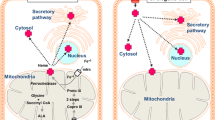
Abstract
The nematode Caenorhabditis elegans (C. elegans) is a convenient tool to evaluate iron metabolism as it shares great orthology with human proteins involved in iron transport, in addition to being transparent and readily available. In this work, we describe how wild-type (N2) C. elegans nematodes in the first larval stage can be loaded with acetomethoxycalcein (CAL-AM) and study it as a whole-organism model for both iron speciation and chelator permeability of the labile iron pool (LIP). This model may be relevant for high throughput assessment of molecules intended for chelation therapy of iron overload diseases.





Similar content being viewed by others
References
Adams PC, Lin E, Barber KR, Grant CWM (1991) Enhanced biliary iron excretion with amphiphilic diethylenetriaminepentaacetic acid. Hepatology 14(6):1230–1234
Adlimoghaddam A, Boeckstaens M, Marini AM, Treberg JR, Brassinga AKC, Weihrauch D (2015) Ammonia excretion in Caenorhabditis elegans: mechanism and evidence of ammonia transport of the Rhesus protein CeRhr-1. J Exp Biol 218(5):675–683. https://doi.org/10.1242/jeb.111856
Alta ECP, Goswami D, Machini MT, Silvestre DM, Nomura CS, Esposito BP (2014) Desferrioxamine-caffeine (DFCAF) as a cell permeant moderator of the oxidative stress caused by iron overload. Biometals 27(6):1351–1360. https://doi.org/10.1007/s10534-014-9795-7
Anderson CP, Leibold EA (2014) Mechanisms of iron metabolism in Caenorhabditis elegans. Front Pharmacol. https://doi.org/10.3389/fphar.2014.00113
Aschner M, Palinski C, Sperling M, Karst U, Schwerdtle T, Bornhorst J (2017) Imaging metals in Caenorhabditis elegans. Metallomics 9(4):357–364. https://doi.org/10.1039/c6mt00265j
Beller G, Lente G, Fabian I (2010) Central role of phenanthroline mono-N-oxide in the decomposition reactions of tris(1,10-phenanthroline)iron(II) and -iron(III) complexes. Inorg Chem 49(9):3968–3970. https://doi.org/10.1021/IC902554b
Brenner S (1974) Genetics of Caenorhabditis-elegans. Genetics 77(1):71–94
Breuer W, Epsztejn S, Cabantchik ZI (1995) Iron acquired from transferrin by K562 cells is delivered into a cytoplasmic pool of chelatable iron(II). J Biol Chem 270(41):24209–24215. https://doi.org/10.1074/jbc.270.41.24209
Bulbake U, Singh A, Domb AJ, Khan W (2019) Therapeutic macromolecular iron chelators. Curr Med Chem 26(2):323–334. https://doi.org/10.2174/0929867325666180904104318
Cabantchik ZI (2014) Labile iron in cells and body fluids: physiology, pathology, and pharmacology. Front Pharmacol. https://doi.org/10.3389/fphar.2014.00045
Carvalho RRV (2019) Bifunctional chelators for the treatment of neurodysfunctions associated with iron. MSc Thesis, University of São Paulo, São Paulo
Chege PM, McColl G (2014) Caenorhabditis elegans: a model to investigate oxidative stress and metal dyshomeostasis in Parkinson's disease. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2014.00089
Cilibrizzi A, Abbate V, Chen YL, Ma YM, Zhou T, Hider RC (2018) Hydroxypyridinone journey into metal chelation. Chem Rev 118(16):7657–7701. https://doi.org/10.1021/acs.chemrev.8b00254
da Silva GS, Shang Z, Kalansuriya P, Capon RJ, Esposito BP (2019) Antioxidant activity and cellular uptake of the hydroxamate-based fungal iron chelators pyridoxatin, desferriastechrome and desferricoprogen. Biometals 32(4):707–715. https://doi.org/10.1007/s10534-019-00202-7
de Lima TG, Benevides FLN, Filho FLE, Farias IS, Dourado DXC, Fontenele EGP, de Moraes MEA, Quidute ARP (2019) Treatment of iron overload syndrome: a general review. Rev Assoc Med Bras 65(9):1216–1222. https://doi.org/10.1590/1806-9282.65.9.1216
Esposito BP, Breuer W, Slotki I, Cabantchik ZI (2002a) Labile iron in parenteral iron formulations and its potential for generating plasma nontransferrin-bound iron in dialysis patients. Eur J Clin Invest 32:42–49. https://doi.org/10.1046/j.1365-2362.2002.0320s1042.x
Esposito BP, Epsztejn S, Breuer W, Cabantchik ZI (2002b) A review of fluorescence methods for assessing labile iron in cells and biological fluids. Anal Biochem 304(1):1–18. https://doi.org/10.1006/abio.2002.5611
Esposito BP, Breuer W, Sirankapracha P, Pootrakul P, Hershko C, Cabantchik ZI (2003) Labile plasma iron in iron overload: redox activity and susceptibility to chelation. Blood 102(7):2670–2677. https://doi.org/10.1182/blood-2003-03-0807
Fagundez DD, Camara DF, Salgueiro WG, Noremberg S, Puntel RL, Piccoli JE, Garcia SC, da Rocha JBT, Aschner M, Avila DS (2015) Behavioral and dopaminergic damage induced by acute iron toxicity in Caenorhabditis elegans. Toxicol Res 4(4):878–884. https://doi.org/10.1039/c4tx00120f
Fajardo C, Costa G, Nande M, Martin M (2016) Three functional biomarkers for monitoring the nanoscale zero-valent iron (nZVI)-induced molecular signature on soil organisms. Water Air Soil Pollut. https://doi.org/10.1007/s11270-016-2901-4
Goel A, Umar S, Nag P, Sharma A, Kumar L, Shamsuzzama Hossain Z, Gayen JR, Nazir A (2015) A dual colorimetric-ratiometric fluorescent probe NAP-3 for selective detection and imaging of endogenous labile iron(III) pools in C. elegans. Chem Commun 51(24):5001–5004. https://doi.org/10.1039/c4cc09798j
Gonzalez-Moragas L, Yu SM, Carenza E, Laromaine A, Roig A (2015) Protective effects of Bovine Serum Albumin on superparamagnetic iron oxide nanoparticles evaluated in the nematode Caenorhabditis elegans. Acs Biomater Sci Eng 1(11):1129–1138. https://doi.org/10.1021/acsbiomaterials.5b00253
Gonzalez-Moragas L, Yu SM, Benseny-Cases N, Sturzenbaum S, Roig A, Laromaine A (2017) Toxicogenomics of iron oxide nanoparticles in the nematode C-elegans. Nanotoxicology 11(5):647–657. https://doi.org/10.1080/17435390.2017.1342011
Guo LE, Liu XY, Wang H, Chen QL, Wang GK, Luo KJ, Mi QL, Zhou Y, Zhang JF (2015) Iron(III)-selective chelation-enhanced fluorescence sensing for in vivo imaging applications. Chem Asian J 10(9):1898–1902. https://doi.org/10.1002/asia.201500415
Hare DJ, Roberts BR, McColl G (2016) Profiling changes to natively-bound metals during Caenorhabditis elegans development. Rsc Adv 6(114):113689–113693. https://doi.org/10.1039/c6ra22084c
Hoss S, Fritzsche A, Meyer C, Bosch J, Meckenstock RU, Totsche KU (2015) Size- and composition-dependent toxicity of synthetic and soil-derived fe oxide colloids for the nematode Caenorhabditis elegans. Environ Sci Technol 49(1):544–552. https://doi.org/10.1021/es503559n
Hu YO, Wang Y, Ye BP, Wang DY (2008) Phenotypic and behavioral defects induced by iron exposure can be transferred to progeny in Caenorhabditis elegans. Biomed Environ Sci 21(6):467–473. https://doi.org/10.1016/s0895-3988(09)60004-0
James SA, Roberts BR, Hare DJ, de Jonge MD, Birchall IE, Jenkins NL, Cherny RA, Bush AI, McColl G (2015) Direct in vivo imaging of ferrous iron dyshomeostasis in ageing Caenorhabditis elegans. Chem Sci 6(5):2952–2962. https://doi.org/10.1039/c5sc00233h
Klang IM, Schilling B, Sorensen DJ, Sahu AK, Kapahi P, Andersen JK, Swoboda P, Killilea DW, Gibson BW, Lithgow GJ (2014) Iron promotes protein insolubility and aging in C. elegans. Aging-Us 6(11):975–991. https://doi.org/10.18632/aging.100689
Lai CH, Chou CY, Ch'ang LY, Liu CS, Lin W (2000) Identification of novel human genes evolutionarily conserved in Caenorhabditis elegans by comparative proteomics. Genome Res 10(5):703–713. https://doi.org/10.1101/gr.10.5.703
Ma Y, Zhou T, Kong X, Hider RC (2012) Chelating agents for the treatment of systemic iron overload. Curr Med Chem 19(17):2816–2827. https://doi.org/10.2174/092986712800609724
McDonald R (1966) Deferoxamine and diethylenetriaminepentaacetic acid (DTPA) in thalassemia. J Pediatr 69(4):563–571
Motekaitis RJ, Martell AE (1994) The iron(III) and iron(II) complexes of nitrilotriacetic acid. J Coord Chem 31(1):67–78. https://doi.org/10.1080/00958979408022546
Ponka P, Richardson DR, Edward JT, Chubb FL (1994) Iron chelators of the pyridoxal isonicotinoyl hydrazone class - relationship of the lipophilicity of the apochelator to its ability to mobilize iron from reticulocytes in-vitro. Can J Physiol Pharmacol 72(6):659–666. https://doi.org/10.1139/y94-093
Ruszkiewicz JA, Pinkas A, Miah MR, Weitz RL, Lawes MJA, Akinyemi AJ, Ijomone OM, Aschner M (2018) C-elegans as a model in developmental neurotoxicology. Toxicol Appl Pharmacol 354:126–135. https://doi.org/10.1016/j.taap.2018.03.016
Saari LA, Seitz WR (1984) Immobilized calcein for metal-ion preconcentration. Anal Chem 56(4):810–813. https://doi.org/10.1021/ac00268a051
Samuni AM, Afeworki M, Stein W, Yordanov AT, DeGraff W, Krishna MC, Mitchell JB, Brechbiel MW (2001) Multifunctional antioxidant activity of HBED iron chelator. Free Radic Biol Med 30(2):170–177. https://doi.org/10.1016/s0891-5849(00)00459-7
Sandell EB, Spindler DC (1949) The soluble complex of ferric iron and 8-hydroxyquinoline. J Am Chem Soc 71(11):3806–3808
Shvartsman M, Fibach E, Cabantchik ZI (2010) Transferrin-iron routing to the cytosol and mitochondria as studied by live and real-time fluorescence. Biochem J 429:185–193. https://doi.org/10.1042/bj20100213
Sihn LM, de Melo FM, Toma HE, Serrano SHP, Esposito BP (2019) A new ferrous diflunisal complex and its effects on biopools of labile iron. J Trace Elem Med Biol 51:65–72. https://doi.org/10.1016/j.jtemb.2018.10.001
Soares MV, Puntel RL, Avila DS (2018) Resveratrol attenuates iron-induced toxicity in a chronic post-treatment paradigm in Caenorhabditis elegans. Free Radic Res 52(9):939–951. https://doi.org/10.1080/10715762.2018.1492119
Sulston J, Hodgkin J (1988) Methods. The Nematode Caenorhabditis elegans, vol 17. Cold Spring Harbor Monographs. https://doi.org/10.1101/0.587-606
Taylor AA, Tsuji JS, Garry MR, McArdle ME, Goodfellow WL, Adams WJ, Menzie CA (2020) Critical review of exposure and effects: implications for setting regulatory health criteria for ingested copper. Environ Manag 65(1):131–159. https://doi.org/10.1007/s00267-019-01234-y
Valentini S, Cabreiro F, Ackerman D, Alam MM, Kunze MBA, Kay CWM, Gems D (2012) Manipulation of in vivo iron levels can alter resistance to oxidative stress without affecting ageing in the nematode C. elegans. Mech Ageing Dev 133(5):282–290. https://doi.org/10.1016/j.mad.2012.03.003
Wang HZ, Jiang X, Wu JY, Zhang LQ, Huang JF, Zhang YR, Zou XJ, Liang B (2016) Iron overload coordinately promotes ferritin expression and fat accumulation in Caenorhabditis elegans. Genetics 203 (1):241-+. https://doi.org/10.1534/genetics.116.186742
Wu QL, Li YP, Tang M, Wang DY (2012) Evaluation of environmental safety concentrations of DMSA coated Fe2O3-NPs using different assay systems in nematode Caenorhabditis elegans. PLoS ONE. https://doi.org/10.1371/journal.pone.0043729
Wu TS, Xu HS, Liang X, Tang M (2019) Caenorhabditis elegans as a complete model organism for biosafety assessments of nanoparticles. Chemosphere 221:708–726. https://doi.org/10.1016/j.chemosphere.2019.01.021
Xu JZ, Marzetti E, Seo AY, Kim JS, Prolla TA, Leeuwenburgh C (2010) The emerging role of iron dyshomeostasis in the mitochondrial decay of aging. Mech Ageing Dev 131(7–8):487–493. https://doi.org/10.1016/j.mad.2010.04.007
Yang YF, Chen PJ, Liao VHC (2016) Nanoscale zerovalent iron (nZVI) at environmentally relevant concentrations induced multigenerational reproductive toxicity in Caenorhabditis elegans. Chemosphere 150:615–623. https://doi.org/10.1016/j.chemosphere.2016.01.068
Yang YF, Lin YJ, Liao CM (2017) Toxicity-based toxicokinetic/toxicodynamic assessment of bioaccumulation and nanotoxicity of zerovalent iron nanoparticles in Caenorhabditis elegans. Int J Nanomed 12:4607–4621. https://doi.org/10.2147/ijn.s138790
Acknowledgements
The authors are grateful for the kind assistance of Dr. Tao Ke with the microscopy images. BPE acknowledges funding from the São Paulo Research Foundation (FAPESP 18/19684-0) and a travel grant from the Coordination of Superior Level Staff Improvement (CAPES 88887.371500/2019-00). MA was supported in part by grants from the National Institute of Environmental Health Sciences (NIEHS) R01ES10563 and R01ES07331.
Funding
FAPESP 18/19684-0; CAPES 88887.371500/2019-00; NIEHS R01ES10563 and R01ES07331.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Espósito, B.P., Martins, A.C., de Carvalho, R.R.V. et al. High throughput fluorimetric assessment of iron traffic and chelation in iron-overloaded Caenorhabditis elegans. Biometals 33, 255–267 (2020). https://doi.org/10.1007/s10534-020-00250-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-020-00250-4