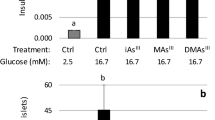
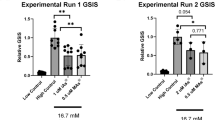
Abstract
Pancreatic beta cells produce and release insulin, a hormone that regulates blood glucose levels, and their dysfunction contributes to the development of diabetes mellitus. Zinc deficiency and inorganic arsenic exposure both independently associate with the development of diabetes, although the effects of their combination on pancreatic beta cell health and function remain unknown. We hypothesized zinc deficiency increases the toxicity associated with arsenic exposure, causing an increased susceptibility to DNA damage and disruption of insulin production. Zinc deficiency decreased cell proliferation by 30% in pancreatic INS-1 rat insulinoma cells. Arsenic exposure (0, 50 or 500 ppb exposures) significantly decreased cell proliferation, and increased mRNA levels of genes involved in stress response (Mt1, Mt2, Hmox1) and DNA damage (p53, Ogg1). When co-exposed to both zinc deficiency and arsenic, zinc deficiency attenuated this response to arsenic, decreasing the expression of Mt1, Hmox1, and Ogg1, and significantly increasing DNA double-strand breaks 2.9-fold. Arsenic exposure decreased insulin expression, but co-exposure did not decrease insulin levels beyond the arsenic alone condition, but did result in a further 33% decline in cell proliferation at the 500 ppb arsenic dose, and a significant increase in beta cell apoptosis. These results suggest zinc deficiency and arsenic, both independently and in combination, adversely affect pancreatic beta cell health and both factors should be considered in the evaluation of health outcomes for susceptible populations.






Similar content being viewed by others
Abbreviations
- qPCR:
-
Quantitative real-time PCR
- T1D:
-
Type I diabetes
- T2D:
-
Type II diabetes
- ZA:
-
Zinc adequate
- ZD:
-
Zinc deficient
References
Abdul KS, Jayasinghe SS, Chandana EP, Jayasumana C, De Silva PM (2015) Arsenic and human health effects: a review. Environ Toxicol Pharmacol 40:828–846. https://doi.org/10.1016/j.etap.2015.09.016
Afridi HI et al (2014) Interaction between essential elements selenium and zinc with cadmium and mercury in samples from hypertensive patients. Biol Trace Elem Res 160:185–196. https://doi.org/10.1007/s12011-014-0048-y
Ayotte JD, Medalie L, Qi SL, Backer LC, Nolan BT (2017) Estimating the high-arsenic domestic-well population in the conterminous United States. Environ Sci Technol 51:12443–12454. https://doi.org/10.1021/acs.est.7b02881
Beaver LM et al (2017) Combinatorial effects of zinc deficiency and arsenic exposure on zebrafish (Danio rerio) development. PLoS ONE 12:e0183831. https://doi.org/10.1371/journal.pone.0183831
Bell DS (2015) Riceabetes: is the association of type 2 diabetes with rice intake due to a high carbohydrate intake or due to exposure to excess inorganic arsenic? Postgrad Med 127:781–782. https://doi.org/10.1080/00325481.2015.1098518
Berglund M, Lindberg AL, Rahman M, Yunus M, Grander M, Lonnerdal B, Vahter M (2011) Gender and age differences in mixed metal exposure and urinary excretion. Environ Res 111:1271–1279. https://doi.org/10.1016/j.envres.2011.09.002
Bhowmick S et al (2015) Assessment of toxic metals in groundwater and saliva in an arsenic affected area of West Bengal, India: a pilot scale study. Environ Res 142:328–336. https://doi.org/10.1016/j.envres.2015.07.005
Bosco MD, Mohanasundaram DM, Drogemuller CJ, Lang CJ, Zalewski PD, Coates PT (2010) Zinc and zinc transporter regulation in pancreatic islets and the potential role of zinc in islet transplantation. Rev Diabet Stud 7:263–274. https://doi.org/10.1900/RDS.2010.7.263
Butte NF, Calloway DH, Van Duzen JL (1981) Nutritional assessment of pregnant and lactating Navajo women. Am J Clin Nutr 34:2216–2228. https://doi.org/10.1093/ajcn/34.10.2216
Chabosseau P, Rutter GA (2016) Zinc and diabetes. Arch Biochem Biophys 611:79–85. https://doi.org/10.1016/j.abb.2016.05.022
Chafe R, Aslanov R, Sarkar A, Gregory P, Comeau A, Newhook LA (2018) Association of type 1 diabetes and concentrations of drinking water components in Newfoundland and Labrador, Canada. BMJ Open Diabetes Res Care 6:e000466. https://doi.org/10.1136/bmjdrc-2017-000466
Chen YW, Yang CY, Huang CF, Hung DZ, Leung YM, Liu SH (2009) Heavy metals, islet function and diabetes development. Islets 1:169–176. https://doi.org/10.4161/isl.1.3.9262
Costarelli L et al (2010) Distinctive modulation of inflammatory and metabolic parameters in relation to zinc nutritional status in adult overweight/obese subjects. J Nutr Biochem 21:432–437. https://doi.org/10.1016/j.jnutbio.2009.02.001
Dashner-Titus EJ et al (2018) Metal exposure and oxidative stress markers in pregnant Navajo Birth Cohort Study participants. Free Radic Biol Med 124:484–492. https://doi.org/10.1016/j.freeradbiomed.2018.04.579
Davidson HW, Wenzlau JM, O’Brien RM (2014) Zinc transporter 8 (ZnT8) and beta cell function. Trends Endocrinol Metab 25:415–424. https://doi.org/10.1016/j.tem.2014.03.008
Dhawan DK, Chadha VD (2010) Zinc: a promising agent in dietary chemoprevention of cancer. Indian J Med Res 132:676–682
Diaz-Villasenor A, Burns AL, Hiriart M, Cebrian ME, Ostrosky-Wegman P (2007) Arsenic-induced alteration in the expression of genes related to type 2 diabetes mellitus. Toxicol Appl Pharmacol 225:123–133. https://doi.org/10.1016/j.taap.2007.08.019
Ding X, Zhou X, Cooper KL, Huestis J, Hudson LG, Liu KJ (2017) Differential sensitivities of cellular XPA and PARP-1 to arsenite inhibition and zinc rescue. Toxicol Appl Pharmacol 331:108–115. https://doi.org/10.1016/j.taap.2017.05.031
Djuzenova CS et al (2015) A prospective study on histone gamma-H2AX and 53BP1 foci expression in rectal carcinoma patients: correlation with radiation therapy-induced outcome. BMC Cancer 15:856. https://doi.org/10.1186/s12885-015-1890-9
Farris W et al (2003) Insulin-degrading enzyme regulates the levels of insulin, amyloid beta-protein, and the beta-amyloid precursor protein intracellular domain in vivo. Proc Natl Acad Sci USA 100:4162–4167. https://doi.org/10.1073/pnas.0230450100
Ferrannini E, Mari A (2014) Beta-Cell function in type 2 diabetes. Metabolism 63:1217–1227. https://doi.org/10.1016/j.metabol.2014.05.012
Fu Y, Tian W, Pratt EB, Dirling LB, Shyng SL, Meshul CK, Cohen DM (2009) Down-regulation of ZnT8 expression in INS-1 rat pancreatic beta cells reduces insulin content and glucose-inducible insulin secretion. PLoS ONE 4:e5679. https://doi.org/10.1371/journal.pone.0005679
Fu Z, Gilbert ER, Liu D (2013) Regulation of insulin synthesis and secretion and pancreatic Beta-cell dysfunction in diabetes. Curr Diabetes Rev 9:25–53
Gaulke CA, Rolshoven J, Wong CP, Hudson LG, Ho E, Sharpton TJ (2018) Marginal zinc deficiency and environmentally relevant concentrations of arsenic elicit combined effects on the gut microbiome. mSphere. https://doi.org/10.1128/mSphere.00521-18
Grau-Perez M et al (2017) The association of arsenic exposure and metabolism with type 1 and type 2 diabetes in youth: the SEARCH case-control study. Diabetes Care 40:46–53. https://doi.org/10.2337/dc16-0810
Guo L, Lichten LA, Ryu MS, Liuzzi JP, Wang F, Cousins RJ (2010) STAT5-glucocorticoid receptor interaction and MTF-1 regulate the expression of ZnT2 (Slc30a2) in pancreatic acinar cells. Proc Natl Acad Sci USA 107:2818–2823. https://doi.org/10.1073/pnas.0914941107
Hall AG, Kelleher SL, Lonnerdal B, Philipps AF (2005) A graded model of dietary zinc deficiency: effects on growth, insulin-like growth factor-I, and the glucose/insulin axis in weanling rats. J Pediatr Gastroenterol Nutr 41:72–80
Ho E (2004) Zinc deficiency, DNA damage and cancer risk. J Nutr Biochem 15:572–578. https://doi.org/10.1016/j.jnutbio.2004.07.005
Ho E, Ames BN (2002) Low intracellular zinc induces oxidative DNA damage, disrupts p53, NFkappa B, and AP1 DNA binding, and affects DNA repair in a rat glioma cell line. Proc Natl Acad Sci USA 99:16770–16775. https://doi.org/10.1073/pnas.222679399
Ho E, Courtemanche C, Ames BN (2003) Zinc deficiency induces oxidative DNA damage and increases p53 expression in human lung fibroblasts. J Nutr 133:2543–2548. https://doi.org/10.1093/jn/133.8.2543
Howard SG (2018) Developmental exposure to endocrine disrupting chemicals and type 1 diabetes mellitus. Front Endocrinol (Lausanne) 9:513. https://doi.org/10.3389/fendo.2018.00513
Huang SH et al (2010) Transgenic expression of haem oxygenase-1 in pancreatic beta cells protects non-obese mice used as a model of diabetes from autoimmune destruction and prolongs graft survival following islet transplantation. Diabetologia 53:2389–2400. https://doi.org/10.1007/s00125-010-1858-x
Huang CF, Chen YW, Yang CY, Tsai KS, Yang RS, Liu SH (2011) Arsenic and diabetes: current perspectives. Kaohsiung J Med Sci 27:402–410. https://doi.org/10.1016/j.kjms.2011.05.008
Jansen J, Karges W, Rink L (2009) Zinc and diabetes–clinical links and molecular mechanisms. J Nutr Biochem 20:399–417. https://doi.org/10.1016/j.jnutbio.2009.01.009
Jurowski K, Szewczyk B, Nowak G, Piekoszewski W (2014) Biological consequences of zinc deficiency in the pathomechanisms of selected diseases. J Biol Inorg Chem 19:1069–1079. https://doi.org/10.1007/s00775-014-1139-0
Kadeyala PK, Sannadi S, Gottipolu RR (2013) Alterations in apoptotic caspases and antioxidant enzymes in arsenic exposed rat brain regions: reversal effect of essential metals and a chelating agent. Environ Toxicol Pharmacol 36:1150–1166. https://doi.org/10.1016/j.etap.2013.09.021
Karagas MR, Gossai A, Pierce B, Ahsan H (2015) Drinking water arsenic contamination, skin lesions, and malignancies: a systematic review of the global evidence. Curr Environ Health Rep 2:52–68. https://doi.org/10.1007/s40572-014-0040-x
Karatela S, Ward NI, Zeng IS, Paterson J (2018) Status and interrelationship of toenail elements in Pacific children. J Trace Elem Med Biol 46:10–16. https://doi.org/10.1016/j.jtemb.2017.11.004
Kawasaki E (2012) ZnT8 and type 1 diabetes. Endocr J 59:531–537
Lemaire K et al (2009) Insulin crystallization depends on zinc transporter ZnT8 expression, but is not required for normal glucose homeostasis in mice. Proc Natl Acad Sci USA 106:14872–14877. https://doi.org/10.1073/pnas.0906587106
Liu S, Guo X, Wu B, Yu H, Zhang X, Li M (2014) Arsenic induces diabetic effects through beta-cell dysfunction and increased gluconeogenesis in mice. Sci Rep 4:6894. https://doi.org/10.1038/srep06894
Liu X, Sun B, Wang X, Nie J, Chen Z, An Y, Tong J (2016) Synergistic effect of radon and sodium arsenite on DNA damage in HBE cells. Environ Toxicol Pharmacol 41:127–131. https://doi.org/10.1016/j.etap.2015.11.013
Lu TH et al (2011) Arsenic induces pancreatic beta-cell apoptosis via the oxidative stress-regulated mitochondria-dependent and endoplasmic reticulum stress-triggered signaling pathways. Toxicol Lett 201:15–26. https://doi.org/10.1016/j.toxlet.2010.11.019
Martin EM, Styblo M, Fry RC (2017) Genetic and epigenetic mechanisms underlying arsenic-associated diabetes mellitus: a perspective of the current evidence. Epigenomics 9:701–710. https://doi.org/10.2217/epi-2016-0097
Naujokas MF, Anderson B, Ahsan H, Aposhian HV, Graziano JH, Thompson C, Suk WA (2013) The broad scope of health effects from chronic arsenic exposure: update on a worldwide public health problem. Environ Health Perspect 121:295–302. https://doi.org/10.1289/ehp.1205875
Nygaard SB, Larsen A, Knuhtsen A, Rungby J, Smidt K (2014) Effects of zinc supplementation and zinc chelation on in vitro beta-cell function in INS-1E cells. BMC Res Notes 7:84. https://doi.org/10.1186/1756-0500-7-84
Padmaja Divya S et al (2015) Arsenic induces insulin resistance in mouse adipocytes and myotubes via oxidative stress-regulated mitochondrial Sirt3-FOXO3a signaling pathway. Toxicol Sci 146:290–300. https://doi.org/10.1093/toxsci/kfv089
Parthasarathy LS, Chiplonkar SA, Khadilkar AV, Khadilkar VV (2015) Dietary modifications to improve micronutrient status of Indian children and adolescents with type 1 diabetes. Asia Pac J Clin Nutr 24:73–82. https://doi.org/10.6133/apjcn.2015.24.1.04
Praveeena S, Pasula S, Sameera K (2013) Trace elements in diabetes mellitus. J Clin Diagn Res 7:1863–1865. https://doi.org/10.7860/JCDR/2013/5464.3335
Rahman S et al (2016) Status of zinc nutrition in Bangladesh: the underlying associations. J Nutr Sci 5:e25. https://doi.org/10.1017/jns.2016.17
Robinson LK, Hurley LS (1981) Effect of maternal zinc deficiency of food restriction on rat fetal pancreas. 2 Insulin and glucagon. J Nutr 111:869–877. https://doi.org/10.1093/jn/111.5.869
Ruttkay-Nedecky B et al (2013) The role of metallothionein in oxidative stress. Int J Mol Sci 14:6044–6066. https://doi.org/10.3390/ijms14036044
Salmonowicz B, Krzystek-Korpacka M, Noczynska A (2014) Trace elements, magnesium, and the efficacy of antioxidant systems in children with type 1 diabetes mellitus and in their siblings. Adv Clin Exp Med 23:259–268
Samuelsson U, Oikarinen S, Hyoty H, Ludvigsson J (2011) Low zinc in drinking water is associated with the risk of type 1 diabetes in children. Pediatr Diabetes 12:156–164. https://doi.org/10.1111/j.1399-5448.2010.00678.x
Sandoval M et al (2007) p53 response to arsenic exposure in epithelial cells: protein kinase B/Akt involvement. Toxicol Sci 99:126–140. https://doi.org/10.1093/toxsci/kfm153
Sharif R, Thomas P, Zalewski P, Fenech M (2012) The role of zinc in genomic stability. Mutat Res 733:111–121. https://doi.org/10.1016/j.mrfmmm.2011.08.009
Skelin M, Rupnik M, Cencic A (2010) Pancreatic beta cell lines and their applications in diabetes mellitus research. Altex 27:105–113
Smedley PLKDG (2002) A review of the source, behaviour, and distribution of arsenic in natural waters. Appl Geochem 17:517–568
Song Y, Chung CS, Bruno RS, Traber MG, Brown KH, King JC, Ho E (2009a) Dietary zinc restriction and repletion affects DNA integrity in healthy men. Am J Clin Nutr 90:321–328. https://doi.org/10.3945/ajcn.2008.27300
Song Y, Leonard SW, Traber MG, Ho E (2009b) Zinc deficiency affects DNA damage, oxidative stress, antioxidant defenses, and DNA repair in rats. J Nutr 139:1626–1631. https://doi.org/10.3945/jn.109.106369
Song Y, Elias V, Loban A, Scrimgeour AG, Ho E (2010) Marginal zinc deficiency increases oxidative DNA damage in the prostate after chronic exercise. Free Radic Biol Med 48:82–88. https://doi.org/10.1016/j.freeradbiomed.2009.10.030
Sun X, Zhou X, Du L, Liu W, Liu Y, Hudson LG, Liu KJ (2014) Arsenite binding-induced zinc loss from PARP-1 is equivalent to zinc deficiency in reducing PARP-1 activity, leading to inhibition of DNA repair. Toxicol Appl Pharmacol 274:313–318. https://doi.org/10.1016/j.taap.2013.11.010
Syring KE et al (2016) Combined deletion of Slc30a7 and Slc30a8 unmasks a critical role for ZnT8 in glucose-stimulated insulin secretion. Endocrinology 157:4534–4541. https://doi.org/10.1210/en.2016-1573
Thirumoorthy N, Manisenthil Kumar KT, Shyam Sundar A, Panayappan L, Chatterjee M (2007) Metallothionein: an overview. World J Gastroenterol 13:993–996
Tseng CH (2004) The potential biological mechanisms of arsenic-induced diabetes mellitus. Toxicol Appl Pharmacol 197:67–83. https://doi.org/10.1016/j.taap.2004.02.009
Valera P et al (2015) Zinc and other metals deficiencies and risk of type 1 diabetes: an ecological study in the high risk Sardinia Island. PLoS ONE 10:e0141262. https://doi.org/10.1371/journal.pone.0141262
Verbanac D, Milin C, Domitrovic R, Giacometti J, Pantovic R, Ciganj Z (1997) Determination of standard zinc values in the intact tissues of mice by ICP spectrometry. Biol Trace Elem Res 57:91–96. https://doi.org/10.1007/BF02803873
Virtanen SM (2016) Dietary factors in the development of type 1 diabetes. Pediatr Diabetes 17(Suppl 22):49–55. https://doi.org/10.1111/pedi.12341
Wani AL, Ahmad A, Shadab GG, Usmani JA (2017) Possible role of zinc in diminishing lead-related occupational stress-a zinc nutrition concern. Environ Sci Pollut Res Int 24:8682–8691. https://doi.org/10.1007/s11356-017-8569-5
Watson GW et al (2016) HDAC6 activity is not required for basal autophagic flux in metastatic prostate cancer cells. Exp Biol Med (Maywood) 241:1177–1185. https://doi.org/10.1177/1535370215618518
Wei H et al (2018) Molecular mechanism of the increased tissue uptake of trivalent inorganic arsenic in mice with type 1 diabetes mellitus. Biochem Biophys Res Commun 504:393–399. https://doi.org/10.1016/j.bbrc.2018.06.029
Welch AHWD, Helsel DR, Wanty RB (2000) Arsenic in ground water of the United States—occurence and geochemistry. Ground Water 38:589–604
Wessells KR, Brown KH (2012) Estimating the global prevalence of zinc deficiency: results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 7:e50568. https://doi.org/10.1371/journal.pone.0050568
Wong CP, Magnusson KR, Ho E (2013) Increased inflammatory response in aged mice is associated with age-related zinc deficiency and zinc transporter dysregulation. J Nutr Biochem 24:353–359. https://doi.org/10.1016/j.jnutbio.2012.07.005
Wong CP, Rinaldi NA, Ho E (2015) Zinc deficiency enhanced inflammatory response by increasing immune cell activation and inducing IL6 promoter demethylation. Mol Nutr Food Res 59:991–999. https://doi.org/10.1002/mnfr.201400761
Wong CP, Dashner-Titus EJ, Alvarez SC, Chase TT, Hudson LG, Ho E (2019) Zinc deficiency and arsenic exposure can act both independently or cooperatively to affect zinc status, oxidative stress, and inflammatory response. Biol Trace Elem Res. https://doi.org/10.1007/s12011-019-1631-z
World Health Organization (2018) Arsenic. https://www.who.int/news-room/fact-sheets/detail/arsenic. Accessed 23 Dec 2018
Xu H et al (2016) Environmentally relevant concentrations of arsenite induce dose-dependent differential genotoxicity through poly(adp-ribose) polymerase inhibition and oxidative stress in mouse thymus cells. Toxicol Sci 149:31–41. https://doi.org/10.1093/toxsci/kfv211
Yan M, Song Y, Wong CP, Hardin K, Ho E (2008) Zinc deficiency alters DNA damage response genes in normal human prostate epithelial cells. J Nutr 138:667–673. https://doi.org/10.1093/jn/138.4.667
Zhou X, Cooper KL, Huestis J, Xu H, Burchiel SW, Hudson LG, Liu KJ (2016) S-nitrosation on zinc finger motif of PARP-1 as a mechanism of DNA repair inhibition by arsenite. Oncotarget 7:80482–80492. https://doi.org/10.18632/oncotarget.12613
Acknowledgements
We would like to thank Andy Ungerer for his technical assistance with the ICP-OES. This study was supported by a grant from the National Institutes of Health (1R01ES021100 NIH Revision Awards for Creating Virtual Consortium for Translational/Transdisciplinary Environmental Research ViCTER) to LH/EH, as well as the Oregon Agricultural Experimental Station (EH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, A.L., Beaver, L.M., Wong, C.P. et al. Zinc deficiency alters the susceptibility of pancreatic beta cells (INS-1) to arsenic exposure. Biometals 32, 845–859 (2019). https://doi.org/10.1007/s10534-019-00217-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-019-00217-0