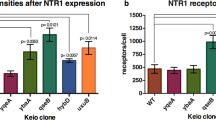
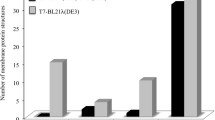
Abstract
The stress response of E. coli to the expression of two recombinant membrane proteins, the E. coli AAA+ protease FtsH and the human G-protein coupled receptor CB1, was examined using several members of a promoter-GFP library. Several genes from the heat-shock and envelope stress regulons (rpoH, clpP, lon, and ftsH) were strongly induced by expression of either membrane protein. Flow cytometry was used to monitor the real-time dynamics of the transcription of these reporter genes in response to membrane protein expression. Co-expression of CB1 and FtsH led to an additive response in these four reporter genes suggesting that the stresses may be occurring via different physiological mechanisms.




Similar content being viewed by others
References
Bartfai T, Benovic JL, Bockaert J et al (2004) The state of GPCR research in 2004. Nat Rev Drug Discov 3:574–626
Cha HJ, Srivastava R, Vakharia VM, Rao G, Bentley WE (1999) Green fluorescent protein as a noninvasive stress probe in resting Escherichia coli cells. Appl Environ Microbiol 65:409–414
Cherezov V, Rosenbaum DM, Hanson MA et al (2007) High-resolution crystal structure of an engineered human beta(2)-adrenergic G protein-coupled receptor. Science 318:1258–1265
Daley DO, Rapp M, Granseth E et al (2005) Global topology analysis of the Escherichia coli inner membrane proteome. Science 308:1321–1323
Funabashi H, Haruyama T, Mie M et al (2002) Non-destructive monitoring of rpoS promoter activity as stress marker for evaluating cellular physiological status. J Biotechnol 95:85–93
Gruber TM, Gross CA (2003) Multiple sigma subunits and the partitioning of bacterial transcription space. Annu Rev Microbiol 57:441–466
Guzman LM, Belin D, Carson MJ, Beckwith J (1995) Tight regulation, modulation, and high-level expression by vectors containing the arabinose P-Bad promoter. J Bacteriol 177:4121–4130
Harle C, Kim I, Angerer A, Braun V (1995) Signal transfer through 3 compartments—transcription initiation of the Escherichia coli ferric citrate transport-system from the cell-surface. EMBO J 14:1430–1438
Ito K, Akiyama Y (2005) Cellular functions, mechanism of action, and regulation of FtsH protease. Annu Rev Microbiol 59:211–231
Kostrzynska M, Leung KT, Lee H, Trevors JT (2002) Green fluorescent protein-based biosensor for detecting SOS-inducing activity of genotoxic compounds. J Microbiol Methods 48:43–51
Kundu TK, Kusano S, Ishihama A (1997) Promoter selectivity of Escherichia coli RNA polymerase sigma(F) holoenzyme involved in transcription of flagellar and chemotaxis genes. J Bacteriol 179:4264–4269
Link AJ, Skretas G, Strauch EM, Chari NS, Georgiou G (2008) Efficient expression of membrane-integrated and detergent-soluble G protein-coupled receptors in E. coli. Protein Sci 17:1857–1863
Lu CH, Albano CR, Bentley WE, Rao GV (2005) Quantitative and kinetic study of oxidative stress regulons using green fluorescent protein. Biotechnol Bioeng 89:574–587
Palczewski K, Kumasaka T, Hori T et al (2000) Crystal structure of rhodopsin: a G protein-coupled receptor. Science 289:739–745
Phillips PC, Otto SP, Whitlock MC (2000) Beyond the average: the evolutionary importance of gene interactions and variability of epistatic effects in epistasis and the evolutionary process. Oxford University Press, New York
Rasmussen SGF, Choi HJ, Rosenbaum DM et al (2007) Crystal structure of the human beta(2) adrenergic G-protein-coupled receptor. Nature 450:383–387
Reitzer L, Schneider BL (2001) Metabolic context and possible physiological themes of sigma(54)-dependent genes in Escherichia coli. Microbiol Mol Biol Rev 65:422–444
Ruiz N, Silhavy TJ (2005) Sensing external stress: watchdogs of the Escherichia coli cell envelope. Curr Opin Microbiol 8:122–126
Santini CL, Bernadac A, Zhang M et al (2001) Translocation of jellyfish green fluorescent protein via the Tat system of Escherichia coli and change of its periplasmic localization in response to osmotic up-shock. J Biol Chem 276:8159–8164
Skerra A (1994) Use of the tetracycline promoter for the tightly regulated production of a murine antibody fragment in Escherichia coli. Gene 151:131–135
Tomoyasu T, Gamer J, Bukau B et al (1995) Escherichia coli FtsH is a membrane-bound, ATP-dependent protease which degrades the heat-shock transcription factor sigma(32). EMBO J 14:2551–2560
Wagner S, Bader ML, Drew D, de Gier JW (2006) Rationalizing membrane protein overexpression. Trends Biotechnol 24:364–371
Wagner S, Baars L, Ytterberg AJ et al (2007) Consequences of membrane protein overexpression in Escherichia coli. Mol Cell Proteomics 6:1527–1550
Wang QP, Kaguni JM (1989) A novel sigma-factor is involved in expression of the Rpoh gene of Escherichia coli. J Bacteriol 171:4248–4253
Weber H, Polen T, Heuveling J, Wendisch VF, Hengge R (2005) Genome-wide analysis of the general stress response network in Escherichia coli: sigma(S)-dependent genes, promoters, and sigma factor selectivity. J Bacteriol 187:1591–1603
Zaslaver A, Bren A, Ronen M et al (2006) A comprehensive library of fluorescent transcriptional reporters for Escherichia coli. Nat Methods 3:623–628
Zhao K, Liu MZ, Burgess RR (2005) The global transcriptional response of Escherichia coli to induced sigma(32) protein involves sigma(32) regulon activation followed by inactivation and degradation of sigma(32) in vivo. J Biol Chem 280:17758–17768
Acknowledgments
This research was supported by Princeton University startup funds and by the Lidow Senior Thesis fund. The authors thank Dr. Eva-Maria Strauch (University of Washington) for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, L.Y., James Link, A. Stress responses to heterologous membrane protein expression in Escherichia coli . Biotechnol Lett 31, 1775–1782 (2009). https://doi.org/10.1007/s10529-009-0075-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-009-0075-5