Abstract
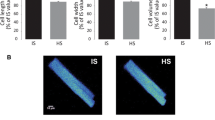
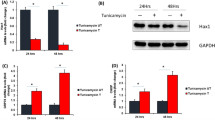
In different pathological situations, cardiac cells undergo hyperosmotic stress and cell shrinkage. This change in cellular volume has been associated with contractile dysfunction and cell death. However, the intracellular mechanisms involved in hyperosmotic stress-induced cell death have not been investigated in depth in adult cardiac myocytes. Given that osmotic stress has been shown to promote endoplasmic reticulum stress (ERS), a recognized trigger for apoptosis, we examined whether hyperosmotic stress triggers ERS in adult cardiac myocytes and if so whether this mechanism mediates hyperosmotic stress-induced cell death. Adult rat cardiomyocytes cultured overnight in a hypertonic solution (HS) containing mannitol as the osmolite, showed increased expression of ERS markers, GRP78, CHOP and cleaved-Caspase-12, compared with myocytes in isotonic solution (IS), suggesting that hyperosmotic stress induces ERS. In addition, HS significantly reduced cell viability and increased TUNEL staining and the expression of active Caspase-3, indicative of apoptosis. These effects were prevented with the addition of the ERS inhibitor, 4-PBA, indicating that hyperosmotic stress-induced apoptosis is mediated by ERS. Hyperosmotic stress-induced apoptosis was also prevented when cells were cultured in the presence of a Ca2+-chelating agent (EGTA) or the CaMKII inhibitor (KN93), suggesting that hyperosmotic stress-induced ERS is mediated by a Ca2+ and CaMKII-dependent mechanism. Similar results were observed when hyperosmotic stress was induced using glucose as the osmolite. We conclude that hyperosmotic stress promotes ERS by a CaMKII-dependent mechanism leading to apoptosis of adult cardiomyocytes. More importantly, we demonstrate that hyperosmotic stress-triggered ERS contributes to hyperglycemia-induced cell death.






Similar content being viewed by others
References
Hoffmann EK, Simonsen LO (1989) Membrane mechanisms in volume and pH regulation in vertebrate cells. Physiol Rev 69:315–382
Barbour HG, Loomis NM, Frankman RW, Warner JH (1924) Heat regulation and water exchange: V. the phase of blood dilution in man. J Physiol 59:300–305
Howarth FC, Qureshi MA, White E (2002) Effects of hyperosmotic shrinking on ventricular myocyte shortening and intracellular Ca(2+) in streptozotocin-induced diabetic rats. Pflugers Arch 444:446–451
Netti VA, Iovane AN, Vatrella MC et al (2016) Dehydration affects cardiovascular nitric oxide synthases and caveolins in growing rats. Eur J Nutr 55:33–43
Galvez A, Morales MP, Eltit JM et al (2001) A rapid and strong apoptotic process is triggered by hyperosmotic stress in cultured rat cardiac myocytes. Cell Tissue Res 304:279–285
Maldonado C, Cea P, Adasme T et al (2005) IGF-1 protects cardiac myocytes from hyperosmotic stress-induced apoptosis via CREB. Biochem Biophys Res Commun 336:1112–1118
Burg MB, Ferraris JD, Dmitrieva NI (2007) Cellular response to hyperosmotic stresses. Physiol Rev 87:1441–1474
Castillero E, Akashi H, Pendrak K et al (2015) Attenuation of the unfolded protein response and endoplasmic reticulum stress after mechanical unloading in dilated cardiomyopathy. Am J Physiol Heart Circ Physiol 309:H459–H470
Chalil S, Pierre N, Bakker AD et al (2015) Aging related ER stress is not responsible for anabolic resistance in mouse skeletal muscle. Biochem Biophys Res Commun 468:702–707
Jia LX, Zhang WM, Zhang HJ et al (2015) Mechanical stretch-induced endoplasmic reticulum stress, apoptosis and inflammation contribute to thoracic aortic aneurysm and dissection. J Pathol 236:373–383
Zhang Y, Ren J (2011) Thapsigargin triggers cardiac contractile dysfunction via NADPH oxidase-mediated mitochondrial dysfunction: Role of Akt dephosphorylation. Free Radic Biol Med 51:2172–2184
Zhu M, Zhou S, Huang Z, Wen J, Li H (2016) Ca2+-dependent endoplasmic reticulum stress regulates mechanical stress-mediated cartilage thinning. J Dent Res 95:889–896
Greenwood M, Greenwood MP, Paton JF, Murphy D (2015) Transcription factor creb3l1 regulates endoplasmic reticulum stress response genes in the osmotically challenged rat hypothalamus. PLoS ONE 10:e0124956
Mendes GC, Reis PA, Calil IP, Carvalho HH, Aragao FJ, Fontes EP (2013) GmNAC30 and GmNAC81 integrate the endoplasmic reticulum stress- and osmotic stress-induced cell death responses through a vacuolar processing enzyme. Proc Natl Acad Sci USA 110:19627–19632
Bohnert KR, McMillan JD, Kumar A (2018) Emerging roles of ER stress and unfolded protein response pathways in skeletal muscle health and disease. J Cell Physiol 233:67–78
Dickhout JG, Carlisle RE, Austin RC (2011) Interrelationship between cardiac hypertrophy, heart failure, and chronic kidney disease: endoplasmic reticulum stress as a mediator of pathogenesis. Circ Res 108:629–642
Liu L, Wu H, Zang J et al (2016) 4-Phenylbutyric acid reveals good beneficial effects on vital organ function via anti-endoplasmic reticulum stress in septic rats. Crit Care Med 44:e689–e701
Battson ML, Lee DM, Gentile CL (2017) Endoplasmic reticulum stress and the development of endothelial dysfunction. Am J Physiol Heart Circ Physiol 312:H355–H367
Li SY, Ren J (2008) Cardiac overexpression of alcohol dehydrogenase exacerbates chronic ethanol ingestion-induced myocardial dysfunction and hypertrophy: role of insulin signaling and ER stress. J Mol Cell Cardiol 44:992–1001
Ozcan U, Yilmaz E, Ozcan L et al (2006) Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 313:1137–1140
Okada K, Minamino T, Tsukamoto Y et al (2004) Prolonged endoplasmic reticulum stress in hypertrophic and failing heart after aortic constriction: possible contribution of endoplasmic reticulum stress to cardiac myocyte apoptosis. Circulation 110:705–712
Wang S, Chen X, Nair S, Sun D, Wang X, Ren J (2017) Deletion of protein tyrosine phosphatase 1B obliterates endoplasmic reticulum stress-induced myocardial dysfunction through regulation of autophagy. Biochim Biophys Acta Mol Basis Dis 1863:3060–3074
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086
Ferri KF, Kroemer G (2001) Organelle-specific initiation of cell death pathways. Nat Cell Biol 3:E255–E263
Zhang Q, Liu J, Chen S et al (2016) Caspase-12 is involved in stretch-induced apoptosis mediated endoplasmic reticulum stress. Apoptosis 21:432–442
Wang XZ, Lawson B, Brewer JW et al (1996) Signals from the stressed endoplasmic reticulum induce C/EBP-homologous protein (CHOP/GADD153). Mol Cell Biol 16:4273–4280
Momoi T (2004) Caspases involved in ER stress-mediated cell death. J Chem Neuroanat 28:101–105
Szegezdi E, Logue SE, Gorman AM, Samali A (2006) Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7:880–885
Hitomi J, Katayama T, Taniguchi M, Honda A, Imaizumi K, Tohyama M (2004) Apoptosis induced by endoplasmic reticulum stress depends on activation of caspase-3 via caspase-12. Neurosci Lett 357:127–130
Roe ND, Ren J (2013) Oxidative activation of Ca(2+)/calmodulin-activated kinase II mediates ER stress-induced cardiac dysfunction and apoptosis. Am J Physiol Heart Circ Physiol 304:H828–H839
Gonano LA, Morell M, Burgos JI et al (2014) Hypotonic swelling promotes nitric oxide release in cardiac ventricular myocytes: impact on swelling-induced negative inotropic effect. Cardiovasc Res 104:456–466
Morell M, Burgos JI, Gonano LA, Vila Petroff M (2018) AMPK-dependent nitric oxide release provides contractile support during hyperosmotic stress. Basic Res Cardiol 113:7
Allen DG, Smith GL (1987) The effects of hypertonicity on tension and intracellular calcium concentration in ferret ventricular muscle. J Physiol 383:425–439
Lado MG, Sheu SS, Fozzard HA (1984) Effects of tonicity on tension and intracellular sodium and calcium activities in sheep heart. Circ Res 54:576–585
Loiselle DS, Stienen GJ, van Hardeveld C et al (1996) The effect of hyperosmolality on the rate of heat production of quiescent trabeculae isolated from the rat heart. J Gen Physiol 108:497–514
Xie H, Zhu PH (2006) Effect of osmotic stress on spontaneous calcium sparks in rat ventricular myocytes. Acta Pharmacol Sin 27:877–887
Fulop M, Tannenbaum H, Dreyer N (1973) Ketotic hyperosmolar coma. Lancet 2:635–639
Pasquel FJ, Umpierrez GE (2014) Hyperosmolar hyperglycemic state: a historic review of the clinical presentation, diagnosis, and treatment. Diabetes Care 37:3124–3131
Vila-Petroff M, Salas MA, Said M et al (2007) CaMKII inhibition protects against necrosis and apoptosis in irreversible ischemia-reperfusion injury. Cardiovasc Res 73:689–698
Reinehr R, Haussinger D (2006) Hyperosmotic activation of the CD95 death receptor system. Acta Physiol (Oxf) 187:199–203
Dmitrieva NI, Michea LF, Rocha GM, Burg MB (2001) Cell cycle delay and apoptosis in response to osmotic stress. Comp Biochem Physiol A 130:411–420
Galvez AS, Ulloa JA, Chiong M et al (2003) Aldose reductase induced by hyperosmotic stress mediates cardiomyocyte apoptosis: differential effects of sorbitol and mannitol. J Biol Chem 278:38484–38494
Jian L, Lu Y, Lu S, Lu C (2016) Chemical chaperone 4-phenylbutyric acid reduces cardiac ischemia/reperfusion injury by alleviating endoplasmic reticulum stress and oxidative stress. Med Sci Monit 22:5218–5227
Jian L, Lu Y, Lu S, Lu C (2016) Chemical chaperone 4-phenylbutyric acid protects H9c2 cardiomyocytes from ischemia/reperfusion injury by attenuating endoplasmic reticulum stress-induced apoptosis. Mol Med Rep 13:4386–4392
Eisner V, Criollo A, Quiroga C et al (2006) Hyperosmotic stress-dependent NFkappaB activation is regulated by reactive oxygen species and IGF-1 in cultured cardiomyocytes. FEBS Lett 580:4495–4500
Shen M, Wang L, Yang G et al (2014) Baicalin protects the cardiomyocytes from ER stress-induced apoptosis: inhibition of CHOP through induction of endothelial nitric oxide synthase. PLoS ONE 9:e88389
Zhu P, Hu S, Jin Q et al (2018) Ripk3 promotes ER stress-induced necroptosis in cardiac IR injury: a mechanism involving calcium overload/XO/ROS/mPTP pathway. Redox Biol 16:157–168
Zhang YH, Zhao CQ, Jiang LS, Dai LY (2011) Cyclic stretch-induced apoptosis in rat annulus fibrosus cells is mediated in part by endoplasmic reticulum stress through nitric oxide production. Eur Spine J 20:1233–1243
Braun AP, Schulman H (1995) The multifunctional calcium/calmodulin-dependent protein kinase: from form to function. Annu Rev Physiol 57:417–445
Zhu W, Woo AY, Yang D, Cheng H, Crow MT, Xiao RP (2007) Activation of CaMKIIdeltaC is a common intermediate of diverse death stimuli-induced heart muscle cell apoptosis. J Biol Chem 282:10833–10839
Sapia L, Palomeque J, Mattiazzi A, Petroff MV (2010) Na+/K+-ATPase inhibition by ouabain induces CaMKII-dependent apoptosis in adult rat cardiac myocytes. J Mol Cell Cardiol 49:459–468
Brocker C, Thompson DC, Vasiliou V (2012) The role of hyperosmotic stress in inflammation and disease. Biomol Concepts 3:345–364
He Y, Zhou L, Fan Z, Liu S, Fang W (2018) Palmitic acid, but not high-glucose, induced myocardial apoptosis is alleviated by Nacetylcysteine due to attenuated mitochondrial-derived ROS accumulation-induced endoplasmic reticulum stress. Cell Death Dis 9:568
Wang L, Shen M, Guo X et al (2017) Volume-sensitive outwardly rectifying chloride channel blockers protect against high glucose-induced apoptosis of cardiomyocytes via autophagy activation. Sci Rep 7:44265
Garcia-Dorado D, Andres-Villarreal M, Ruiz-Meana M, Inserte J, Barba I (2012) Myocardial edema: a translational view. J Mol Cell Cardiol 52:931–939
Cordova-Casanova A, Olmedo I, Riquelme JA et al (2018) Mechanical stretch increases L-type calcium channel stability in cardiomyocytes through a polycystin-1/AKT-dependent mechanism. Biochim Biophys Acta Mol Cell Res 1865:289–296
Cheng WP, Wang BW, Lo HM, Shyu KG (2015) Mechanical stretch induces apoptosis regulator trb3 in cultured cardiomyocytes and volume-overloaded heart. PLoS ONE 10:e0123235
Ron D, Walter P (2007) Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol 8:519–529
Kim I, Xu W, Reed JC (2008) Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov 7:1013–1030
Minamino T, Kitakaze M (2010) ER stress in cardiovascular disease. J Mol Cell Cardiol 48:1105–1110
Xu C, Bailly-Maitre B, Reed JC (2005) Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 115:2656–2664
Yamaguchi O, Higuchi Y, Hirotani S et al (2003) Targeted deletion of apoptosis signal-regulating kinase 1 attenuates left ventricular remodeling. Proc Natl Acad Sci USA 100:15883–15888
Szegezdi E, Duffy A, O’Mahoney ME et al (2006) ER stress contributes to ischemia-induced cardiomyocyte apoptosis. Biochem Biophys Res Commun 349:1406–1411
Li Z, Zhang T, Dai H et al (2008) Endoplasmic reticulum stress is involved in myocardial apoptosis of streptozocin-induced diabetic rats. J Endocrinol 196:565–572
Khan MM, Yang WL, Wang P (2015) Endoplasmic reticulum stress in sepsis. Shock 44:294–304
Ko YE, Lee IH, So HM, Kim HW, Kim YH (2011) Mechanism of glutathione depletion during simulated ischemia-reperfusion of H9c2 cardiac myocytes. Free Radic Res 45:1074–1082
Siami S, Polito A, Porcher R et al (2013) Thirst perception and osmoregulation of vasopressin secretion are altered during recovery from septic shock. PLoS ONE 8:e80190
Zhang Z, Zhao L, Zhou Y et al (2017) Taurine ameliorated homocysteine-induced H9C2 cardiomyocyte apoptosis by modulating endoplasmic reticulum stress. Apoptosis 22:647–661
Li H, Zhang XY, Wu TJ et al (2013) Endoplasmic reticulum stress regulates rat mandibular cartilage thinning under compressive mechanical stress. J Biol Chem 288:18172–18183
Ricardo RA, Bassani RA, Bassani JW (2008) Osmolality- and Na+-dependent effects of hyperosmotic NaCl solution on contractile activity and Ca2+ cycling in rat ventricular myocytes. Pflugers Arch 455:617–626
Chiong M, Parra V, Eisner V et al (2010) Parallel activation of Ca(2+)-induced survival and death pathways in cardiomyocytes by sorbitol-induced hyperosmotic stress. Apoptosis 15:887–903
Erickson JR, Joiner ML, Guan X et al (2008) A dynamic pathway for calcium-independent activation of CaMKII by methionine oxidation. Cell 133:462–474
Palomeque J, Rueda OV, Sapia L et al (2009) Angiotensin II-induced oxidative stress resets the Ca2+ dependence of Ca2+-calmodulin protein kinase II and promotes a death pathway conserved across different species. Circ Res 105:1204–1212
Burgos JI, Yeves AM, Barrena JP, Portiansky EL, Vila-Petroff MG, Ennis IL (2017) Nitric oxide and CaMKII: critical steps in the cardiac contractile response To IGF-1 and swim training. J Mol Cell Cardiol 112:16–26
Zhang T, Brown JH (2004) Role of Ca2+/calmodulin-dependent protein kinase II in cardiac hypertrophy and heart failure. Cardiovasc Res 63:476–486
Skoularigis J, Melidonis A, Westermann D, Georgiopoulou VV, Karagiannis G, Giamouzis G (2016) Interactions between diabetes and the heart. J Diabetes Res 2016:8032517
Pappachan JM, Varughese GI, Sriraman R, Arunagirinathan G (2013) Diabetic cardiomyopathy: pathophysiology, diagnostic evaluation and management. World J Diabetes 4:177–189
Ouyang C, You J, Xie Z (2014) The interplay between autophagy and apoptosis in the diabetic heart. J Mol Cell Cardiol 71:71–80
Badalzadeh R, Mokhtari B, Yavari R (2015) Contribution of apoptosis in myocardial reperfusion injury and loss of cardioprotection in diabetes mellitus. J Physiol Sci 65:201–215
Fiorentino TV, Prioletta A, Zuo P, Folli F (2013) Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr Pharm Des 19:5695–5703
Volpe CMO, Villar-Delfino PH, Dos Anjos PMF, Nogueira-Machado JA (2018) Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death Dis 9:119
Li Z, Zhang T, Dai H et al (2007) Involvement of endoplasmic reticulum stress in myocardial apoptosis of streptozocin-induced diabetic rats. J Clin Biochem Nutr 41:58–67
Liu M, Li Y, Liang B et al (2018) Hydrogen sulfide attenuates myocardial fibrosis in diabetic rats through the JAK/STAT signaling pathway. Int J Mol Med 41:1867–1876
Federico M, Portiansky EL, Sommese L et al (2017) Calcium-calmodulin-dependent protein kinase mediates the intracellular signalling pathways of cardiac apoptosis in mice with impaired glucose tolerance. J Physiol 595:4089–4108
Luo M, Guan X, Luczak ED et al (2013) Diabetes increases mortality after myocardial infarction by oxidizing CaMKII. J Clin Invest 123:1262–1274
Ren J, Gintant GA, Miller RE, Davidoff AJ (1997) High extracellular glucose impairs cardiac E-C coupling in a glycosylation-dependent manner. Am J Physiol 273:H2876–H2883
Ren J, Duan J, Hintz KK, Ren BH (2003) High glucose induces cardiac insulin-like growth factor I resistance in ventricular myocytes: role of Akt and ERK activation. Cardiovasc Res 57:738–748
Hasnain SZ, Prins JB, McGuckin MA (2016) Oxidative and endoplasmic reticulum stress in beta-cell dysfunction in diabetes. J Mol Endocrinol 56:R33–R54
Acknowledgements
The technical support of Mónica Rando and Omar Castillo are gratefully acknowledged.
Funding
This study was supported by Grants PICT 1678 from Fondo para la Investigación Científica y Tecnológica (FONCyT) and PIP 0270 from El Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) to Martín Vila Petroff.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Burgos, J.I., Morell, M., Mariángelo, J.I.E. et al. Hyperosmotic stress promotes endoplasmic reticulum stress-dependent apoptosis in adult rat cardiac myocytes. Apoptosis 24, 785–797 (2019). https://doi.org/10.1007/s10495-019-01558-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-019-01558-4