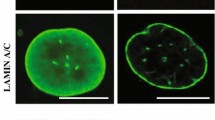
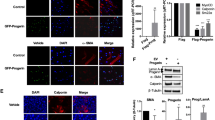
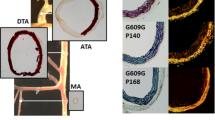
Abstract
Hutchinson-Gilford progeria syndrome (HGPS) is a premature aging disorder caused by a de novo genetic mutation that leads to the accumulation of a splicing isoform of lamin A termed progerin. Progerin expression alters the organization of the nuclear lamina and chromatin. The life expectancy of HGPS patients is severely reduced due to critical cardiovascular defects. Progerin also accumulates in an age-dependent manner in the vascular cells of adults that do not carry genetic mutations associated with HGPS. The molecular mechanisms that lead to vascular dysfunction in HGPS may therefore also play a role in vascular aging. The vascular phenotypic and molecular changes observed in HGPS are strikingly similar to those seen with age, including increased senescence, altered mechanotransduction and stem cell exhaustion. This article discusses the similarities and differences between age-dependent and HGPS-related vascular aging to highlight the relevance of HGPS as a model for vascular aging. Induced pluripotent stem cells derived from HGPS patients are suggested as an attractive model to study vascular aging in order to develop novel approaches to treat cardiovascular disease.

Similar content being viewed by others
Abbreviations
- EC:
-
Endothelial cell
- ECM:
-
Extracellular matrix
- EPC:
-
Endothelial progenitor cell
- ESC:
-
Embryonic stem cell
- HGPS:
-
Hutchinson–Gilford progeria syndrome
- iPSC:
-
Induced pluripotent stem cell
- MSC:
-
Mesenchymal stem cell
- NF:
-
Nuclear factor
- NO:
-
Nitric oxide
- ROS:
-
Reactive oxygen species
- VSMC:
-
Vascular smooth muscle cell
References
Alt EU et al (2012) Aging alters tissue resident mesenchymal stem cell properties. Stem Cell Res 8:215–225. doi:10.1016/j.scr.2011.11.002
Andres V, Gonzalez JM (2009) Role of A-type lamins in signaling, transcription, and chromatin organization. J Cell Biol 187:945–957. doi:10.1083/jcb.200904124
Arancio W, Pizzolanti G, Genovese SI, Pitrone M, Giordano C (2014) Epigenetic involvement in Hutchinson–Gilford progeria syndrome: a mini-review. Gerontology 60:197–203. doi:10.1159/000357206
Bellantuono I, Sanguinetti G, Keith WN (2012) Progeroid syndromes: models for stem cell aging? Biogerontology 13:63–75. doi:10.1007/s10522-011-9347-2
Benson EK, Lee SW, Aaronson SA (2010) Role of progerin-induced telomere dysfunction in HGPS premature cellular senescence. J Cell Sci 123:2605–2612. doi:10.1242/jcs.067306
Blasco MA (2005) Telomeres and human disease: ageing, cancer and beyond. Nat Rev Genet 6:611–622. doi:10.1038/nrg1656
Blondel S et al (2014) Induced pluripotent stem cells reveal functional differences between drugs currently investigated in patients with hutchinson-gilford progeria syndrome. Stem Cells Transl Med 3:510–519. doi:10.5966/sctm.2013-0168
Bond AR, Iftikhar S, Bharath AA, Weinberg PD (2011) Morphological evidence for a change in the pattern of aortic wall shear stress with age. Arterioscler Thromb Vasc Biol 31:543–550. doi:10.1161/ATVBAHA.110.219683
Bonello-Palot N et al (2014) Prelamin A accumulation in endothelial cells induces premature senescence and functional impairment. Atherosclerosis 237:45–52. doi:10.1016/j.atherosclerosis.2014.08.036
Booth-Gauthier EA, Du V, Ghibaudo M, Rape AD, Dahl KN, Ladoux B (2013) Hutchinson–Gilford progeria syndrome alters nuclear shape and reduces cell motility in three dimensional model substrates. Integr Biol (Camb) 5:569–577. doi:10.1039/c3ib20231c
Burtner CR, Kennedy BK (2010) Progeria syndromes and ageing: what is the connection? Nat Rev Mol Cell Biol 11:567–578. doi:10.1038/nrm2944
Candelario J, Sudhakar S, Navarro S, Reddy S, Comai L (2008) Perturbation of wild-type lamin A metabolism results in a progeroid phenotype. Aging Cell 7:355–367. doi:10.1111/j.1474-9726.2008.00393.x
Candelario J, Borrego S, Reddy S, Comai L (2011) Accumulation of distinct prelamin A variants in human diploid fibroblasts differentially affects cell homeostasis. Exp Cell Res 317:319–329. doi:10.1016/j.yexcr.2010.10.014
Cao K, Capell BC, Erdos MR, Djabali K, Collins FS (2007) A lamin A protein isoform overexpressed in Hutchinson–Gilford progeria syndrome interferes with mitosis in progeria and normal cells. Proc Natl Acad Sci USA 104:4949–4954. doi:10.1073/pnas.0611640104
Cao K et al (2011) Progerin and telomere dysfunction collaborate to trigger cellular senescence in normal human fibroblasts. J Clin Invest 121:2833–2844. doi:10.1172/JCI43578
Chang E, Harley CB (1995) Telomere length and replicative aging in human vascular tissues. Proc Natl Acad Sci USA 92:11190–11194
Chang EI et al (2007) Age decreases endothelial progenitor cell recruitment through decreases in hypoxia-inducible factor 1alpha stabilization during ischemia. Circulation 116:2818–2829. doi:10.1161/CIRCULATIONAHA.107.715847
Chiba S (2006) Notch signaling in stem cell systems. Stem Cells 24:2437–2447. doi:10.1634/stemcells.2005-0661
Constantinescu D, Gray HL, Sammak PJ, Schatten GP, Csoka AB (2006) Lamin A/C expression is a marker of mouse and human embryonic stem cell differentiation. Stem Cells 24:177–185. doi:10.1634/stemcells.2004-0159
Csoka AB et al (2004) Genome-scale expression profiling of Hutchinson-Gilford progeria syndrome reveals widespread transcriptional misregulation leading to mesodermal/mesenchymal defects and accelerated atherosclerosis. Aging Cell 3:235–243. doi:10.1111/j.1474-9728.2004.00105.x
Dahl KN, Scaffidi P, Islam MF, Yodh AG, Wilson KL, Misteli T (2006) Distinct structural and mechanical properties of the nuclear lamina in Hutchinson–Gilford progeria syndrome. Proc Natl Acad Sci USA 103:10271–10276. doi:10.1073/pnas.0601058103
Datta S, Snow CJ, Paschal BM (2014) A pathway linking oxidative stress and the Ran GTPase system in progeria. Mol Biol Cell 25:1202–1215. doi:10.1091/mbc.E13-07-0430
de Castro Bras LE, Toba H, Baicu CF, Zile MR, Weintraub ST, Lindsey ML, Bradshaw AD (2014) Age and SPARC change the extracellular matrix composition of the left ventricle. Biomed Res Int 2014:810562. doi:10.1155/2014/810562
De Sandre-Giovannoli A et al (2003) Lamin a truncation in Hutchinson–Gilford progeria. Science 300:2055. doi:10.1126/science.1084125
De Vos WH et al (2010) Increased plasticity of the nuclear envelope and hypermobility of telomeres due to the loss of A-type lamins. Biochim Biophys Acta 1800:448–458. doi:10.1016/j.bbagen.2010.01.002
Decker ML, Chavez E, Vulto I, Lansdorp PM (2009) Telomere length in Hutchinson–Gilford progeria syndrome. Mech Ageing Dev 130:377–383. doi:10.1016/j.mad.2009.03.001
Deschenes-Simard X et al (2013) Tumor suppressor activity of the ERK/MAPK pathway by promoting selective protein degradation. Genes Dev 27:900–915. doi:10.1101/gad.203984.112
Eckersley-Maslin MA, Bergmann JH, Lazar Z, Spector DL (2013) Lamin A/C is expressed in pluripotent mouse embryonic stem cells. Nucleus 4:53–60. doi:10.4161/nucl.23384
Eckes B et al (1998) Impaired mechanical stability, migration and contractile capacity in vimentin-deficient fibroblasts. J Cell Sci 111(Pt13):1897–1907
Eriksson M et al (2003) Recurrent de novo point mutations in lamin A cause Hutchinson-Gilford progeria syndrome. Nature 423:293–298. doi:10.1038/nature01629
Espada J et al (2008) Nuclear envelope defects cause stem cell dysfunction in premature-aging mice. J Cell Biol 181:27–35. doi:10.1083/jcb.200801096
Esue O, Carson AA, Tseng Y, Wirtz D (2006) A direct interaction between actin and vimentin filaments mediated by the tail domain of vimentin. J Biol Chem 281:30393–30399. doi:10.1074/jbc.M605452200
Evanko SP, Angello JC, Wight TN (1999) Formation of hyaluronan- and versican-rich pericellular matrix is required for proliferation and migration of vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 19:1004–1013
Flores I, Canela A, Vera E, Tejera A, Cotsarelis G, Blasco MA (2008) The longest telomeres: a general signature of adult stem cell compartments. Genes Dev 22:654–667. doi:10.1101/gad.451008
Frangos SG, Gahtan V, Sumpio B (1999) Localization of atherosclerosis: role of hemodynamics. Arch Surg 134:1142–1149
Fyhrquist F, Saijonmaa O, Strandberg T (2013) The roles of senescence and telomere shortening in cardiovascular disease. Nat Rev Cardiol 10:274–283. doi:10.1038/nrcardio.2013.30
Geiger H, Denkinger M, Schirmbeck R (2014) Hematopoietic stem cell aging. Curr Opin Immunol 29:86–92. doi:10.1016/j.coi.2014.05.002
Gerhard-Herman M et al (2012) Mechanisms of premature vascular aging in children with Hutchinson–Gilford progeria syndrome. Hypertension 59:92–97. doi:10.1161/HYPERTENSIONAHA.111.180919
Goldman RD et al (2004) Accumulation of mutant lamin A causes progressive changes in nuclear architecture in Hutchinson–Gilford progeria syndrome. Proc Natl Acad Sci USA 101:8963–8968. doi:10.1073/pnas.0402943101
Halaschek-Wiener J, Brooks-Wilson A (2007) Progeria of stem cells: stem cell exhaustion in Hutchinson-Gilford progeria syndrome. J Gerontol A Biol Sci Med Sci 62:3–8
Harten IA et al (2011) Age-dependent loss of MMP-3 in Hutchinson-Gilford progeria syndrome. J Gerontol A Biol Sci Med Sci 66:1201–1207. doi:10.1093/gerona/glr137
Hayashi T et al (2006) Endothelial cellular senescence is inhibited by nitric oxide: implications in atherosclerosis associated with menopause and diabetes. Proc Natl Acad Sci USA 103:17018–17023. doi:10.1073/pnas.0607873103
Hazel AL, Pedley TJ (2000) Vascular endothelial cells minimize the total force on their nuclei. Biophys J 78:47–54. doi:10.1016/S0006-3495(00)76571-4
Heiss C, Keymel S, Niesler U, Ziemann J, Kelm M, Kalka C (2005) Impaired progenitor cell activity in age-related endothelial dysfunction. J Am Coll Cardiol 45:1441–1448. doi:10.1016/j.jacc.2004.12.074
Helmlinger G, Geiger RV, Schreck S, Nerem RM (1991) Effects of pulsatile flow on cultured vascular endothelial cell morphology. J Biomech Eng 113:123–131
Hennekam RC (2006) Hutchinson–Gilford progeria syndrome: review of the phenotype. Am J Med Genet A 140:2603–2624. doi:10.1002/ajmg.a.31346
Hernandez L et al (2010) Functional coupling between the extracellular matrix and nuclear lamina by Wnt signaling in progeria. Dev Cell 19:413–425. doi:10.1016/j.devcel.2010.08.013
Ho JC et al (2011) Generation of induced pluripotent stem cell lines from 3 distinct laminopathies bearing heterogeneous mutations in lamin A/C. Aging (Albany NY) 3:380–390
Hoffmann J, Haendeler J, Aicher A, Rossig L, Vasa M, Zeiher AM, Dimmeler S (2001) Aging enhances the sensitivity of endothelial cells toward apoptotic stimuli: important role of nitric oxide. Circ Res 89:709–715
Huang S, Chen L, Libina N, Janes J, Martin GM, Campisi J, Oshima J (2005) Correction of cellular phenotypes of Hutchinson-Gilford Progeria cells by RNA interference. Hum Genet 118:444–450. doi:10.1007/s00439-005-0051-7
Huang S, Risques RA, Martin GM, Rabinovitch PS, Oshima J (2008) Accelerated telomere shortening and replicative senescence in human fibroblasts overexpressing mutant and wild-type lamin A. Exp Cell Res 314:82–91. doi:10.1016/j.yexcr.2007.08.004
Humphrey JD, Dufresne ER, Schwartz MA (2014) Mechanotransduction and extracellular matrix homeostasis. Nat Rev Mol Cell Biol 15:802–812. doi:10.1038/nrm3896
Kalinowski A, Yaron PN, Qin Z, Shenoy S, Buehler MJ, Losche M, Dahl KN (2014) Interfacial binding and aggregation of lamin A tail domains associated with Hutchinson–Gilford progeria syndrome. Biophys Chem 195:43–48. doi:10.1016/j.bpc.2014.08.005
Kudlow BA, Stanfel MN, Burtner CR, Johnston ED, Kennedy BK (2008) Suppression of proliferative defects associated with processing-defective lamin A mutants by hTERT or inactivation of p53. Mol Biol Cell 19:5238–5248. doi:10.1091/mbc.E08-05-0492
Kurz DJ, Decary S, Hong Y, Trivier E, Akhmedov A, Erusalimsky JD (2004) Chronic oxidative stress compromises telomere integrity and accelerates the onset of senescence in human endothelial cells. J Cell Sci 117:2417–2426. doi:10.1242/jcs.01097
Lammerding J et al (2004) Lamin A/C deficiency causes defective nuclear mechanics and mechanotransduction. J Clin Invest 113:370–378. doi:10.1172/JCI19670
Lee SJ, Park SH (2013) Arterial ageing. Korean Circ J 43:73–79. doi:10.4070/kcj.2013.43.2.73
Liu B et al (2005) Genomic instability in laminopathy-based premature aging. Nat Med 11:780–785. doi:10.1038/nm1266
Liu Y, Rusinol A, Sinensky M, Wang Y, Zou Y (2006) DNA damage responses in progeroid syndromes arise from defective maturation of prelamin A. J Cell Sci 119:4644–4649. doi:10.1242/jcs.03263
Liu Y, Wang Y, Rusinol AE, Sinensky MS, Liu J, Shell SM, Zou Y (2008) Involvement of xeroderma pigmentosum group A (XPA) in progeria arising from defective maturation of prelamin A. FASEB J 22:603–611. doi:10.1096/fj.07-8598com
Liu GH et al (2011) Recapitulation of premature ageing with iPSCs from Hutchinson–Gilford progeria syndrome. Nature 472:221–225. doi:10.1038/nature09879
Liu B, Zhou S, Liu X, Zhou K, Zhang F, Zhou Z (2013) Accumulation of prelamin A compromises NF-kappaB-regulated B-lymphopoiesis in a progeria mouse model. Longev Healthspan 2:1. doi:10.1186/2046-2395-2-1
Lo CY et al (2014) An upregulation in the expression of vanilloid transient potential channels 2 enhances hypotonicity-induced cytosolic Ca(2)(+) rise in human induced pluripotent stem cell model of Hutchinson-Gillford Progeria. PLoS ONE 9:e87273. doi:10.1371/journal.pone.0087273
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153:1194–1217. doi:10.1016/j.cell.2013.05.039
Matsumoto Y et al (2009) Reduced number and function of endothelial progenitor cells in patients with aortic valve stenosis: a novel concept for valvular endothelial cell repair. Eur Heart J 30:346–355. doi:10.1093/eurheartj/ehn501
Matthews C et al (2006) Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ Res 99:156–164. doi:10.1161/01.RES.0000233315.38086.bc
McClintock D, Ratner D, Lokuge M, Owens DM, Gordon LB, Collins FS, Djabali K (2007) The mutant form of lamin A that causes Hutchinson-Gilford progeria is a biomarker of cellular aging in human skin. PLoS ONE 2:e1269. doi:10.1371/journal.pone.0001269
Merideth MA et al (2008) Phenotype and course of Hutchinson–Gilford progeria syndrome. N Engl J Med 358:592–604. doi:10.1056/NEJMoa0706898
Minamino T, Komuro I (2008) Vascular aging: insights from studies on cellular senescence, stem cell aging, and progeroid syndromes. Nat Clin Pract Cardiovasc Med 5:637–648. doi:10.1038/ncpcardio1324
Miyoshi N, Oubrahim H, Chock PB, Stadtman ER (2006) Age-dependent cell death and the role of ATP in hydrogen peroxide-induced apoptosis and necrosis. Proc Natl Acad Sci USA 103:1727–1731. doi:10.1073/pnas.0510346103
Moiseeva O, Lessard F, Acevedo-Aquino M, Vernier M, Tsantrizos YS, Ferbeyre G (2015) Mutant lamin A links prophase to a p53 independent senescence program. Cell Cycle 14:2408–2421. doi:10.1080/15384101.2015.1053671
Mun GI, Lee SJ, An SM, Kim IK, Boo YC (2009) Differential gene expression in young and senescent endothelial cells under static and laminar shear stress conditions. Free Radic Biol Med 47:291–299. doi:10.1016/j.freeradbiomed.2009.04.032
Nakano-Kurimoto R et al (2009) Replicative senescence of vascular smooth muscle cells enhances the calcification through initiating the osteoblastic transition. Am J Physiol Heart Circ Physiol 297:H1673–1684. doi:10.1152/ajpheart.00455.2009
Nishio K, Inoue A (2005) Senescence-associated alterations of cytoskeleton: extraordinary production of vimentin that anchors cytoplasmic p53 in senescent human fibroblasts. Histochem Cell Biol 123:263–273. doi:10.1007/s00418-005-0766-5
Okuda K, Khan MY, Skurnick J, Kimura M, Aviv H, Aviv A (2000) Telomere attrition of the human abdominal aorta: relationships with age and atherosclerosis. Atherosclerosis 152:391–398
Olive M et al (2010) Cardiovascular pathology in Hutchinson–Gilford progeria: correlation with the vascular pathology of aging. Arterioscler Thromb Vasc Biol 30:2301–2309. doi:10.1161/ATVBAHA.110.209460
Osorio FG et al (2012) Nuclear lamina defects cause ATM-dependent NF-kappaB activation and link accelerated aging to a systemic inflammatory response. Genes Dev 26:2311–2324. doi:10.1101/gad.197954.112
Pacheco LM, Gomez LA, Dias J, Ziebarth NM, Howard GA, Schiller PC (2014) Progerin expression disrupts critical adult stem cell functions involved in tissue repair. Aging (Albany NY) 6:1049–1063
Pekovic V, Hutchison CJ (2008) Adult stem cell maintenance and tissue regeneration in the ageing context: the role for A-type lamins as intrinsic modulators of ageing in adult stem cells and their niches. J Anat 213:5–25. doi:10.1111/j.1469-7580.2008.00928.x
Pendas AM et al (2002) Defective prelamin A processing and muscular and adipocyte alterations in Zmpste24 metalloproteinase-deficient mice. Nat Genet 31:94–99. doi:10.1038/ng871
Philip JT, Dahl KN (2008) Nuclear mechanotransduction: response of the lamina to extracellular stress with implications in aging. J Biomech 41:3164–3170. doi:10.1016/j.jbiomech.2008.08.024
Qin Z, Kalinowski A, Dahl KN, Buehler MJ (2011) Structure and stability of the lamin A tail domain and HGPS mutant. J Struct Biol 175:425–433. doi:10.1016/j.jsb.2011.05.015
Ragnauth CD et al (2010) Prelamin A acts to accelerate smooth muscle cell senescence and is a novel biomarker of human vascular aging. Circulation 121:2200–2210. doi:10.1161/CIRCULATIONAHA.109.902056
Raz V et al (2008) The nuclear lamina promotes telomere aggregation and centromere peripheral localization during senescence of human mesenchymal stem cells. J Cell Sci 121:4018–4028. doi:10.1242/jcs.034876
Rennenberg RJ, Kessels AG, Schurgers LJ, van Engelshoven JM, de Leeuw PW, Kroon AA (2009) Vascular calcifications as a marker of increased cardiovascular risk: a meta-analysis. Vasc Health Risk Manag 5:185–197
Ribeiro AJ, Khanna P, Sukumar A, Dong C, Dahl KN (2014) Nuclear stiffening inhibits migration of invasive melanoma cells. Cell Mol Bioeng 7:544–551. doi:10.1007/s12195-014-0358-3
Rodriguez S, Coppede F, Sagelius H, Eriksson M (2009) Increased expression of the Hutchinson–Gilford progeria syndrome truncated lamin A transcript during cell aging. Eur J Hum Genet 17:928–937. doi:10.1038/ejhg.2008.270
Rosengardten Y, McKenna T, Grochova D, Eriksson M (2011) Stem cell depletion in Hutchinson–Gilford progeria syndrome. Aging Cell 10:1011–1020. doi:10.1111/j.1474-9726.2011.00743.x
Rossi DJ, Jamieson CH, Weissman IL (2008) Stems cells and the pathways to aging and cancer. Cell 132:681–696. doi:10.1016/j.cell.2008.01.036
Rouleau L, Rossi J, Leask RL (2010) The response of human aortic endothelial cells in a stenotic hemodynamic environment: effect of duration, magnitude, and spatial gradients in wall shear stress. J Biomech Eng 132:071015
Rusinol AE, Sinensky MS (2006) Farnesylated lamins, progeroid syndromes and farnesyl transferase inhibitors. J Cell Sci 119:3265–3272. doi:10.1242/jcs.03156
Satoh M, Ishikawa Y, Takahashi Y, Itoh T, Minami Y, Nakamura M (2008) Association between oxidative DNA damage and telomere shortening in circulating endothelial progenitor cells obtained from metabolic syndrome patients with coronary artery disease. Atherosclerosis 198:347–353. doi:10.1016/j.atherosclerosis.2007.09.040
Sawabe M (2010) Vascular aging: from molecular mechanism to clinical significance. Geriatr Gerontol Int 10(Suppl 1):S213–220. doi:10.1111/j.1447-0594.2010.00603.x
Scaffidi P, Misteli T (2006) Lamin A-dependent nuclear defects in human aging. Science 312:1059–1063. doi:10.1126/science.1127168
Scaffidi P, Misteli T (2008) Lamin A-dependent misregulation of adult stem cells associated with accelerated ageing. Nat Cell Biol 10:452–459. doi:10.1038/ncb1708
Sedelnikova OA, Horikawa I, Zimonjic DB, Popescu NC, Bonner WM, Barrett JC (2004) Senescing human cells and ageing mice accumulate DNA lesions with unrepairable double-strand breaks. Nat Cell Biol 6:168–170. doi:10.1038/ncb1095
Sedelnikova OA, Horikawa I, Redon C, Nakamura A, Zimonjic DB, Popescu NC, Bonner WM (2008) Delayed kinetics of DNA double-strand break processing in normal and pathological aging. Aging Cell 7:89–100. doi:10.1111/j.1474-9726.2007.00354.x
Serio RN (2011) Unraveling the mysteries of aging through a Hutchinson-Gilford progeria syndrome model. Rejuvenation Res 14:133–141. doi:10.1089/rej.2010.1088
Shanahan CM (2013) Mechanisms of vascular calcification in CKD-evidence for premature ageing? Nat Rev Nephrol 9:661–670. doi:10.1038/nrneph.2013.176
Sharpless NE, DePinho RA (2007) How stem cells age and why this makes us grow old. Nat Rev Mol Cell Biol 8:703–713. doi:10.1038/nrm2241
Slack C, Alic N, Foley A, Cabecinha M, Hoddinott MP, Partridge L (2015) The Ras-Erk-ETS-Signaling pathway is a drug target for longevity. Cell 162:72–83. doi:10.1016/j.cell.2015.06.023
Snow CJ, Dar A, Dutta A, Kehlenbach RH, Paschal BM (2013) Defective nuclear import of Tpr in Progeria reflects the Ran sensitivity of large cargo transport. J Cell Biol 201:541–557. doi:10.1083/jcb.201212117
Song M, San H, Anderson SA, Cannon RO III, Orlic D (2014) Shear stress-induced mechanotransduction protein deregulation and vasculopathy in a mouse model of progeria. Stem Cell Res Ther 5:41. doi:10.1186/scrt429
Stehbens WE, Wakefield SJ, Gilbert-Barness E, Olson RE, Ackerman J (1999) Histological and ultrastructural features of atherosclerosis in progeria. Cardiovasc Pathol 8:29–39
Stehbens WE, Delahunt B, Shozawa T, Gilbert-Barness E (2001) Smooth muscle cell depletion and collagen types in progeric arteries. Cardiovasc Pathol 10:133–136
Stochaj U, Kodiha M, Shum-Tim D, Colmegna I (2013) Implications of multipotent mesenchymal stromal cell aging. Regen Med 8:211–222. doi:10.2217/rme.13.10
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676. doi:10.1016/j.cell.2006.07.024
Varela I et al (2005) Accelerated ageing in mice deficient in Zmpste24 protease is linked to p53 signalling activation. Nature 437:564–568. doi:10.1038/nature04019
Varga R et al (2006) Progressive vascular smooth muscle cell defects in a mouse model of Hutchinson–Gilford progeria syndrome. Proc Natl Acad Sci USA 103:3250–3255. doi:10.1073/pnas.0600012103
Vasa M et al (2001) Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circ Res 89:E1–7
Vasile E, Tomita Y, Brown LF, Kocher O, Dvorak HF (2001) Differential expression of thymosin beta-10 by early passage and senescent vascular endothelium is modulated by VPF/VEGF: evidence for senescent endothelial cells in vivo at sites of atherosclerosis. FASEB J 15:458–466. doi:10.1096/fj.00-0051com
Verstraeten VL, Ji JY, Cummings KS, Lee RT, Lammerding J (2008) Increased mechanosensitivity and nuclear stiffness in Hutchinson–Gilford progeria cells: effects of farnesyltransferase inhibitors. Aging Cell 7:383–393. doi:10.1111/j.1474-9726.2008.00382.x
Villa-Bellosta R, Rivera-Torres J, Osorio FG, Acin-Perez R, Enriquez JA, Lopez-Otin C, Andres V (2013) Defective extracellular pyrophosphate metabolism promotes vascular calcification in a mouse model of Hutchinson–Gilford progeria syndrome that is ameliorated on pyrophosphate treatment. Circulation 127:2442–2451. doi:10.1161/CIRCULATIONAHA.112.000571
Viteri G, Chung YW, Stadtman ER (2010) Effect of progerin on the accumulation of oxidized proteins in fibroblasts from Hutchinson Gilford progeria patients. Mech Ageing Dev 131:2–8. doi:10.1016/j.mad.2009.11.006
Voghel G et al (2007) Cellular senescence in endothelial cells from atherosclerotic patients is accelerated by oxidative stress associated with cardiovascular risk factors. Mech Ageing Dev 128:662–671. doi:10.1016/j.mad.2007.09.006
Wang JC, Bennett M (2012) Aging and atherosclerosis: mechanisms, functional consequences, and potential therapeutics for cellular senescence. Circ Res 111:245–259. doi:10.1161/CIRCRESAHA.111.261388
Wang L et al (2012) A proteomic study of Hutchinson–Gilford progeria syndrome: application of 2D-chromotography in a premature aging disease. Biochem Biophys Res Commun 417:1119–1126. doi:10.1016/j.bbrc.2011.12.056
Wang C, Baker BM, Chen CS, Schwartz MA (2013) Endothelial cell sensing of flow direction. Arterioscler Thromb Vasc Biol 33:2130–2136. doi:10.1161/ATVBAHA.113.301826
Wang M, Jiang L, Monticone RE, Lakatta EG (2014) Proinflammation: the key to arterial aging. Trends Endocrinol Metab 25:72–79. doi:10.1016/j.tem.2013.10.002
Wenzel V et al (2012) Naive adult stem cells from patients with Hutchinson-Gilford progeria syndrome express low levels of progerin in vivo. Biol Open 1:516–526. doi:10.1242/bio.20121149
Wu H, Roks AJ (2014) Genomic instability and vascular aging: a focus on nucleotide excision repair. Trends Cardiovasc Med 24:61–68. doi:10.1016/j.tcm.2013.06.005
Zhang J et al (2011) A human iPSC model of Hutchinson Gilford Progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell 8:31–45. doi:10.1016/j.stem.2010.12.002
Zhang H, Kieckhaefer JE, Cao K (2013) Mouse models of laminopathies. Aging Cell 12:2–10. doi:10.1111/acel.12021
Zhang H, Xiong ZM, Cao K (2014) Mechanisms controlling the smooth muscle cell death in progeria via down-regulation of poly(ADP-ribose) polymerase 1. Proc Natl Acad Sci USA 111:E2261–2270. doi:10.1073/pnas.1320843111
Zuela N, Bar DZ, Gruenbaum Y (2012) Lamins in development, tissue maintenance and stress. EMBO Rep 13:1070–1078. doi:10.1038/embor.2012.167
Zuo B et al (2012) Influences of lamin A levels on induction of pluripotent stem cells. Biol Open 1:1118–1127. doi:10.1242/bio.20121586
Acknowledgments
We thank Dr. Gerardo Ferbeyre (Université de Montréal) for reviewing this manuscript, and for his insightful comments and discussion. We also thank Professor Gaétan Laroche (Université Laval and Centre de recherche du CHU de Québec) and William L. Stanford (Ottawa Hospital Research Institute and University of Ottawa) for useful discussions. We thank the Canadian Stem Cell Network (JB, AG and CH), Pfizer Ltd and Pfizer Canada Inc. (See the Potential postdoctoral fellowship awarded to CH), the Canadian Institutes of Health Research (MOP-142285), Saint-Gobain Ceramics & Plastics Inc., the ThéCell Network and the Centre québécois sur les matériaux fonctionnels (CQMF) strategic cluster of the Fonds de recherche du Québec—Nature et technologies for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brassard, J.A., Fekete, N., Garnier, A. et al. Hutchinson–Gilford progeria syndrome as a model for vascular aging. Biogerontology 17, 129–145 (2016). https://doi.org/10.1007/s10522-015-9602-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-015-9602-z