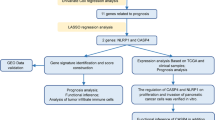
Abstract
Background
Pyroptosis, as a type of inflammatory programmed cell death, has been studied in inflammatory diseases and numerous cancers but its role in pancreatic ductal adenocarcinoma (PDAC) remains further exploration.
Methods
A TCGA-PDAC cohort was enrolled for bioinformatics analysis to investigate the effect of pyroptosis on the prognosis and drug sensitivity of patients. PA-TU-8988T and CFPAC-1 cells were selected for investigating the role of GSDMC in PDAC.
Results
A distinct classification pattern of PDAC mediated by 21 pyroptosis-related genes (PRGs) was identified. It was suggested that higher pyroptosis activity was associated with poor prognosis of patients and higher tumor proliferation rates. We further established a prognostic model based on three PRGs (GSDMC, CASP4 and NLRP1) and the TCGA-PDAC cohort was classified into low and high-risk subgroups. It is noteworthy that the high-risk group showed significantly higher tumor proliferation rates and was proved to be highly correlated with oxaliplatin resistance. Further experiments suggested that overexpression of GSDMC promoted the proliferation and oxaliplatin resistance of PA-TU-8988T cells in vitro and vivo, while downregulation of GSDMC showed opposite effects in CFPAC-1 cells. Finally, we found that the activation of pentose phosphate pathway (PPP) was the mechanism by which GSDMC overexpression promoted the proliferation and oxaliplatin resistance of pancreatic cancer cells.
Conclusions
In this study, we found that higher pyroptosis activity is associated with worse prognosis and oxaliplatin resistance of PDAC patients. In addition, as a core effector of pyroptosis, GSDMC promoted proliferation and oxaliplatin resistance of pancreatic cancer cells, which will provide new therapeutic target for PDAC patients.







Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, CA Cancer J Clin, 2023. 73(1): p. 17–48
Mizrahi JD, Surana R, Valle JW, Shroff RT (2020) Pancreatic cancer. Lancet 395(10242):2008–2020
Strobel O, Neoptolemos J, Jager D, Buchler MW (2019) Optimizing the outcomes of pancreatic cancer surgery. Nat Rev Clin Oncol 16(1):11–26
Neoptolemos JP, Kleeff J, Michl P, Costello E, Greenhalf W, Palmer DH (2018) Therapeutic developments in pancreatic cancer: current and future perspectives. Nat Rev Gastroenterol Hepatol 15(6):333–348
Morrison AH, Byrne KT, Vonderheide RH (2018) Immunotherapy and Prevention of Pancreatic Cancer. Trends Cancer 4(6):418–428
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM (2014) Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 74(11):2913–2921
Kovacs SB, Miao EA (2017) Gasdermins: effectors of pyroptosis. Trends Cell Biol 27(9):673–684
Aglietti RA, Dueber EC (2017) Recent insights into the Molecular Mechanisms underlying pyroptosis and Gasdermin Family Functions. Trends Immunol 38(4):261–271
Shi J, Gao W, Shao F (2017) Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem Sci 42(4):245–254
Broz P, Pelegrin P, Shao F (2020) The gasdermins, a protein family executing cell death and inflammation. Nat Rev Immunol 20(3):143–157
Wu D, Wei C, Li Y, Yang X, Zhou S (2021) Pyroptosis, a new breakthrough in Cancer Treatment. Front Oncol 11:698811
Wang G, Gao H, Dai S, Li M, Gao Y, Yin L, Zhang K, Zhang J, Jiang K, Miao Y, Lu Z (2023) Metformin inhibits neutrophil extracellular traps-promoted pancreatic carcinogenesis in obese mice. Cancer Lett 562:216155
Zychlinsky A, Prevost MC, Sansonetti PJ (1992) Shigella flexneri induces apoptosis in infected macrophages. Nature 358(6382):167–169
Ruan J, Wang S, Wang J (2020) Mechanism and regulation of pyroptosis-mediated in cancer cell death. Chem Biol Interact 323:109052
Gao J, Qiu X, Xi G, Liu H, Zhang F, Lv T, Song Y (2018) Downregulation of GSDMD attenuates tumor proliferation via the intrinsic mitochondrial apoptotic pathway and inhibition of EGFR/Akt signaling and predicts a good prognosis in nonsmall cell lung cancer. Oncol Rep 40(4):1971–1984
Zhou B, Zhang JY, Liu XS, Chen HZ, Ai YL, Cheng K, Sun RY, Zhou D, Han J, Wu Q (2018) Tom20 senses iron-activated ROS signaling to promote melanoma cell pyroptosis. Cell Res 28(12):1171–1185
Cui J, Zhou Z, Yang H, Jiao F, Li N, Gao Y, Wang L, Chen J, Quan M (2019) MST1 suppresses pancreatic Cancer Progression via ROS-Induced pyroptosis. Mol Cancer Res 17(6):1316–1325
Ding M, Liu J, Lv H, Zhu Y, Chen Y, Peng H, Fan S, Chen X (2023) Knocking down GALNT6 promotes pyroptosis of pancreatic ductal adenocarcinoma cells through NF-kappaB/NLRP3/GSDMD and GSDME signaling pathway. Front Oncol 13:1097772
Watabe K, Ito A, Asada H, Endo Y, Kobayashi T, Nakamoto K, Itami S, Takao S, Shinomura Y, Aikou T, Yoshikawa K, Matsuzawa Y, Kitamura Y, Nojima H (2001) Structure, expression and chromosome mapping of MLZE, a novel gene which is preferentially expressed in metastatic melanoma cells. Jpn J Cancer Res 92(2):140–151
Sun K, Chen RX, Li JZ, Luo ZX (2022) LINC00511/hsa-miR-573 axis-mediated high expression of Gasdermin C associates with dismal prognosis and tumor immune infiltration of breast cancer. Sci Rep 12(1):14788
Miguchi M, Hinoi T, Shimomura M, Adachi T, Saito Y, Niitsu H, Kochi M, Sada H, Sotomaru Y, Ikenoue T, Shigeyasu K, Tanakaya K, Kitadai Y, Sentani K, Oue N, Yasui W, Ohdan H (2016) Gasdermin C is upregulated by inactivation of transforming growth factor beta receptor type II in the Presence of Mutated apc, promoting Colorectal Cancer Proliferation. PLoS ONE 11(11):e0166422
Yan C, Niu Y, Li F, Zhao W, Ma L (2022) System analysis based on the pyroptosis-related genes identifies GSDMC as a novel therapy target for pancreatic adenocarcinoma. J Transl Med 20(1):455
Kondo T, Kanai M, Matsubara J, Yamaguchi D, Ura T, Kou T, Itani T, Nomura M, Funakoshi T, Yokoyama A, Doi K, Tamaoki M, Yoshimura M, Uza N, Yamada T, Masui T, Minamiguchi S, Matsumoto S, Ishikawa H, Muto M (2023) Association between homologous recombination gene variants and efficacy of oxaliplatin-based chemotherapy in advanced pancreatic cancer: prospective multicenter observational study. Med Oncol 40(5):144
Xia R, Hu C, Ye Y, Zhang X, Li T, He R, Zheng S, Wen X, Chen R (2023) HNF1A regulates oxaliplatin resistance in pancreatic cancer by targeting 53BP1 Int J Oncol, 62(4)
Chiu CF, Park JM, Chen HH, Mau CZ, Chen PS, Su YH, Chen HA, Liu YR, Hsieh TH, Chiu CC, Hung SW, Kuo CY, Chen YM, Chang CF (2022) Organic cation transporter 2 activation enhances sensitivity to oxaliplatin in human pancreatic ductal adenocarcinoma. Biomed Pharmacother 153:113520
Wu LS, Liu Y, Wang XW, Xu B, Lin YL, Song Y, Dong Y, Liu JL, Wang XJ, Liu S, Kong P, Han M, Li BH (2020) LPS enhances the chemosensitivity of oxaliplatin in HT29 cells via GSDMD-Mediated pyroptosis. Cancer Manag Res 12:10397–10409
Yin X, Tang B, Li JH, Wang Y, Zhang L, Xie XY, Zhang BH, Qiu SJ, Wu WZ, Ren ZG (2017) ID1 promotes hepatocellular carcinoma proliferation and confers chemoresistance to oxaliplatin by activating pentose phosphate pathway. J Exp Clin Cancer Res 36(1):166
Wang X, Zhang H, Yin S, Yang Y, Yang H, Yang J, Zhou Z, Li S, Ying G, Ba Y (2022) lncRNA-encoded pep-AP attenuates the pentose phosphate pathway and sensitizes colorectal cancer cells to Oxaliplatin. EMBO Rep 23(1):e53140
Lin J, Xia L, Oyang L, Liang J, Tan S, Wu N, Yi P, Pan Q, Rao S, Han Y, Tang Y, Su M, Luo X, Yang Y, Chen X, Yang L, Zhou Y, Liao Q (2022) The POU2F1-ALDOA axis promotes the proliferation and chemoresistance of colon cancer cells by enhancing glycolysis and the pentose phosphate pathway activity. Oncogene 41(7):1024–1039
Acknowledgements
Not applicable.
Funding
This work was supported by the grant from the National Natural Science Foundation of China (82173206, 82272974), Natural Science Foundation of Jiangsu Province (BK20221416), Construction Program of Jiangsu Provincial Clinical Research Center Support System (BL2014084), the Project of Invigorating Health Care through Science, Technology and Education, Jiangsu Provincial Medical Outstanding Talent (to YM, JCRCA2016009), Clinical Science and Technology Climbing program - “Spark” basic research project (ZJ202124). All samples used in current study were obtained from Pancreas Biobank, The First Affiliated Hospital with Nanjing Medical University, which is a part of Jiangsu Biobank of Clinical Resource.
Author information
Authors and Affiliations
Contributions
Guangfu Wang designed and drafted the manuscript. Shangnan Dai and Jin Chen conducted the analysis. Yong Gao, Lingdi Yin and Jinfan Zhang conducted the literature search. Kuirong Jiang, Yi Miao and Zipeng Lu designed, led, supervised the study, and critically revised the manuscript. All authors approved the final draft submitted, read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, G., Chen, J., Dai, S. et al. High pyroptosis activity in pancreatic adenocarcinoma: poor prognosis and oxaliplatin resistance. Apoptosis 29, 344–356 (2024). https://doi.org/10.1007/s10495-023-01901-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01901-w