Abstract
Objective
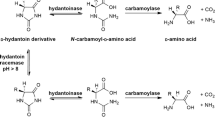
To investigate the expression and immobilization of recombinant cis-epoxysuccinate hydrolase (ESH), and its application in the biological production of l-(+)-tartaric acid.
Results
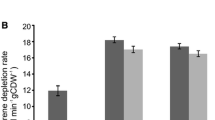
E. coli BL21 (DE3)/pET11a-ESH (His) was engineered to express recombinant ESH. The enzyme had an activity of 262 U mg−1. The recombinant ESH was immobilized on agarose Ni-IDA matrix with metal ion affinity interaction to improve its thermostability and pH stability. The immobilization efficiency and activity yield were 94 and 95%, respectively. The specific catalytic efficiency of immobilized ESH was 104 mg U−1 h−1 during the continuous enzymatic production process.
Conclusion
ESH with a histidine tag was immobilized and used for the continuous production of l-(+)-tartaric acid.




Similar content being viewed by others
References
Bao WN, Pan HF, Zhang ZH, Cheng YQ, Xie ZP, Zhang JG, Li YQ (2014) Analysis of essential amino acid residues for catalytic activity of cis-epoxysuccinate hydrolase from Bordetella sp. BK-52. Appl Microbiol Biotechnol 98:1641–1649
Cao L, van Langen L, Sheldon RA (2003) Immobilized enzymes:carrier-bound or carrier-free. Curr Opin Biotech 14:387–394
Liu ZQ, Li Y, Xu YY, Ping LF, Zheng YG (2007) Cloning, sequencing, expression of a novel epoxide hydrolase gene from Rhodococcus opacus in Escherichia coli and characterization of enzyme. Appl Microbiol Biotechnol 74:99–106
Lynch HC, Yang Y (2004) Degradation products of clavulanic acid promote clavulanic acid production in cultures of Streptomyces clavuligerus. Enzyme Microb Technol 34:48–54
Mateo C, Palomo JM, Fernández-Lorente G, Guisán JM, Fernández-Lafuente R (2007) Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzyme Microb Technol 40:1451–1463
Pan HF, Xie ZP, Bao WN, Cheng YQ, Zhang JG, Li YQ (2011) Site-directed mutagenesis of epoxide hydrolase to probe catalytic amino acid residues and reaction mechanism. FEBS Lett 585:2545–2550
Steinreiber A, Faber K (2001) Microbial epoxide hydroxylases for preparative biotransformations. Curr Opin Biotechnol 12:552–558
Tischer W, Wedekind F (1999) Immobilized enzymes: methods and applications. Top Curr Chem 200:95–126
Vasu V, Kumaresan J, Babu MG, Meenakshisundaram S (2012) Active site analysis of cis-epoxysuccinate hydrolase from Nocardia tartaricans using homology modeling and site-directed mutagenesis. Appl Microbiol Biotechnol 93:2377–2386
Wang S, Cui GZ, Song XF, Feng YG, Cui Q (2012) Efficiency and stability enhancement of cis-epoxysuccinic acid hydrolase by fusion with a carbohydrate binding module and immobilization onto cellulose. Appl Biochem Biotechnol 168:708–717
Wang ZQ, Wang YS, Su ZG (2013) Purification and characterization of a cis-epoxysuccinic acid hydrolase from Nocardia tartaricans CAS-52, and expression in Escherichia coli. Appl Microbiol Biotechnol 97:2433–2441
Wang ZQ, Wang YS, Shi H, Su ZG (2014) Improvement of the production efficiency of l-(+)-tartaric acid by heterogeneous whole-cell bioconversion. Appl Biochem Biotechnol 172:3989–4001
Willaert R, de Vuyst L (2006) Continuous production of L (+)-tartaric acid from cis-epoxysuccinate using a membrane recycle reactor. Appl Microbiol Biotechnol 71:155–163
Acknowledgements
This work was supported by the Major State Basic Research Development Program of China, Grant No. 2013CB733604.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Z., Su, M., Li, Y. et al. Production of tartaric acid using immobilized recominant cis-epoxysuccinate hydrolase. Biotechnol Lett 39, 1859–1863 (2017). https://doi.org/10.1007/s10529-017-2419-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-017-2419-x