Abstract
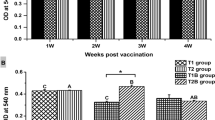
In this study, efficacy of three antigenic preparations from the fish pathogen Aeromonas hydrophila was evaluated as vaccine candidate in rohu Labeo rohita. The rabbit anti-rohu globulin conjugate was prepared in rabbit using sera obtained from healthy adult rohu. Three antigens, namely formalin inactivated A. hydrophila (FAH), FAH mixed with Freund’s incomplete adjuvant in a 1:1 (v/v) ratio (FAH + A), and extra cellular product were prepared. Protection of rohu against A. hydrophila infection was tested at days 10, 20, and 30 post-vaccination. At the end of the vaccination, fish were challenged with A. hydrophila (dose: 1 × 107 cfu ml−1) and relative per cent of survival was recorded up to 60th day of post-challenge. Study of cellular immunological parameters including antigen-specific leucocyte proliferation, antigen-specific nitric oxide production, and superoxide anion production exhibited significantly higher (p < 0.05) values on 10th and 20th day of post-vaccination compared with control. Among all the antigenic groups, FAH + A showed most significant responses compared with the other groups and produced maximum antibody on 30th day following vaccination. Among the humoral immune responses, lysozyme activity showed almost similar trend like cellular parameters. The result of the challenge study showed a higher level of survival in all the vaccinated groups, especially in FAH + A group. Our results suggest that vaccine FAH + A can effectively protect rohu against A. hydrophila infection and could offer an appropriate strategy to prevent this infection in rohu farms.





Similar content being viewed by others
References
Alexander JB, Ingram GA (1992) Noncellular non-specific defence mechanisms of fish. Ann Rev Fish Dis 2:249–279
Amend DF (1981) Potency testing of fish vaccines. International symposium on fish biologics: serodiagnostics and vaccines. Dev Biol Stand 49:447–454
APHA, Awwa, WEF (1998) Standard methods for the examination of water and waste water, 20th edn. American Public Health Association, American Water Works Association, Water Environment Association, Washington, pp 413–426
Azad IS, Shankar KM, Mohan CV, Kalita B (1999) Biofilm vaccine of Aeromonas hydrophila—standardization of dose and duration for oral vaccination of carps. Fish Shellfish Immunol 9:519–528
Azad IS, Shankar KM, Mohan CV, Kalitav B (2000) Uptake and processing of biofilm and free-cell vaccines of Aeromonas hydrophila in Indian major carps and common carp following oral vaccination-antigen localization by a monoclonal antibody. Dis Aquat Organ 43:103–108
Bastardo A, Ravelo C, Castro N et al (2012) Effectiveness of bivalent vaccines against Aeromonas hydrophila and Lactococcus garvieae infections in rainbow trout Oncorhynchus mykiss (Walbaum). Fish Shellfish Immunol 32:756–761
Behera T, Nanda PK, Mohanty C, Mohapatra D, Swain P et al (2010) Parenteral immunization of fish, Labeo rohita with Poly d, l-lactide-co-glycolic acid (PLGA) encapsulated antigen microparticles promotes innate and adaptive immune responses. Fish Shellfish Immunol 28:320–325
Bondad-Reantaso MG, Suba singhe RP, Arthur JR et al (2005) Disease and health management in Asian aquaculture. Vet Parasitol 132:249–272
Chandran MR, Aruna BV, Logambal SM, Michael RD (2002) Immunisation of Indian major carps against Aeromonas hydrophila by intraperitoneal injection. Fish Shellfish Immunol 13:1–9
CIFA (2004) Annual report Central Institute of Freshwater Aquaculture, Bhubaneswar
Das P, Joardar SN, Abraham TJ, Kamilya D, Batabyal S (2009) Dynamic changes in immune-effector characteristics of Indian major carp, rohu (Labeo rohita) sensitized with Aeromonas hydrophila. Indian J Comp Microbiol Immunol Infect Dis 30:45–49
Dash S, Das SK, Samal J, Ojha PK, Patra JK, Thatoi H (2011) Dose dependence specific and non-specific immune responses of Indian major carp (L. rohita Ham) to intraperitoneal injection of formalin killed Aeromonas hydrophila whole cell vaccine. Vet Res Commun 35:541–552
Di Giulio RT, Habig C, Gallagher EP (1993) Effects of black rock harbor sediments on indices of biotransformation, oxidative stress, and DNA integrity in channel catfish. Aquat Toxicol 26:1–22
Diamond RD, Krzesicki R, Jao W (1978) Damage to pseudohyphal forms of Candida albicans by neutrophils in the absence of serum in vitro. J Clin Invest 61:349–359
Ellis AE (1990) Lysozyme assay. In: Stolen JS, Fletcher EC, Aderson DP, Robetrson DS, van Muiswinkel WB (eds) Techniques in fish immunology. SOS publications, USA, pp 101–103
Fridovich I (1995) Superoxide radical and superoxide dismutases. Ann Rev Biochem 64:97–112
Giri SS, Sukumaran V, Oviya M (2013) Potential probiotic Lactobacillus plantarum VSG3 improves the growth, immunity, and disease resistance of tropical freshwater fish, Labeo rohita. Fish Shellfish Immunol 34:660–666
Green IC, Wagner DA, Glogowski J, Skipper PL et al (1982) Analysis of nitrate, nitrite and (15 N) nitrate in biological fluids. Anal Biochem 126:131–138
Gross KA, Powell MD, Butler R, Morrison RN, Nowak BF (2005) Changes in the innate immune response of Atlantic salmon, Salmo salar L., exposed to experimental infection with Neoparamoeba sp. J Fish Dis 28:293–299
Gudding R, Lillehaug A, Evensen O (1999) Recent development in fish vaccinology. Vet Immunol Immunopathol 72:203–212
Hastings TS (1988) Furunculosis vaccines. In: Ellis AE (ed) Fish vaccination. Academic Press, New York, pp 93–111
John MB, Chandran MR, Aruna BV, Anbarasu K (2002) Production of superoxide anion by head kidney leukocytes of Indian major carps immunised with bacterins of Aeromonas hydrophila. Fish Shellfish Immunol 12:201–207
Kamilya D, Maiti TK, Joardar SN, Mal BC (2006) Adjuvant effect of mushroom glucan and bovine lactoferrin upon Aeromonas hydrophila vaccination in catla, Catla catla (Hamilton). J Fish Dis 29:331–337
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–273
Lund V, Jorgensen T, Holm KO, Eggest G (1991) Humoral immune response in Atlantic salmon, Salmo salar L., to cellular and extracellular antigens of Aeromonas salmonicida. J Fish Dis 14:443–452
Marsden MJ, Secombes CJ (1997) The influence of vaccine preparations on the induction of antigen specific responsiveness in rainbow trout, Oncorhynchus mykiss. Fish Shellfish Immunol 7:455–469
Midtlyng PJ, Reitan LJ, Lillehaug A, Ramstad A (1996) Protection, immune responses and side effects in Atlantic salmon (Salmo salar) vaccinated against furunculosis by different procedures. Fish Shellfish Immunol 6:599–613
Murray CK, Fletcher TCF (1976) The immunohistochemical localization of lysozyme in plaice, Pleuronectes platessa, tissues. J Fish Biol 9:324–329
Saikia D, Kamilya D (2012) Immune responses and protection in catla (Catla catla) vaccinated against epizootic ulcerative syndrome. Fish Shellfish Immunol 32:353–359
Secombes CJ (1990) Isolation of salmonid macrophages and analysis of their killing activity. In: Stolen JS, Fletcher TC, Anderson DP, Robertson BS, van Muiswinkel WB (eds) Techniques in fish immunology, vol I. SOS Publications, Fair Haven, pp 137–154
Swain P, Behura A, Dash S, Nayak SK (2007) Serum antibody response of Indian major carp, Labeo rohita to three species of pathogenic bacteria; Aeromonas hydrophila, Edwardsiella tarda and Pseudomonas fluorescens. Vet Immunol Immunopathol 117:137–141
Yin Z, Lam TJ, Sin YM (1997) Cytokine-mediated antimicrobial immune response of catfish, Clarias gariepinus, as a defence against Aeromonas hydrophila. Fish Shellfish Immunol 7:93–104
Acknowledgments
Author SSS like to acknowledge Erasmus Mundus-European Union framework MOVER for his post doctoral fellowship. SSG acknowledges DST, Government of India for awarding INSPIRE doctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Shib Sankar Sen and Sib Sankar Giri have contributed equally in this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sen, S.S., Giri, S.S. & Sukumaran, V. Immune responses and protection in rohu vaccinated against Aeromonas hydrophila infection. Aquacult Int 22, 1637–1648 (2014). https://doi.org/10.1007/s10499-014-9770-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-014-9770-x