Abstract
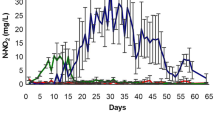
This research aimed to elucidate the effect of Chlorella sp. addition on biofloc quality and growth of Macrobrachium rosenbergii juveniles in biofloc systems. Four treatments of freshwater prawn nursery culture systems were tested, i.e., control system with clear water system at a regular water exchange at about 50% every 2 weeks; control system with Chlorella sp. addition with regular water exchange and weekly addition of Chlorella sp.; biofloc system without Chlorella sp. addition; and biofloc system with Chlorella sp. addition. Freshwater prawn juvenile at an average initial body weight of 0.25 g was randomly distributed into 16 units of aquarium with a working volume of 100 L at a density of 20 prawn/aquarium (50 prawn/m2). Molasses was added every day into biofloc treatments at an estimated C/N ratio of 6.25. Chlorella sp. was added into the microalgae treatments at the initiation of the experiment and every 2 weeks at 10% of the total volume to reach an estimated final density of about 105/mL. Microbial aggregation was inhibited by vanillin (a quorum sensing inhibitor) addition regardless the presence of Chlorella sp. The microalgae addition resulted in biofloc with higher density, larger particle size, and higher protein content. Different amino acid and fatty acid profiles of biofloc were observed between the biofloc treatments. Essential amino acid index of biofloc with the addition of Chlorella sp. was about 39% higher compared to that of biofloc without Chlorella sp. addition. The growth of freshwater prawn in biofloc systems was generally higher than those of the control with and without Chlorella sp. addition. In conclusion, the addition of Chlorella sp. could improve biofloc quality and the growth performance of freshwater prawn juveniles.


Similar content being viewed by others
References
APHA (1998) Standard methods for the examination of water and wastewater. American Public Health Association. Inc., Washington DC
AOAC Official Method (2000) AOAC official methods of analysis, 17thEd edn. AOAC, Gathersburg
Arai S (1981) A purified diet for coho salmon Oncorhynchus kisutch fry. Bull Jpn Soc Sci Fish 47:547–550. https://doi.org/10.2331/suisan.47.547
Avnimelech Y (1999) Carbon/nitrogen ratio as a control element in aquaculture systems. Aquaculture 176:227–235. https://doi.org/10.1016/S0044-8486(99)00085-X
Ballester ELC, Marzarotto SA, Silva de Castro C, Frozza A, Pastore I, Abreu PC (2017) Productive performance of juvenile freshwater prawns Macrobrachium rosenbergii in biofloc system. Aquac Res 48:4748–4755. https://doi.org/10.1111/are.13296
Bhavan PS, Radhakrishnan S, Seenivasan C, Shanthi R, Poongodi R, Kannan S (2010) Proximate composition and profiles of amino acids and fatty acids in the muscle of adult males and females of commercially viable prawn species Macrobrachium rosenbergii collected from natural culture environments. Int J Biol 2:107–119. https://doi.org/10.5539/ijb.v2n2p107
Chen YC (2001) Immobilized microalga Scenedesmus quadricauda (Chlorophyta, Chlorococcales) for long-term storage and for application for water quality control in fish culture. Aquaculture 195:71–80. https://doi.org/10.1016/S0044-8486(00)00540-8
Cirri E, Pohnert G (2019) Algae−bacteria interactions that balance the planktonic microbiome. New Phytol 223:100–106
Crab R, Chielens B, Wille M, Bossier P, Verstraete W (2010) The effect of different carbon sources on the nutritional value of bioflocs, a feed for Macrobrachium rosenbergii postlarvae. Aquac Res 41:559–567. https://doi.org/10.1111/j.1365-2109.2009.02353.x
De Schryver P, Crab R, Defoirdt T et al (2008) The basics of bio-flocs technology: the added value for aquaculture. Aquaculture 277:125–137. https://doi.org/10.1016/j.aquaculture.2008.02.019
Ebeling JM, Timmons MB, Bisogni JJ (2006) Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia-nitrogen in aquaculture systems. Aquaculture 257:346–358. https://doi.org/10.1016/j.aquaculture.2006.03.019
Ekasari J, Angela D, Waluyo SH, Bachtiar T, Surawidjaja EH, Bossier P, de Schryver P (2014) The size of biofloc determines the nutritional composition and the nitrogen recovery by aquaculture animals. Aquaculture 426-427:426–427. https://doi.org/10.1016/j.aquaculture.2014.01.023
Ekasari J, Crab R, Verstraete W (2010) Primary nutritional content of bio-flocs cultured with different organic carbon sources and salinity. HAYATI J Biosci 17:125–130. https://doi.org/10.4308/hjb.17.3.125
Fatimah N, Pande GSJ, Natrah FMI, Meritha WW, Widanarni, Sucipto A, Ekasari J (2019) The role of microbial quorum sensing on the characteristics and functionality of bioflocs in aquaculture systems. Aquaculture 504:420–426. https://doi.org/10.1016/j.aquaculture.2019.02.022
Fimbres-Acedo YE, Magallón-Servín P, Garza-Torres R, Emerenciano MGC, Servín-Villegas R, Endo M, Fitzsimmons KM, Magallón-Barajas FJ (2020) Oreochromis niloticus aquaculture with biofloc technology, photoautotrophic conditions and Chlorella microalgae. Aquac Res 51:3323–3346. https://doi.org/10.1111/are.14668
Fuentes JL, Garbayo I, Cuaresma M, et al (2016) Impact of microalgae-bacteria interactions on the production of algal biomass and associated compounds. Mar. Drugs 14
Hargreaves JA (2006) Photosynthetic suspended-growth systems in aquaculture. Aquac Eng 34:344–363. https://doi.org/10.1016/j.aquaeng.2005.08.009
Jiménez-Ordaz FJ, Cadena-Roa MA, Pacheco-Vega JM, Rojas-Contreras M, Tovar-Ramírez D, Arce-Amezquita PM (2021) Microalgae and probiotic bacteria as biofloc inducers in a hyper-intensive Pacific white shrimp (Penaeus vannamei) culture. Lat Am J Aquat Res 49:155–168
Ju ZY, Forster I, Conquest L, Dominy W, Kuo WC, David Horgen F (2008) Determination of microbial community structures of shrimp floc cultures by biomarkers and analysis of floc amino acid profiles. Aquac Res 39:118–133. https://doi.org/10.1111/j.1365-2109.2007.01856.x
Jung JY, Damusaru JH, Park Y, Kim K, Seong M, Je HW, Kim S, Bai SC (2017) Autotrophic biofloc technology system (ABFT) using Chlorella vulgaris and Scenedesmus obliquus positively affects performance of Nile tilapia (Oreochromis niloticus). Algal Res 27:259–264. https://doi.org/10.1016/j.algal.2017.09.021
Khanjani MH, Sharifinia M (2020) Biofloc technology as a promising tool to improve aquaculture production. Rev Aquac 1–15. https://doi.org/10.1111/raq.12412
Lavens P, Sorgeloos P (1996) Manual on the production and use of live food for aquaculture (No. 361). Food and Agriculture Organization (FAO)
Lee J, Cho DH, Ramanan R, Kim BH, Oh HM, Kim HS (2013) Microalgae-associated bacteria play a key role in the flocculation of Chlorella vulgaris. Bioresour Technol 131:195–201. https://doi.org/10.1016/j.biortech.2012.11.130
Llario F, Rodilla M, Escrivá J, Falco S, Sebastiá-Frasquet MT (2019) Phytoplankton evolution during the creation of a biofloc system for shrimp culture. Int J Environ Sci Technol 16:211–222. https://doi.org/10.1007/s13762-018-1655-5
Lv J, Wang Y, Zhong C, Li Y, Hao W, Zhu J (2014) The effect of quorum sensing and extracellular proteins on the microbial attachment of aerobic granular activated sludge. Bioresour Technol 152:53–58. https://doi.org/10.1016/j.biortech.2013.10.097
Martins TG, Odebrecht C, Jensen LV, D ' Oca MGM, Wasielesky W Jr (2016) The contribution of diatoms to bioflocs lipid content and the performance of juvenile Litopenaeus vannamei (Boone, 1931) in a BFT culture system. Aquac Res 47:1315–1326. https://doi.org/10.1111/are.12592
Miao S, Sun L, Bu H, Zhu J, Chen G (2017a) Effect of molasses addition at C:N ratio of 20:1 on the water quality and growth performance of giant freshwater prawn (Macrobrachium rosenbergii). Aquac Int 25:1409–1425. https://doi.org/10.1007/s10499-017-0124-3
Miao S, Zhu J, Zhao C, Sun L, Zhang X, Chen G (2017b) Effects of C/N ratio control combined with probiotics on the immune response, disease resistance, intestinal microbiota and morphology of giant freshwater prawn (Macrobrachium rosenbergii). Aquaculture 476:125–133. https://doi.org/10.1016/j.aquaculture.2017.04.027
Natrah FMI, Bossier P, Sorgeloos P, Yusoff FM, Defoirdt T (2014) Significance of microalgal-bacterial interactions for aquaculture. Rev Aquac 6:48–61. https://doi.org/10.1111/raq.12024
Nguyen TDP, Le TVA, Show PL et al (2019) Bioflocculation formation of microalgae-bacteria in enhancing microalgae harvesting and nutrient removal from wastewater effluent. Bioresour Technol 272:34–39. https://doi.org/10.1016/j.biortech.2018.09.146
Pande GSJ, Natrah FMI, Flandez AVB, Kumar U, Niu Y, Bossier P, Defoirdt T (2015) Isolation of AHL-degrading bacteria from micro-algal cultures and their impact on algal growth and on virulence of Vibrio campbellii to prawn larvae. Appl Microbiol Biotechnol 99:10805–10813. https://doi.org/10.1007/s00253-015-6918-1
Panigrahi A, Saranya C, Ambiganandam K, Sundaram M, Sivakumar MR, Kumaraguru vasagam KP (2020) Evaluation of biofloc generation protocols to adopt high density nursery rearing of Penaeus vannamei for better growth performances, protective responses and immuno modulation in biofloc based technology. Aquaculture 522:735095. https://doi.org/10.1016/j.aquaculture.2020.735095
Peñaflorida VD (1989) An evaluation of indigenous protein sources as potential component in the diet formulation for tiger prawn, Penaeus monodon, using essential amino acid index (EAAI). Aquaculture 83:319–330. https://doi.org/10.1016/0044-8486(89)90043-4
Pérez-Fuentes JA, Pérez-Rostro CI, Hernández-Vergara MP (2013) Pond-reared Malaysian prawn Macrobrachium rosenbergii with the biofloc system. Aquaculture 400–401:105–110. https://doi.org/10.1016/j.aquaculture.2013.02.028
Rohmana D, Surawidjaja EH, Sukenda S, Ekasari J (2014) Water quality and production performance of catfish–prawn co-culture with organic carbon source addition. Aquac Int 23:267–276. https://doi.org/10.1007/s10499-014-9814-2
Salehizadeh H, Vossoughi M, Alemzadeh I (2000) Some investigations on bioflocculant producing bacteria. Biochem Eng J 5:39–44. https://doi.org/10.1016/S1369-703X(99)00066-2
Schrader KK, Green BW, Perschbacher PW (2011) Aquacultural Engineering Development of phytoplankton communities and common off-flavors in a biofloc technology system used for the culture of channel catfish ( Ictalurus punctatus ). Aquac Eng 45:118–126. https://doi.org/10.1016/j.aquaeng.2011.08.004
Subashchandrabose SR, Ramakrishnan B, Megharaj M, Venkateswarlu K, Naidu R (2011) Consortia of cyanobacteria/microalgae and bacteria: biotechnological potential. Biotechnol Adv 29:896–907. https://doi.org/10.1016/j.biotechadv.2011.07.009
Sun S, Weber-Shirk M, Lion LW (2016) Characterization of flocs and floc size distributions using image analysis. Environ Eng Sci 33:25–34. https://doi.org/10.1089/ees.2015.0311
Wang Y, Yang Y, Ma F, Xuan L, Xu Y, Huo H, Zhou D, Dong S (2015) Optimization of Chlorella vulgaris and bioflocculant-producing bacteria co-culture: enhancing microalgae harvesting and lipid content. Lett Appl Microbiol 60:497–503. https://doi.org/10.1111/lam.12403
Yang W, Zheng Z, Lu K, Zheng C, du Y, Wang J, Zhu J (2020) Manipulating the phytoplankton community has the potential to create a stable bacterioplankton community in a shrimp rearing environment. Aquaculture 520:734789. https://doi.org/10.1016/j.aquaculture.2019.734789
Acknowledgements
We thank the Directorate General of Higher Education of The Ministry of Research, Technology, and Higher Education (Grant No. 1408/IT3.11/PN/2017) for the research funding. We also thank Miss Restu Malianty, Miss Retno Meliasari, and Miss Lina Mulyani for the technical and administrative assistance during the research implementation.
Funding
The research was funded by the Directorate General of Higher Education of The Ministry of Research, Technology, and Higher Education (Grant No. 1408/IT3.11/PN/2017).
Author information
Authors and Affiliations
Contributions
Julie Ekasari: Conceptualization, methodology, data curation, writing - original draft, writing - review and editing. Utomo Adi Nugroho: Investigation, resources, data curation, writing - original draft. Nurul Fatimah: Investigation, writing - original draft. Deasy Angela: Investigation, writing - original draft. Yuni Puji Hastuti: Conceptualization, writing - original draft. Pande Gde Sasmita Julyantoro: Conceptualization, writing - original draft. F.M.I. Natrah: Conceptualization, writing - original draft.
Corresponding author
Ethics declarations
Ethic approval
The experimental procedures have complied the ethical guidelines from the Animal Care and Use Committee of IPB University, Indonesia.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Simon Goddek
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ekasari, J., Nugroho, U.A., Fatimah, N. et al. Improvement of biofloc quality and growth of Macrobrachium rosenbergii in biofloc systems by Chlorella addition. Aquacult Int 29, 2305–2317 (2021). https://doi.org/10.1007/s10499-021-00750-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00750-1