Abstract
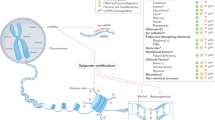
Alzheimer’s disease (AD), the most common form of dementia among the elderly, manifests mostly late in adult life. However, it is presently unclear when the disease process starts and how long the pathobiochemical processes take to develop. Our goal is to address the timing and nature of triggers that lead to AD. To explain the etiology of AD, we have recently proposed a “Latent Early-life Associated Regulation” (LEARn) model, which postulates a latent expression of specific genes triggered at the developmental stage. This model integrates both the neuropathological features (e.g., amyloid-loaded plaques and tau-laden tangles) and environmental factors (e.g., diet, metal exposure, and hormones) associated with the disease. Environmental agents perturb gene regulation in a long-term fashion, beginning at early developmental stages, but these perturbations do not have pathological results until significantly later in life. The LEARn model operates through the regulatory region (promoter) of the gene and by affecting the methylation status within the promoter of specific genes.

Similar content being viewed by others
References
Barker DJ, Eriksson JG, Forsen T et al (2002) Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol 31:1235–1239. doi:10.1093/ije/31.6.1235
Basha MR, Wei W, Bakheet SA et al (2005) The fetal basis of amyloidogenesis: exposure to lead and latent overexpression of amyloid precursor protein and beta-amyloid in the aging brain. J Neurosci 25:823–829. doi:10.1523/JNEUROSCI.4335-04.2005
Bellingham SA, Lahiri DK, Maloney B et al (2004) Copper depletion down-regulates expression of the Alzheimer’s disease amyloid-beta precursor protein gene. J Biol Chem 279:20378–20386. doi:10.1074/jbc.M400805200
Bolin CM, Basha R, Cox D et al (2006) Exposure to lead and the developmental origin of oxidative DNA damage in the aging brain. FASEB J 20:788–790
Buehlmeyer K, Doering F, Daniel H et al (2008) Alteration of gene expression in rat colon mucosa after exercise. Ann Anat 190:71–80. doi:10.1016/j.aanat.2007.04.002
Campos AC, Molognoni F, Melo FH et al (2007) Oxidative stress modulates DNA methylation during melanocyte anchorage blockade associated with malignant transformation. Neoplasia 9:1111–1121. doi:10.1593/neo.07712
Chan A, Shea TB (2006) Supplementation with apple juice attenuates presenilin-1 overexpression during dietary and genetically-induced oxidative stress. J Alzheimers Dis 10:353–358
De la Burde B, Choate MS (1972) Does asymptomatic lead exposure in children have latent sequelae? J Pediatrics 81:1088–1091
Dobosy JR, Selker EU (2001) Emerging connections between DNA methylation and histone acetylation. Cell Mol Life Sci 58:721–727. doi:10.1007/PL00000895
Fowler BA, Whittaker MH, Lipsky M et al (2004) Oxidative stress induced by lead, cadmium and arsenic mixtures: 30-day, 90-day, and 180-day drinking water studies in rats: an overview. Biometals 17:567–568. doi:10.1023/B:BIOM.0000045740.52182.9d
Hebert LE, Scherr PA, Bienias JL et al (2003) Alzheimer disease in the US population: prevalence estimates using the 2000 census. Arch Neurol 60:1119–1122. doi:10.1001/archneur.60.8.1119
Kivipelto M, Solomon A (2008) Alzheimer’s disease—the ways of prevention. J Nutr Health Aging 12:89S–94S
Knudson AG Jr (1971) Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA 68:820–823. doi:10.1073/pnas.68.4.820
Lahiri DK, Farlow MR, Sambamurti K et al (2003) A critical analysis of new molecular targets and strategies for drug developments in Alzheimer’s disease. Curr Drug Targets 4:97–112. doi:10.2174/1389450033346957
Lahiri DK, Chen D, Ge YW et al (2004) Dietary supplementation with melatonin reduces levels of amyloid beta-peptides in the murine cerebral cortex. J Pineal Res 36:224–231. doi:10.1111/j.1600-079X.2004.00121.x
Lahiri DK, Wavrant De-Vrieze F, Ge Y-W et al (2005) Characterization of two APP gene promoter polymorphisms that appear to influence risk of late-onset Alzheimer’s disease. Neurobiol Aging 26:1329–1341. doi:10.1016/j.neurobiolaging.2004.11.005
Lahiri DK, Maloney B, Basha MR et al (2007) How and when environmental agents and dietary factors affect the course of Alzheimer’s disease: the “LEARn” model (Latent Early Associated Regulation) may explain the triggering of AD. Curr Alzheimer Res 4:219–228. doi:10.2174/156720507780362164
Lu T, Pan Y, Kao SY et al (2004) Gene regulation and DNA damage in the ageing human brain. Nature 429:883–891. doi:10.1038/nature02661
Maloney B, Ge Y-W, Alley GM et al (2007) Important differences between human and mouse APOE gene promoters with implications for Alzheimer’s disease. J Neurochem 103:1237–1257. doi:10.1111/j.1471-4159.2007.04831.x
Sambamurti K, Suram A, Venugopal C et al (2006) A partial failure of membrane protein turnover may cause Alzheimer’s disease: a new hypothesis. Curr Alzheimer Res 3:81–90. doi:10.2174/156720506775697142
Takiguchi M, Achanzar WE, Qu W et al (2003) Effects of cadmium on DNA-(cytosine-5) methyltransferase activity and DNA methylation status during cadmium-induced cellular transformation. Exp Cell Res 286:355–365. doi:10.1016/S0014-4827(03)00062-4
Valinluck V, Tsai HH, Rogstad DK et al (2004) Oxidative damage to methyl-CpG sequences inhibits the binding of the methyl-CpG binding domain (MBD) of methyl-CpG binding protein 2 (MeCP2). Nucleic Acids Res 32:4100–4108. doi:10.1093/nar/gkh739
Wu J, Basha MR, Brock B et al (2008) Alzheimer’s disease (AD)-like pathology in aged monkeys after infantile exposure to environmental metal lead (Pb): evidence for a developmental origin and environmental link for AD. J Neurosci 28:3–9. doi:10.1523/JNEUROSCI.4405-07.2008
Acknowledgments
This work was supported in part by grants from Alzheimer’s Associations (Zenith award) and the National Institutes of Health (AG18379 and AG18884) to DKL.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lahiri, D.K., Zawia, N.H., Greig, N.H. et al. Early-life events may trigger biochemical pathways for Alzheimer’s disease: the “LEARn” model. Biogerontology 9, 375–379 (2008). https://doi.org/10.1007/s10522-008-9162-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-008-9162-6