousa Cavalcante, G. Monteiro, Gemcitabine: Metabolism and molecular mechanisms of action, sensitivity and chemoresistance in pancreatic cancer. Eur. J. Pharmacol. 741, 8–16 (2014). https://doi.org/10.1016/j.ejphar.2014.07.041
R. Farias-Eisner, F. Teng, M. Oliveira, R. Leuchter, B. Karlan, L.D. Lagasse, J.S. Berek, The influence of tumor grade, distribution, and extent of carcinomatosis in minimal residual stage III epithelial ovarian cancer after optimal primary cytoreductive surgery. Gynecol. Oncol. 55(1), 108–110 (1994). https://doi.org/10.1006/gyno.1994.1257
E. Fokas, E. O'Neill, A. Gordon-Weeks, S. Mukherjee, W.G. McKenna, R.J. Muschel, Pancreatic ductal adenocarcinoma: From genetics to biology to radiobiology to oncoimmunology and all the way back to the clinic. Biochim. Biophys. Acta 1855(1), 61–82 (2015). https://doi.org/10.1016/j.bbcan.2014.12.001
L.K. Folkes, P. O'Neill, DNA damage induced by nitric oxide during ionizing radiation is enhanced at replication. Nitric Oxide 34, 47–55 (2013). https://doi.org/10.1016/j.niox.2013.04.005
L.J. Frederiksen, R. Sullivan, L.R. Maxwell, S.K. Macdonald-Goodfellow, M.A. Adams, B.M. Bennett, et al., Chemosensitization of cancer in vitro and in vivo by nitric oxide signaling. Clin. Cancer Res. 13(7), 2199–2206 (2007). https://doi.org/10.1158/1078-0432.CCR-06-1807
D. Fukumura, S. Kashiwagi, R.K. Jain, The role of nitric oxide in tumour progression. Nat. Rev. Cancer 6(7), 521–534 (2006). https://doi.org/10.1038/nrc1910
A.F. Hezel, A.C. Kimmelman, B.Z. Stanger, N. Bardeesy, R.A. Depinho, Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 20(10), 1218–1249 (2006). https://doi.org/10.1101/gad.1415606
S.W. Hung, H.R. Mody, R. Govindarajan, Overcoming nucleoside analog chemoresistance of pancreatic cancer: A therapeutic challenge. Cancer Lett. 320(2), 138–149 (2012). https://doi.org/10.1016/j.canlet.2012.03.007
A. Jemal, R. Siegel, E. Ward, T. Murray, J. Xu, M.J. Thun, Cancer statistics, 2007. CA Cancer J. Clin. 57(1), 43–66 (2007)
Y. Kang, J. Ling, R. Suzuki, D. Roife, X. Chopin-Laly, M.J. Truty, et al., SMAD4 regulates cell motility through transcription of N-cadherin in human pancreatic ductal epithelium. PLoS One 9(9), e107948 (2014). https://doi.org/10.1371/journal.pone.0107948
L.K. Keefer, Fifty years of diazeniumdiolate research. From laboratory curiosity to broad-spectrum biomedical advances. ACS Chem. Biol. 6(11), 1147–1155 (2011). https://doi.org/10.1021/cb200274r
E. Kogias, N. Osterberg, B. Baumer, N. Psarras, C. Koentges, A. Papazoglou, et al., Growth-inhibitory and chemosensitizing effects of the glutathione-S-transferase-pi-activated nitric oxide donor PABA/NO in malignant gliomas. Int. J. Cancer 130(5), 1184–1194 (2012). https://doi.org/10.1002/ijc.26106
R. Leone, P. Giussani, S. De Palma, C. Fania, D. Capitanio, M. Vasso, et al., Proteomic analysis of human glioblastoma cell lines differently resistant to a nitric oxide releasing agent. Mol. BioSyst. 11(6), 1612–1621 (2015). https://doi.org/10.1039/c4mb00725e
K. Luberice, D. Downs, B. Sadowitz, S. Ross, A. Rosemurgy, Has survival improved following resection for pancreatic adenocarcinoma? Am. J. Surg. 214(2), 341–346 (2017). https://doi.org/10.1016/j.amjsurg.2017.05.007
J.F. Quinn, M.R. Whittaker, T.P. Davis, Delivering nitric oxide with nanoparticles. J. Control. Release 205, 190–205 (2015). https://doi.org/10.1016/j.jconrel.2015.02.007
D.P. Ryan, T.S. Hong, N. Bardeesy, Pancreatic adenocarcinoma. N. Engl. J. Med. 371(11), 1039–1049 (2014). https://doi.org/10.1056/NEJMra1404198
J.H. Shin, S.K. Metzger, M.H. Schoenfisch, Synthesis of nitric oxide-releasing silica nanoparticles. J. Am. Chem. Soc. 129(15), 4612–4619 (2007). https://doi.org/10.1021/ja0674338
D.J. Stuehr, M.A. Marletta, Mammalian nitrate biosynthesis: Mouse macrophages produce nitrite and nitrate in response to Escherichia coli lipopolysaccharide. Proc. Natl. Acad. Sci. U. S. A. 82(22), 7738–7742 (1985)
F. Wang, X. Xia, C. Yang, J. Shen, J. Mai, H.C. Kim, et al., SMAD4 gene mutation renders pancreatic cancer resistance to radiotherapy through promotion of autophagy. Clin. Cancer Res. 24, 3176 (2018). https://doi.org/10.1158/1078-0432.CCR-17-3435
J.M. Winter, M.F. Brennan, L.H. Tang, M.I. D'Angelica, R.P. Dematteo, Y. Fong, et al., Survival after resection of pancreatic adenocarcinoma: Results from a single institution over three decades. Ann. Surg. Oncol. 19(1), 169–175 (2012). https://doi.org/10.1245/s10434-011-1900-3
H. Ying, P. Dey, W. Yao, A.C. Kimmelman, G.F. Draetta, A. Maitra, R.A. DePinho, Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 30(4), 355–385 (2016). https://doi.org/10.1101/gad.275776.115
M. Zijlstra, N. Bernards, I.H. de Hingh, A.J. van de Wouw, S.H. Goey, E.M. Jacobs, et al., Does long-term survival exist in pancreatic adenocarcinoma? Acta Oncol. 55(3), 259–264 (2016). https://doi.org/10.3109/0284186X.2015.1096020
Financial Support
The authors acknowledge financial support from the following source: Physical Sciences in Oncology - National Cancer Institute of Health grant (U54CA143837).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
There are no financial disclosures or conflicts of interest to report for any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Araujo-Gutierrez, R., Van Eps, J.L., Kirui, D. et al. Enhancement of gemcitabine cytotoxicity in pancreatic adenocarcinoma through controlled release of nitric oxide. Biomed Microdevices 21, 23 (2019). https://doi.org/10.1007/s10544-019-0375-z
Published:
DOI: https://doi.org/10.1007/s10544-019-0375-z