Abstract
Objectives
To identify genes that affected protein expression in Chinese hamster ovary (CHO) cells was significant, and we identified the changes in the transcriptome and the functional gene sets that would contribute to increase expression of recombinant protein.
Results
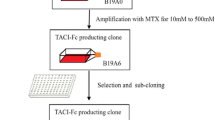
Here two sub-clones from a methotrexate-treated parental recombinant CHO cell line were selected. The two sub-clones, with different expression levels (qp were 42.8 pg/cell/day and 14.0 pg/cell/day), were analyzed through RNA-seq. More than 600 genes were identified as differently expressed, and we found that the differentially expressed genes were involved in processes such as RNA processing, transcription, protein catabolism, and protein transport. Among these, we cloned genes encoding proteins that were involved in transcription and protein transport to investigate their effect on protein production.
Conclusions
We found that some genes involved in transcription and protein transport would improve recombinant protein production in CHO cells.







Similar content being viewed by others
Abbreviations
- CHO cells:
-
Chinese hamster ovary cells
- MTX:
-
Methotrexate
- TACI:
-
The transmembrane activator and CAML interactor
- Fc:
-
The fragment crystallizable
- ELISA:
-
Enzyme linked immunosorbent assay
- IMDM:
-
Iscove’s modified Dulbecco’s medium
- FBS:
-
Fetal bovine serum
- qPCR:
-
Quantitative polymerase chain reaction
References
Abramian DS, Glebov OK (1981) Intraclonal heterogeneity and instability of CHO-K1 Chinese hamster cells in sensitivity to ultraviolet light and resistance to 8-azaguanine. Tsitologiia 23(9):1031–1040
Allegue C, Coll M, Mates J, Campuzano O, Iglesias A, Sobrino B, Brion M, Amigo J, Carracedo A, Brugada P, Brugada J, Brugada R (2015) Genetic analysis of arrhythmogenic diseases in the era of NGS: the complexity of clinical decision-making in brugada syndrome. PLoS ONE 10(7):e0133037
Allen GC, Spiker S, Thompson WF (2000) Use of matrix attachment regions (MARs) to minimize transgene silencing. Plant Mol Biol 43:361–376
Beckmann TF, Kramer O, Klausing S, Heinrich C, Thute T, Buntemeyer H, Hoffrogge R, Noll T (2012) Effects of high passage cultivation on CHO cells: a global analysis. Appl Microbiol Biotechnol 94(3):659–671
Benton T, Chen T, McEntee M, Fox B, King D, Crombie R, Thomas TC, Bebbington C (2002) The use of UCOE vectors in combination with a preadapted serum free, suspension cell line allows for rapid production of large quantities of protein. Cytotechnology 38:43–46
Butler M, Meneses-Acosta A (2012) Recent advances in technology supporting biopharmaceutical production from mammalian cells. Appl Microbiol Biotechnol 96(4):885–894
Chen K, Li D, Li H, Li B, Li J (2017) Genetic analysis of heterogeneous sub-clones in recombinant Chinese hamster ovary cells. Appl Microbiol Biotechnol 101(14):5785–5797
Davies SL, Lovelady CS, Grainger RK, Racher AJ, Young RJ, James DC (2013) Functional heterogeneity and heritability in CHO cell populations. Biotechnol Bioeng 110(1):260–274
Feichtinger J, Hernandez I, Fischer C, Hanscho M, Auer N, Hackl M, Jadhav V, Baumann M, Krempl PM, Schmidl C, Farlik M, Schuster M, Merkel A, Sommer A, Heath S, Rico D, Bock C, Thallinger GG, Borth N (2016) Comprehensive genome and epigenome characterization of CHO cells in response to evolutionary pressures and over time. Biotechnol Bioeng 113(10):2241–2253
Fomina-Yadlin D, Mujacic M, Maggiora K, Quesnell G, Saleem R, McGrew JT (2015) Transcriptome analysis of a CHO cell line expressing a recombinant therapeutic protein treated with inducers of protein expression. J Biotechnol 212:106–115
Gan W, Zhang C, Siu KY, Satoh A, Tanner JA, Yu S (2017) ULK1 phosphorylates Sec23A and mediates autophagy-induced inhibition of ER-to-Golgi traffic. BMC Cell Biol 18(1):22
Hammond S, Lee KH (2012) RNA interference of cofilin in Chinese hamster ovary cells improves recombinant protein productivity. Biotechnol Bioeng 109(2):528–535
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4(1):44–57
Kaas CS, Kristensen C, Betenbaugh MJ, Andersen MR (2015) Sequencing the CHO DXB11 genome reveals regional variations in genomic stability and haploidy. BMC Genomics 16:160
Kantardjieff A, Jacob NM, Yee JC, Epstein E, Kok YJ, Philp R, Betenbaugh M, Hu WS (2010) Transcriptome and proteome analysis of Chinese hamster ovary cells under low temperature and butyrate treatment. J Biotechnol 145(2):143–159
Kim JY, Kim YG, Lee GM (2012) CHO cells in biotechnology for production of recombinant proteins: current state and further potential. Appl Microbiol Biotechnol 93(3):917–930
Lee KH, Tsutsui T, Honda K, Asano R, Kumagai I, Ohtake H, Omasa T (2013) Generation of high- producing cell lines by overexpression of cell division cycle 25 homolog A in Chinese hamster ovary cells. J Biosci Bioeng 116(6):754–760
Li D, Xie K, Ding G, Li J, Chen K, Li H, Qian J, Jiang C, Fang J (2014) Tumor resistance to anti-VEGF therapy through up-regulation of VEGF-C expression. Cancer Lett 346(1):45–52
Li H, Chen K, Wang Z, Li D, Lin J, Yu C, Yu F, Wang X, Huang L, Jiang C, Gu H, Fang J (2016) Genetic analysis of the clonal stability of Chinese hamster ovary cells for recombinant protein production. Mol BioSyst 12(1):102–109
Nagalakshmi U, Wang Z, Waern K, Shou C, Raha D, Gerstein M, Snyder M (2008) The transcriptional landscape of the yeast genome defined by RNA sequencing. Science 320(5881):1344–1349
Nishimiya D, Ogura Y, Sakurai H, Takahashi T (2012) Identification of antibody-interacting proteins that contribute to the production of recombinant antibody in mammalian cells. Appl Microbiol Biotechnol 96(4):971–979
Nishimiya D, Mano T, Miyadai K, Yoshida H, Takahashi T (2013) Overexpression of CHOP alone and in combination with chaperones is effective in improving antibody production in mammalian cells. Appl Microbiol Biotechnol 97(6):2531–2539
Omasa T, Takami T, Ohya T, Kiyama E, Hayashi T, Nishii H, Miki H, Kobayashi K, Honda K, Ohtake H (2008) Overexpression of GADD34 enhances production of recombinant human antithrombin III in Chinese hamster ovary cells. J Biosci Bioeng 106(6):568–573
Park JH, Wang Z, Jeong HJ, Park HH, Kim BG, Tan WS, Choi SS, Park TH (2012) Enhancement of recombinant human EPO production and glycosylation in serum-free suspension culture of CHO cells through expression and supplementation of 30Kc19. Appl Microbiol Biotechnol 96(3):671–683
Peng RW, Abellan E, Fussenegger M (2011) Differential effect of exocytic SNAREs on the production of recombinant proteins in mammalian cells. Biotechnol Bioeng 108(3):611–620
Pilbrough W, Munro TP, Gray P (2009) Intraclonal protein expression heterogeneity in recombinant CHO cells. PLoS ONE 4(12):e8432
Takasea K, Ohtsukia T, Migitaa O, Torub M, Inadac T, Yamakawa-Kobayashia K, Arinamia T (2001) Association of ZNF74 gene genotypes with age-at-onset of schizophrenia. Schizophr Res 52:161–165
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28(5):511–515
Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, Pimentel H, Salzberg SL, Rinn JL, Pachter L (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7(3):562–578
Vishwanathan N, Le H, Jacob NM, Tsao Y-S, Ng S-W, Loo B, Liu Z, Kantardjieff A, Hu W-S (2014) Transcriptome dynamics of transgene amplification in Chinese Hamster Ovary Cells. Biotechnol Bioeng 111(3):518–528
Xu X, Nagarajan H, Lewis NE, Pan S, Cai Z, Liu X, Chen W, Xie M, Wang W, Hammond S, Andersen MR, Neff N, Passarelli B, Koh W, Fan HC, Wang J, Gui Y, Lee KH, Betenbaugh MJ, Quake SR, Famili I, Palsson BO, Wang J (2011) The genomic sequence of the Chinese hamster ovary (CHO)-K1 cell line. Nat Biotechnol 29(8):735–741
Acknowledgements
This study was supported by grants from the National Basic Research Program of China (973 Program) 2015CB553706, the National Natural Science Foundation of China (NSFC31270987 and NSFC31470896), Suzhou Applicational Basic Project, China (SYG201509), the Natural Science Foundation of Jiangsu Province(BK201150278), and the Fundamental Research Funds for the Central Universities 22120170036. We would like to thank Editage [www.editage.cn] for English language editing.
Supporting information
Supplementary Table 1—Primers used for amplifying each gene for construction of the expression plasmids.
Supplementary Table 2—Overall transcriptome and gene ontology (GO) analysis of the genes differentially expressed by the CHO-L and CHO-H cell lines.
Supplementary Table 3—All genes differentially expressed in the CHO-L and CHO-H cell lines.
Supplementary Fig. 1—Effect of selected genes on the expression of the Fc fragment in CHO-K1 cells
Supplementary Fig. 2—Effect of selected genes on the expression of the Herceptin in CHO-K1 cells.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest with the contents of this article. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, K., Li, D., Li, H. et al. Improved recombinant protein production by regulation of transcription and protein transport in Chinese hamster ovary cells. Biotechnol Lett 41, 719–732 (2019). https://doi.org/10.1007/s10529-019-02681-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02681-7