Abstract
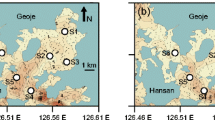
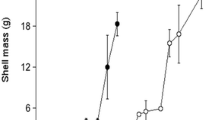
In Eastern Canada, an increasing number of pests, predators and pathogens in downriver areas (close to the ocean) have led the aquaculture industry to consider growing oysters in upriver areas (close to the river source). In this study, oyster growth performance was compared between downriver and upriver environments by means of stock transfer experiments within the Richibucto estuary. In May 2009, seed oysters (~26 mm shell height) originating from two downriver sites (salinity ~20–30 ‰) were transferred upriver (salinity ~5–20 ‰). Follow-up measurements in October 2009 revealed that the seed transferred upriver grew and survived as well as seed that remained downriver, while the mortality rates of adult oysters (shell height ~66 mm) were lower at the upriver site. Meat content was unaffected in adult oysters transferred upriver. However, there were indications that the upriver environment promoted shell growth (mm) in adult oysters. Oysters transferred upriver had a gain in shell height (LSM ± SE) over the oysters that remained downriver (2.7 ± 0.5 vs. 1.8 ± 0.5 mm) and in shell width [Median (95 % CI); 2.8 mm (1.9, 3.6) vs. 1.0 mm (0.3, 1.2)]. Therefore, the holding of adult oysters upriver during the spring–summer period confers productivity advantages on top of protection from diseases and predation. By contrast, productivity losses were recorded when relocating adult oysters originating from the upriver environment. More specifically, final organic meat content were approximately 35 % less in adult oysters transferred downriver compared to those that remained upriver (0.48 ± 0.04 vs. 0.74 ± 0.04 g). Results suggest that transfers along the river impact physiological processes such as gametogenesis and shell formation in adult oysters.



Similar content being viewed by others
References
Albright BW, Abbe GR, McCollough CB, Barker LS, Dungan CF (2007) Growth and mortality of dermo-disease-free juvenile oysters (Crassostrea virginica) at three salinity regimes in an enzootic area of Chesapeake Bay. J Shellfish Res 26:451–463
Andrews JD, Haven D, Quayle DB (1959) Freshwater kill of oysters (Crassostrea virginica) in James River, Virginia. Proc Natl Shellfish Assoc 49:29–49
Bataller EE, Boghen AD, Burt MDB (1999) Comparative growth of the eastern oyster Crassostrea virginica (Gmelin) reared at low and high salinities in New Brunswick, Canada. J Shellfish Res 18:107–114
Bayne BL (2004) Phenotypic flexibility and physiological tradeoffs in the feeding and growth of marine bivalve molluscs. Integr Comp Biol 44:425–432
Berger VJ, Kharazova AD (1997) Mechanisms of salinity adaptations in marine molluscs. Hydrobiologia 355:115–126
Boettcher KJ, Smolowitz R, Lewis EJ, Allam B, Dicherson H, Ford S, Huq A, Reece K, Rivara G, Woodley CM (2006) Juvenile oyster disease (JOD) in Crassostrea virginica: synthesis of knowledge and recommendations. J Shellfish Res 25:683–686
Bushek D, Ford SE, Burt I (2012) Long-term patterns of an estuarine pathogen along a salinity gradient. J Mar Res 70:225–251
Butler PA (1949) Gametogenesis in the oyster under conditions of depressed salinity. Biol Bull 96:263–269
Chanley PE (1958) Survival of some juvenile bivalves in water of low salinity. Proc Natl Shellfish Assoc 48:52–65
Chu FL, La Peyre JF, Burreson CS (1993) Perkinsus marinus infection and potential defense-related activities in eastern oysters, Crassostrea virginica: salinity effects. J Invertebr Pathol 62:226–232
Comeau LA (2014) Spring awakening temperature and survival of sediment-covered eastern oysters Crassostrea virginica. Aquaculture 430:188–194
Comeau LA, Mayrand E, Mallet A (2012) Winter quiescence and spring awakening of the eastern oyster Crassostrea virginica at its northernmost distribution limit. J Mar Biol 159:2269–2279
Dohoo I, Martin W, Stryhn H (2009) Veterinary epidemiologic research. VER Inc., Charlottetown
Doiron S (2008) Manuel de référence de l’ostréiculteur. Ministère de l’agriculture et de l’aquaculture, NB
Doiron S, Maillet MJ (2010) Rapport d’activités 2009: Évaluation de la croissance des huîtres en suspension le long de la côte-est du Nouveau-Brunswick. Ministère de l’agriculture et de l’aquaculture, NB
Drent J, Luttikhuizen PC, Piersma T (2004) Morphological dynamics in the foraging apparatus of a deposit feeding marine bivalve: phenotypic plasticity and heritable effects. Funct Ecol 18:349–356
Eierman LE, Hare MP (2013) Survival of oyster larvae in different salinities depends on source population within an estuary. J Exp Mar Biol Ecol 449:61–68
Eldridge PJ, Eversole AG (1982) Compensatory growth and mortality of the hard clam, Mercenaria mercenaria (Linnaeus, 1758). Veliger 24:276–278
Elston RA, Ford SE (2011) Shellfish diseases and health management. In: Shumway SE (ed) Shellfish aquaculture and the environment. Wiley, Oxford, pp 359–394
Engle JB (1946) Commercial aspects of the upper Chesapeake Bay oyster bars in light of recent oyster mortalities. In: Proceedings of the National Shellfish Association, pp 42–46
Environment Canada (1964–2010) Water surveys of Canada: water level and streamflow statistics. Station no. 01BS001. http://www.wsc.ec.gc.ca. Cited 20 July 2014
Ernande B, Boudry P, Clobert J, Haures J (2004) Plasticity in resource allocation based life history traits in the Pacific oyster, Crassostrea gigas. I. Spatial variation in food abundance. J Evol Biol 17:342–356
Ewart JW, Ford SE (1993) History and impact of MSX and dermo diseases on oyster stocks in the northeast region. Northeastern Regional Aquaculture Center, University of Massachusetts, Massachusetts
Filgueira R, Guyondet T, Comeau LA, Grant J (2014) A fully-spatial ecosystem-DEB model of oyster (Crassostrea virginica) carrying capacity in the Richibucto Estuary, Eastern Canada. J Mar Syst 136:42–54
Fisher WS, Chintala MM, Moline MA (1989) Annual variation of estuarine and oceanic oyster Crassostrea virginica Gmelin hemocyte capacity. J Exp Mar Biol Ecol 127:105–120
Ford SE, Haskin HH (1988) Management strategies for MSX (Haplosporidium nelsoni) disease in eastern oysters. Am Fish Soc Spec Publ 18:249–256
Ford SE, Smolowitz R (2007) Infection dynamics of an oyster parasite in its newly expanded range. Mar Biol 151:119–133
Foreman MG (1977) Manual for tidal heights analysis and previsions. Pac Mar Sci Rep 77–10:101
Hamdoun AM, Cheney DP, Cherr GN (2003) Phenotypic plasticity of HSP70 and HSP70 gene expression in the Pacific oyster (Crassostrea gigas): implications for thermal limits and induction of thermal tolerance. Biol Bull 205:160–169
Honkoop PJC (2003) Physiological costs of reproduction in the Sydney rock oyster Saccostrea glomerata. Oecologia 135:176–183
Hoverman JT, Relyea RA (2007) How flexible is phenotypic plasticity? Developmental windows for trait induction and reversal. Ecology 88:693–705
Hoverman JT, Auld JR, Relyea RA (2005) Putting prey back together again: integrating predator-induced behavior, morphology, and life history. Oecologia 144:481–491
Ingle RM, Dawson CE Jr (1952) Growth of the American oyster, Crassostrea virginica (Gmelin) in Florida waters. Bull Mar Sci 2:393–404
Kooijman SALM (2000) Dynamic energy and mass budgets in biological systems. Cambridge University Press, Cambridge
Kraeuter JN, Ford S, Cummings M (2007) Oyster growth analysis: a comparison of methods. J Shellfish Res 26:479–491
La Peyre MK, Gossman B, La Peyre JF (2009) Defining optimal freshwater flow for oyster production: effects of freshet rate and magnitude of change and duration on eastern oysters and Perkinsus marinus infection. Estuaries Coasts 32:522–534
Landry T, Hardy M, Ouellette M & MacNair NG (2001) Monitoring the life cycle of the northern quahaug, Mercenaria mercenaria, in Prince Edward Island. Canadian Technical Report Fisheries Aquatic Sciences 2355
Loosanoff VL (1953) Behavior of oysters in water of low salinities. Proc Natl Shellfish Assoc 43:135–151
Mallet AL, Carver C, Hardy M (2009) The effect of floating bag management strategies on biofouling, oyster growth and biodeposition levels. Aquaculture 287:315–323
Mallet AL, Carver CE, Doiron S, Thériault MH (2013) Growth performance of eastern oysters Crassostrea viriginica in Atlantic Canada: effect of the culture gear. Aquaculture 1:396–399
Mayrand E, Sonier T, Comeau LA (2013) Hot water immersion lowers survival, shell growth rate and lysosomal membrane stability of oysters Crassostrea virginica (Gmelin). Aquac Res. doi:10.1111/are.12364
McGladdery SE, Zurbrigg RE (2006) Canada’s approach to aquatic animal biosecurity: experience and evolution. In: Scarfe DA, Lee C-S, O’Bryen PJ (eds) Aquaculture biosecurity: prevention, control, and eradication of aquatic animal disease. Blackwell Publishing, Ames
Medcof JC, Needler AWH (1941) The influence of temperature and salinity on the condition of oysters (Ostrea virginica). J Fish Res Board Can 5:253–257
Méthé D, Léger M (1994) Assessment of oyster spat growth and collection in Bouctouche Bay, New Brunswick, Canada 1987 to 1993. New Brunswick Department of Fisheries and Aquaculture, Bouctouche
North EW, King DM, Xu J, Hood RR, Newell RIE, Paynter K, Kellogg ML, Liddel MK, Boesch DF (2010) Linking optimization and ecological models in a decision support tool for oyster restoration and management. Ecol Appl 20:851–866
Paynter KT, Burreson EM (1991) Effects of Perkinsus marinus infection in the eastern oyster, Crassostrea virginica: II. Disease development and impact on growth at different salinities. J Shellfish Res 10:425–431
Pollack JB, Kim H, Morgan EK, Montagna PA (2011) Role of flood disturbance in natural oyster (Crassostrea virginica) population maintenance in an estuary in South Texas, USA. Estuaries Coasts 34:187–197
Pouvreau S, Bourles Y, Lefebvre S, Gangnery A, Alunno-Bruscia M (2006) Application of a dynamic energy budget model to the Pacific oyster, Crassostrea gigas, reared under various environmental conditions. J Sea Res 56:156–167
Ragone LM, Burreson EM (1993) Effect of salinity on infection progression and pathogenicity of Perkinsus marinus in the eastern oyster, Crassostrea virginica (Gmelin). J Shellfish Res 12:1–7
Sanford E, Kelly MW (2011) Local adaptation in marine invertebrates. Annu Rev Mar Sci 3:509–535
Shaw WN (1966) The growth and mortality of seed oysters, Crassostrea virginica, from Broad Creek, Chesapeake Bay, Maryland, in high-and low-salinity waters. Proc Natl Shellfish Assoc 56:59–63
Shumway SE (1996) Natural environment factors. In: Kennedy VS, Newell RIE, Eble AF (eds) The eastern oyster Crassostrea virginica. Maryland Sea Grant College, College Park, pp 467–503
Turcotte-Lanteigne A, Ferguson E (2008) Ecosystem overview of the Richibucto watershed in New Brunswick. Can Manuscr Rep Fish Aquat Sci 2847:182
Vasseur L, Catto NR (2008) Canada Atlantique. In: Lemmen DS, Warren FJ, Lacroix J, Bush E (eds) Vivre avec les changements climatiques au Canada, 2007th edn. Gouvernement du Canada, Ottawa, pp 119–170
Wallace DH (1966) Oysters in the estuarine environment. Am Fish Soc Spec Publ 3:68–73
Weisberg S (2005) Applied linear regression, 3rd edn. Wiley, New Jersey
Wells HW (1961) The fauna of oyster beds, with special reference to the salinity factors. Ecol Monogr 31:239–266
Acknowledgments
This project was funded by the Aquaculture Collaborative Research and Development Program (ACRDP) in partnership with the Elsipogtog First Nation (Project No. MG–08–01–006). The authors would like to thank Rémi Sonier for his expertise in the field and Dr John Martell for his critical review of the manuscript. Special thanks as well to our industry partner, the Elsipogtog First Nation represented by Mr. Blayne Peters for much needed assistance and guidance in the field.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Méthé, D., Comeau, L.A., Stryhn, H. et al. Survival and growth performance of Crassostrea virginica along an estuarine gradient. Aquacult Int 23, 1089–1103 (2015). https://doi.org/10.1007/s10499-014-9866-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-014-9866-3