Abstract
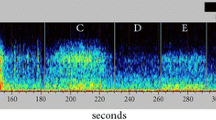
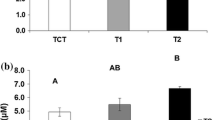
A controlled experiment mimicking Atlantic salmon (Salmo salar) pre-smolt farming conditions showed that fish exposed to 150 and 175% super oxygenated water produced higher levels of carbon dioxide with the subsequent decrease in water pH compared to control fish exposed to 100% O2. At the 7th day of exposure the hyperoxic fish showed larger individual variation in swimming activity compared to the controls. The individual variance in activity, tail beat frequency and scattering in the tanks among super oxygenated fish decreased from the 7th to the 21st day of exposure. The behavioural effects of hyperoxia were seen in relation to altered feed consumption halfway through the experiment, lower body weight, and altered haematological variables at day 21 of exposure. Plasma chloride was reduced in the exposed fish and haemoglobin decreased with increasing oxygen saturations. Plasma cortisol was elevated only in the 150% oxygenated group at day 21, while no effect on osmolality was recorded. The alterations in physiology and behaviour from day 7 to day 21 may be explained in terms of acclimation to increased oxygen saturations. This study shows that behaviour may be used as an indication of impaired water quality that may influence animal welfare negatively and eventually prevent an efficient production.


Similar content being viewed by others
References
Beitnger TL (1990) Behavioural reactions for the assessment of stress in fishes. J Great Lakes Res 16:495–528
Brauner CJ, Seidelin M, Madsen SS, Jensen FB (2000) Effects of freshwater hyperoxia and hypercapnia and their influences on subsequent seawater transfer in Atlantic salmon Salmo salar smolts. Can J Fish Aquat Sci 57:2054–2064. doi:10.1139/cjfas-57-10-2054
Caldwell AC, Hinshaw J (1994) Physiological and haematological responses in rainbow trout subjected to supplemental dissolved oxygen in fish culture. Aquaculture 126:183–193. doi:10.1016/0044-8486(94)90259-3
Caldwell AC, Hinshaw J (1995) Tolerance of rainbow trout to dissolved oxygen supplementation and a Yersinia ruckeri challenge. J Aquat Anim Health 7:168–171. doi :10.1577/1548-8667(1995)007<0168:CTORTT>2.3.CO;2
Claireaux G, Webber DM, Kerr SR, Boutilier RG (1995) Physiology and behaviour of free-swimming Atlantic cod Gadus morhua facing fluctuating salinity and oxygenation conditions. J Exp Biol 198:61–69
Dawkins MS (2004) Using behaviour to assess animal welfare. Anim Welf 13:s3–s7
Dejours P, Toulmond A, Truchot JP (1977) The effect of hyperoxia on the breathing of marine fishes. Comp Biochem Physiol 58A:409–411. doi:10.1016/0300-9629(77)90164-5
Depledge MH (1994) The rational basis for the use of biomarkers as ecotoxicological tools. In: Fossi MC, Leonzio C (eds) Nondestructive biomarkers in vertebrates. Lewis, Boca Raton, pp 271–295
Edsall DA, Smith CE (1991) Performance of rainbow trout and Snake River cutthroat trout reared in oxygen saturated water. Aquaculture 90:251–259. doi:10.1016/0044-8486(90)90249-M
Fernö A, Huse I, Juell JE, Bjordal Å (1995) Vertical distribution of Atlantic salmon Salmo salar L. in net pens: trade-off between surface light avoidance and food attraction. Aquaculture 132:285–296. doi:10.1016/0044-8486(94)00384-Z
Fivelstad S, Binde M (1994) Effects of reduced waterflow (increased loading) in soft water on Atlantic salmon smolts Salmo salar L. while maintaining oxygen at constant level by oxygenation of the inlet water. Aquac Eng 13:211–218. doi:10.1016/0144-8609(94)90004-3
Gilmour KM, Perry SF (1994) The effects of hypoxia, hyperoxia or hypercapnia on the acid-base disequilibrium in the arterial blood of rainbow trout. J Exp Biol 192:269–284
Goss GG, Wood CM, Laurent P, Perry SF (1994) Morphological responses of the rainbow trout Oncorhynchus mykiss gill to hyperoxia, base (NaHCO3) and acid (HCl) infusions. Fish Physiol Biochem 12:465–477. doi:10.1007/BF00004449
Helland SJ, Grisdale-Helland B, Nerland S (1996) A simple method for the measurement of daily feed intake of groups of fish in tanks. Aquaculture 139:157–163. doi:10.1016/0044-8486(95)01145-5
Huntingford F, Adams C (2005) Behavioural syndromes in farmed fish: implications for production and welfare. Behaviour 142:1213–1227. doi:10.1163/156853905774539382
Huntingford FA, Adams C, Braithwaite VA, Kadri S, Pottinger TG, Sandøe P et al (2006) Current issues in fish welfare. J Fish Biol 68:332–372. doi:10.1111/j.0022-1112.2006.001046.x
Juell JE (1995) The behaviour of Atlantic salmon in relation to efficient cage-rearing. Rev Fish Biol Fish 5:320–335. doi:10.1007/BF00043005
Kolok AS, Plaisance EP, Abdelghani A (1998) Individual variation in the swimming performance of fishes: an overlooked source of variation in toxicity studies. Environ Toxicol Chem 17:282–285. doi :10.1897/1551-5028(1998)017<0282:IVITSP>2.3.CO;2
Kramer DL (1987) Dissolved oxygen and fish behaviour. Environ Biol Fishes 18:81–92. doi:10.1007/BF00002597
Larsen BK, Jensen FB (1997) Influence of ionic composition on acid-base regulation in rainbow trout Oncorhynchus mykiss exposed to environmental hypercapnia. Fish Physiol Biochem 16:157–170. doi:10.1007/BF00004672
Liepelt A, Karbe L, Westendorf J (1995) Induction of DNA strand breaks in rainbow trout Oncorhynchus mykiss under hypoxic and hyperoxic conditions. Aquat Toxicol 33:177–181. doi:10.1016/0166-445X(95)00016-W
Little EE, Finger SE (1990) Swimming behaviour as an indicator of sublethal toxicity in fish. Environ Toxicol Chem 9:15–19
Lygren B, Hamre K, Waagbø R (2000) Effect of induced hyperoxia on the antioxidant status of Atlantic salmon Salmo salar L. fed three different levels of dietary vitamin E. Aquac Res 31:401–407. doi:10.1046/j.1365-2109.2000.00459.x
Øverli Ø, Sørensen C, Nilsson GE (2006) Behavioural indicators of stress-coping style in rainbow trout: do males and females react differently to novelty? Physiol Behav 87:506–512. doi:10.1016/j.physbeh.2005.11.012
Pickering AD (1998) Stress responses of farmed fish. In: Black KD, Pickering AD (eds) Biology of farmed fish. Sheffield Academic Press, University of Sheffield, UK, pp 222–255
Portz DE, Woodley CM, Cech JJ (2006) Stress-associated impacts of short-term holding on fishes. Rev Fish Biol Fish 16:125–170. doi:10.1007/s11160-006-9012-z
Priede IG, Solbé JF, Nott JE, O`Grandy KT, Cragg-Hine D (1988) Behaviour of adult Atlantic salmon Salmo salar L., in the estuary of the River Ribble in relation to variations in dissolved oxygen and tidal flow. J Fish Biol 33:133–139. doi:10.1111/j.1095-8649.1988.tb05567.x
Ritola O, Kiuru T, Koponen K, Mölsä H, Hänninen O, Lindström-Seppä P (1999) Rainbow trout Oncorhynchus mykiss exposed to oxygen supersaturation and handling stress: plasma cortisol and hepatic glutathione status. Acta Biol Hung 50:215–227
Ritola O, Livingstone DR, Peters LD, Lindström-Seppä P (2002) Antioxidant processes are affected in juvenile rainbow trout Oncorhynchus mykiss exposed to ozone and oxygen-supersaturated water. Aquaculture 210:1–19. doi:10.1016/S0044-8486(01)00823-7
Ruyet Person-Le J, Pichavant K, Vacher C, Le Bayon N, Sévère A, Boeuf G (2002) Effects of O2 supersaturation on metabolism and growth in juvenile turbot Scophthalmus maximus L. Aquaculture 205:373–383. doi:10.1016/S0044-8486(01)00689-5
Schurmann H, Steffensen JF (1994) Spontaneous swimming activity of Atlantic cod Gadus morhua exposed to graded hypoxia at three temperatures. J Exp Biol 197:129–142
Shelton RGJ, Johnstone ADF (1995) Preliminary aquarium observations of the schooling behaviour of Atlantic salmon Salmo salar L., smolts on transfer to sea water. Fish Res 24:73–78. doi:10.1016/0165-7836(94)00363-2
Steffensen JF, Farrell AP (1998) Swimming performance, venous oxygen tension cardiac performance of coronary-ligated rainbow trout Oncorhynchus mykiss exposed to progressive hypoxia. Comp Biochem Physiol 119A:585–592
Tornbull J, Bel A, Adams C, Bron J, Huntingford F (2005) Stocking density and welfare of cage farmed Atlantic salmon: application of a multivariate analysis. Aquaculture 243:121–132. doi:10.1016/j.aquaculture.2004.09.022
Wannamaker CM, Rice JA (2000) Effects of hypoxia on movements and behaviour of selected estuarine organisms from the south-eastern United States. J Exp Mar Biol Ecol 249:145–163. doi:10.1016/S0022-0981(00)00160-X
Wibe ÅE (2003) Behavioural effects of environmental pollution in threespine stickleback Gasterosteus aculeatus. Doctor Scient thesis, Norwegian University of Science and Technology
Wibe ÅE, Billing A, Rosenqvist G, Jenssen BM (2002) Butyl benzyl phthalate affects shoaling behaviour and bottom-dwelling behaviour in threespine stickleback. Environ Res 89A:180–187. doi:10.1006/enrs.2002.4360
Wilhelm D, Torres MA, Zaniboni E, Pedrosa RC (2005) Effect of different oxygen tensions on weight gain, feed conversion, and antioxidant status in piapara Leporinus elongates (Valenciennes, 1847). Aquaculture 244:349–357. doi:10.1016/j.aquaculture.2004.11.024
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice Hall, New Jersey
Acknowledgements
We would like to thank the technical staff at AKVAFORSK for help during the experimental setup. We are also grateful to the laboratory personnel at AKVAFORSK and Peter Torjessen (Hormone Laboratory, Aker University Hospital) for performing the physiological analyses. Finally, we wish to thank two anonymous reviewers for valuable comments on an earlier draft of this manuscript. This experiment was financially supported by the Research Council of Norway (project no. 133964/120).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Espmark, Å.M., Baeverfjord, G. Effects of hyperoxia on behavioural and physiological variables in farmed Atlantic salmon (Salmo salar) parr. Aquacult Int 17, 341–353 (2009). https://doi.org/10.1007/s10499-008-9206-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-008-9206-6