Abstract
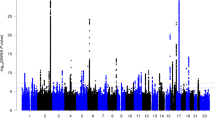
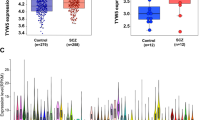
Handedness is an elusive human behavioral phenotypes and the genetic basis of it remains unclear until now. The aim of this study is to evaluate the genetic correlations between left-handedness and multiple mental disorders, and explored the genes detected by genetic correlations. In this study, linkage disequilibrium score regression (LDSC) analysis was conducted to evaluate the genetic correlations between left-handedness and multiple mental disorders. The significant genetic correlation was only observed between left-handedness and schizophrenia (SCZ). For the observed genetic correlation, transcriptome-wide association study (TWAS) was performed to identify the genes associated with left-handedness and SCZ, including brain RNA-seq (CBR) and brain RNA-seq splicing (CBRS). We detected several common genes associated with both left-handedness and SCZ, such as YWHAH, MAPT and ANO10. The common genes shared by left-handedness and SCZ were subjected to gene set enrichment analysis. Our study provides a novel clue for understanding the genetic correlation between left-handedness and SCZ.

Similar content being viewed by others
References
Annett M (1998) Handedness and cerebral dominance: the right shift theory. J Neuropsychiatry Clin Neurosci 10:459–469. https://doi.org/10.1176/jnp.10.4.459
Bian Y, Bian QT, Wang ZX, Hospital BH (2014) A case-control study of handedness distribution in patients with schizophrenia. Journal of Clinical Psychiatry
Buliksullivan B et al (2015) An atlas of genetic correlations across human diseases and traits. Nat Genet 47:1236–1241
Bulik-Sullivan BK, Loh PR, Finucane HK, Ripke S, Yang J (2015) LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat Genet 47:291–295. https://doi.org/10.1038/ng.3211
Canela-Xandri O, Rawlik K (2018) An atlas of genetic associations in UK Biobank. Nat Genet 50(11):1593–1599. https://doi.org/10.1038/s41588-018-0248-z
Carter CJ (2007) eIF2B and oligodendrocyte survival: where nature and nurture meet in bipolar disorder and schizophrenia? Schizophr Bull 33:1343–1353. https://doi.org/10.1093/schbul/sbm007
Clyde D (2017) Disease genomics: transitioning from association to causation with eQTLs. Nat Rev Genet 18:271. https://doi.org/10.1038/nrg.2017.22
Consortium, ASDWGoTPG (2017) Meta-analysis of GWAS of over 16,000 individuals with autism spectrum disorder highlights a novel locus at 10q24.32 and a significant overlap with schizophrenia. Mol Autism 8:21. https://doi.org/10.1186/s13229-017-0137-9
Consortium, BDaSWGotPGCEadrveBDaSWGotPG (2018) Genomic dissection of bipolar disorder and schizophrenia, including 28 subphenotypes. Cell 173:1705–1715.e1716. https://doi.org/10.1016/j.cell.2018.05.046
Dollfus S et al (2012) Familial sinistrality and handedness in patients with first episode schizophrenia: the EUFEST study. Laterality 17:217–224. https://doi.org/10.1080/1357650x.2011.558510
Duncan LE et al (2018) Largest GWAS of PTSD (N=20 070) yields genetic overlap with schizophrenia and sex differences in heritability. Mol Psychiatry 23:666–673. https://doi.org/10.1038/mp.2017.77
Grover D et al (2010) Family-based association of YWHAH in psychotic bipolar disorder American. Am J Med Genet B 150B:977–983
Gusev A, Ko A (2016) Integrative approaches for large-scale transcriptome-wide association studies. Nat Genet 48:245–252. https://doi.org/10.1038/ng.3506
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57. https://doi.org/10.1038/nprot.2008.211
Jiang T et al (2017) Transcriptome-wide association study revealed two novel genes associated with nonobstructive azoospermia in a Chinese population. Fertil Steril 108:1056–1062.e1054. https://doi.org/10.1016/j.fertnstert.2017.09.023
Karayiorgou M, Simon TJ, Gogos JA (2010) 22q11.2 microdeletions: linking DNA structural variation to brain dysfunction and schizophrenia. Nat Rev Neurosci 11:402–416. https://doi.org/10.1038/nrn2841
Knaus TA, Kamps J, Foundas AL (2016) Handedness in children with autism spectrum disorder perceptual and motor skills. Percept Mot Skills 122:542–559. https://doi.org/10.1177/0031512516637021
Lang B, Alrahbeni TM, Clair DS, Blackwood DH, McCaig CD, Shen S (2012) HDAC9 is implicated in schizophrenia and expressed specifically in post-mitotic neurons but not in adult neural stem cells. Am J Stem Cells 1:31–41
Larimore J et al (2011) The schizophrenia susceptibility factor dysbindin and its associated complex sort cargoes from cell bodies to the synapse. Mol Biol Cell 22:4854–4867. https://doi.org/10.1091/mbc.E11-07-0592
Lin GN et al (2015) Spatiotemporal 16p11.2 protein network implicates cortical late mid-fetal brain development and KCTD13-Cul3-RhoA pathway in psychiatric diseases. Neuron 85:742–754. https://doi.org/10.1016/j.neuron.2015.01.010
Marzullo G, Boklage CE (2011) Bimodal rhythms of general conceptions and the birth-month phenomenon in schizophrenia, neural tube defects, and laterality: a solstitial hypothesis. Birth Defects Res A 91:249–257. https://doi.org/10.1002/bdra.20806
Marzullo G, Fraser FC (2005) Similar rhythms of seasonal conceptions in neural tube defects and schizophrenia: a hypothesis of oxidant stress and the photoperiod. Birth Defects Res A 73:1–5. https://doi.org/10.1002/bdra.20100
Momeni P et al (2010) Familial early onset frontotemporal dementia caused by a novel S356T MAPT mutation, initially diagnosed as schizophrenia. Clin Neurol Neurosurg 112:917–920. https://doi.org/10.1016/j.clineuro.2010.07.015
Neale BM et al (2010) Meta-analysis of genome-wide association studies of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 49:884–897. https://doi.org/10.1016/j.jaac.2010.06.008
Nicolae DL, Gamazon E, Zhang W, Duan S, Dolan ME, Cox NJ (2010) Trait-associated SNPs are more likely to be eQTLs: annotation to enhance discovery from GWAS. PLoS Genet 6:e1000888. https://doi.org/10.1371/journal.pgen.1000888
Oldmeadow C et al (2014) Combined analysis of exon splicing and genome wide polymorphism data predict schizophrenia risk loci. J Psychiatr Res 52:44–49. https://doi.org/10.1016/j.jpsychires.2014.01.011
Otowa T et al (2016) Meta-analysis of genome-wide association studies of anxiety disorders. Mol Psychiatry 21:1485. https://doi.org/10.1038/mp.2016.11
Pappa I et al (2016) A genome-wide approach to children's aggressive behavior: the EAGLE consortium. Am J Med Genet B 171:562–572. https://doi.org/10.1002/ajmg.b.32333
Ravichandran C, Shinn AK, Ongur D, Perlis RH, Cohen B (2017) Frequency of non-right-handedness in bipolar disorder and schizophrenia Psychiatry research. Psychiatry Res 253:267–269. https://doi.org/10.1016/j.psychres.2017.04.011
Roveletlecrux A et al (2010) Frontotemporal dementia phenotype associated with MAPT gene duplication. J Alzheimers Dis 21:897
Shimizu A, Endo M, Yamaguchi N, Torii H, Isaki K (1985) Hand preference in schizophrenics and handedness conversion in their childhood. Acta Psychiatr Scand 72:259–265. https://doi.org/10.1111/j.1600-0447.1985.tb02604.x
Simoes EN, Carvalho ALN, Schmidt SL (2017) What does handedness reveal about ADHD? An analysis based on CPT performance. Res Dev Disabil 65:46–56. https://doi.org/10.1016/j.ridd.2017.04.009
Strockens F, Gunturkun O, Ocklenburg S (2013) Limb preferences in non-human vertebrates. Laterality 18:536–575. https://doi.org/10.1080/1357650x.2012.723008
Toyooka K et al (1999) 14-3-3 protein eta chain gene (YWHAH) polymorphism and its genetic association with schizophrenia. Am J Med Genet 88:164–167
Tsuang HC et al (2011) Handedness and schizotypy in non-psychotic relatives of patients with schizophrenia. Laterality 16:690–706. https://doi.org/10.1080/1357650x.2010.511646
Wray NR et al (2018) Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat Genet 50:668–681. https://doi.org/10.1038/s41588-018-0090-3
Yan Z, Kim E (2016) Synaptic Actin Dysregulation, a convergent mechanism of mental disorders? J Neurosci 36:11411–11417
Zammit S, Lewis S, Gunnell D, Smith GD (2007) Schizophrenia and neural tube defects: comparisons from an epidemiological perspective. Schizophr Bull 33:853–858. https://doi.org/10.1093/schbul/sbl041
Zanardini R, Ciani M, Benussi L, Ghidoni R (2016) Molecular pathways bridging frontotemporal lobar degeneration and psychiatric disorders. Front Aging Neurosci 8:10. https://doi.org/10.3389/fnagi.2016.00010
Acknowledgements
This work was supported by the National Natural Scientific Foundation of China [Grant Nos. 81472925, 81673112]; the Key projects of international cooperation among governments in scientific and technological innovation [2016YFE0119100]; and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
This article does not contain any studies with human participants and there is no need for informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, B., Liang, C., Li, P. et al. Evaluating the Genetic Correlations Between Left-Handedness and Mental Disorder Using Linkage Disequilibrium Score Regression and Transcriptome-Wide Association Study. Biochem Genet 58, 348–358 (2020). https://doi.org/10.1007/s10528-020-09952-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-020-09952-3