Abstract
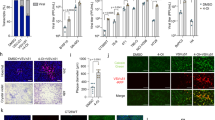
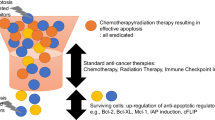
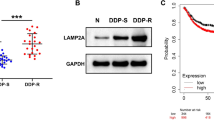
In this study, we demonstrated that survivin downregulation with TRAIL expression greatly enhanced the cytotoxic death of pancreatic cancer cells after gemcitabine treatment. Using real-time RT-PCR, we analyzed five survivin shRNAs to identify the best target sequence for suppression of human survivin, with the goal of treating gemcitabine-resistant pancreatic cancer cells. Survivin shRNA 5, corresponding to target 5, showed the greatest reduction in survivin mRNA levels. Furthermore, combined treatment with survivin shRNA-expressing adenovirus with gemcitabine plus TRAIL decreased uncleaved PARP and increased consequent PARP cleavage, which was correlated with the greatest levels of survivin downregulation and cell death. These results indicate that survivin functions as a common mediator of gemcitabine- and TRAIL-induced cell death. Using a nude mouse model implanted with MiaPaCa-2 pancreatic cancer cells, we observed tumor regression induced by an oncolytic adenovirus expressing survivin shRNA and TRAIL plus gemcitabine. Together, our findings provide a strong rationale for treating pancreatic cancer patients with both gemcitabine and oncolytic adenovirus armed with survivin shRNA and TRAIL.







Similar content being viewed by others
Abbreviations
- RT-PCR:
-
Reverse-transcription polymerase chain reaction
- TRAIL:
-
Tumor necrosis factor-related apoptosis-inducing ligand
- MTS:
-
3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium
- FLIP:
-
FLICE-inhibitory protein
- IAP:
-
Inhibitor of apoptosis protein
- BIR:
-
Baculovirus IAP repeat
- FACS:
-
Fluorescence-activated cell sorter
- FITC:
-
Fluorescein isothiocyanate
- PI:
-
Propidium iodide
- XIAP:
-
X-linked inhibitor of apoptosis protein
- HEK-293:
-
Human embryonic kidney-293
- PARP:
-
Poly (ADP-ribose) polymerase
References
Kim MP, Gallick GE (2008) Gemcitabine resistance in pancreatic cancer: picking the key players. Clin Cancer Res: Off J Am Assoc Cancer Res 14:1284–1285
Hilbig A, Oettle H (2008) Gemcitabine in the treatment of metastatic pancreatic cancer. Expert Rev Anticancer Ther 8:511–523
Hidalgo M (2010) Pancreatic cancer. N Engl J Med 362:1605–1617
Long J, Zhang Y, Yu X, Yang J, LeBrun DG, Chen C et al (2011) Overcoming drug resistance in pancreatic cancer. Expert Opin Ther Targets 15:817–828
Goldstein D, El-Maraghi RH, Hammel P, Heinemann V, Kunzmann V, Sastre J et al (2015) nab-Paclitaxel plus gemcitabine for metastatic pancreatic cancer: long-term survival from a phase III trial. J Nat Cancer Inst 107:dju413
Lo Re G, Santeufemia DA, Foltran L, Bidoli E, Basso SM, Lumachi F (2015) Prognostic factors of survival in patients treated with nab-paclitaxel plus gemcitabine regimen for advanced or metastatic pancreatic cancer: a single institutional experience. Oncotarget 6:8255–8260
Ambrosini G, Adida C, Altieri DC (1997) A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 3:917–921
Uchida H, Tanaka T, Sasaki K, Kato K, Dehari H, Ito Y et al (2004) Adenovirus-mediated transfer of siRNA against survivin induced apoptosis and attenuated tumor cell growth in vitro and in vivo. Mol Ther: J Am Soc Gene Ther 10:162–171
Altieri DC (2008) New wirings in the survivin networks. Oncogene 27:6276–6284
Azuhata T, Scott D, Griffith TS, Miller M, Sandler AD (2006) Survivin inhibits apoptosis induced by TRAIL, and the ratio between survivin and TRAIL receptors is predictive of recurrent disease in neuroblastoma. J Pediatr Surg 41:1431–1440
Kanwar JR, Kamalapuram SK, Kanwar RK (2013) Survivin signaling in clinical oncology: a multifaceted dragon. Med Res Rev 33:765–789
Liu WS, Yan HJ, Qin RY, Tian R, Wang M, Jiang JX et al (2009) siRNA directed against survivin enhances pancreatic cancer cell gemcitabine chemosensitivity. Dig Dis Sci 54:89–96
Yoon DH, Shin JS, Jin DH, Hong SW, Jung KA, Kim SM et al (2012) The survivin suppressant YM155 potentiates chemosensitivity to gemcitabine in the human pancreatic cancer cell line MiaPaCa-2. Anticancer Res 32:1681–1688
Altieri DC (2008) Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer 8:61–70
Zhang R, Ma L, Zheng M, Ren J, Wang T, Meng Y et al (2010) Survivin knockdown by short hairpin RNA abrogates the growth of human hepatocellular carcinoma xenografts in nude mice. Cancer Gene Ther 17:275–288
Li QX, Zhao J, Liu JY, Jia LT, Huang HY, Xu YM et al (2006) Survivin stable knockdown by siRNA inhibits tumor cell growth and angiogenesis in breast and cervical cancers. Cancer Biol Ther 5:860–866
Ambrosini G, Adida C, Sirugo G, Altieri DC (1998) Induction of apoptosis and inhibition of cell proliferation by survivin gene targeting. J Biol Chem 273:11177–11182
Olie RA, Simoes-Wust AP, Baumann B, Leech SH, Fabbro D, Stahel RA et al (2000) A novel antisense oligonucleotide targeting survivin expression induces apoptosis and sensitizes lung cancer cells to chemotherapy. Cancer Res 60:2805–2809
Pennati M, Colella G, Folini M, Citti L, Daidone MG, Zaffaroni N (2002) Ribozyme-mediated attenuation of survivin expression sensitizes human melanoma cells to cisplatin-induced apoptosis. J Clin Investig 109:285–286
Shen W, Wang CY, Wang XH, Fu ZX (2009) Oncolytic adenovirus mediated Survivin knockdown by RNA interference suppresses human colorectal carcinoma growth in vitro and in vivo. J Exp Clin Cancer Res 28:81
Kunnumakkara AB, Guha S, Krishnan S, Diagaradjane P, Gelovani J, Aggarwal BB (2007) Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-kappaB-regulated gene products. Cancer Res 67:3853–3861
Wang S, El-Deiry WS (2003) TRAIL and apoptosis induction by TNF-family death receptors. Oncogene 22:8628–8633
Kauh J, Fan S, Xia M, Yue P, Yang L, Khuri FR et al (2010) c-FLIP degradation mediates sensitization of pancreatic cancer cells to TRAIL-induced apoptosis by the histone deacetylase inhibitor LBH589. PLoS ONE 5:e10376
Fulda S, Meyer E, Debatin KM (2002) Inhibition of TRAIL-induced apoptosis by Bcl-2 overexpression. Oncogene 21:2283–2294
Zhu H, Guo W, Zhang L, Davis JJ, Wu S, Teraishi F et al (2005) Enhancing TRAIL-induced apoptosis by Bcl-X(L) siRNA. Cancer Biol Ther 4:393–397
Zhang L, Fang B (2005) Mechanisms of resistance to TRAIL-induced apoptosis in cancer. Cancer Gene Ther 12:228–237
Premkumar DR, Jane EP, Foster KA, Pollack IF (2013) Survivin inhibitor YM-155 sensitizes tumor necrosis factor- related apoptosis-inducing ligand-resistant glioma cells to apoptosis through Mcl-1 downregulation and by engaging the mitochondrial death pathway. J Pharmacol Exp Ther 346:201–210
He SQ, Rehman H, Gong MG, Zhao YZ, Huang ZY, Li CH et al (2007) Inhibiting survivin expression enhances TRAIL-induced tumoricidal activity in human hepatocellular carcinoma via cell cycle arrest. Cancer Biol Ther 6:1247–1257
Xu ZW, Kleeff J, Friess H, Buchler MW, Solioz M (2003) Synergistic cytotoxic effect of TRAIL and gemcitabine in pancreatic cancer cells. Anticancer Res 23:251–258
Mao L, Yang C, Li L, Nai L, Fan L, Wang J et al (2014) Replication-competent adenovirus expressing TRAIL synergistically potentiates the antitumor effect of gemcitabine in bladder cancer cells. Tumour Biol: J Int Soc Oncodev Biol Med 35:5937–5944
Yang J, Ouyang J, Ouyang L, Ouyang L, Chen Y (2013) Inhibition of cell proliferation and increase of chemosensitivity by simultaneous knockdown of XIAP and survivin in pancreatic carcinoma cells. Oncol Res 21:43–50
Retzer-Lidl M, Schmid RM, Schneider G (2007) Inhibition of CDK4 impairs proliferation of pancreatic cancer cells and sensitizes towards TRAIL-induced apoptosis via downregulation of survivin. Int J Cancer 121:66–75
Kim SY, Kang S, Song JJ, Kim JH (2013) The effectiveness of the oncolytic activity induced by Ad5/F35 adenoviral vector is dependent on the cumulative cellular conditions of survival and autophagy. Int J Oncol 42:1337–1348
Seol DW, Billiar TR (1999) A caspase-9 variant missing the catalytic site is an endogenous inhibitor of apoptosis. J Biol Chem 274:2072–2076
Seol JW, Chaudhari AA, Lee YJ, Kang HS, Kim IS, Kim NS et al (2007) Regulation of DR-5 protein and mitochondrial transmembrane potential by gemcitabine, a possible mechanism of gemcitabine-enhanced TRAIL-induced apoptosis. Oncol Rep 18:523–529
Rajeshkumar NV, Rasheed ZA, Garcia-Garcia E, Lopez-Rios F, Fujiwara K, Matsui WH et al (2010) A combination of DR5 agonistic monoclonal antibody with gemcitabine targets pancreatic cancer stem cells and results in long-term disease control in human pancreatic cancer model. Mol Cancer Ther 9:2582–2592
Arlt A, Muerkoster SS, Schafer H (2013) Targeting apoptosis pathways in pancreatic cancer. Cancer Lett 332:346–358
Ibrahim SM, Ringel J, Schmidt C, Ringel B, Muller P, Koczan D et al (2001) Pancreatic adenocarcinoma cell lines show variable susceptibility to TRAIL-mediated cell death. Pancreas 23:72–79
Alemany R (2012) Chapter four-design of improved oncolytic adenoviruses. Adv Cancer Res 115:93–114
Alemany R (2013) Viruses in cancer treatment. Clin Trans Oncol: Offl Publ Fed Span Oncol Soc Nat Cancer Inst Mexico 15:182–188
Sharma A, Tandon M, Bangari DS, Mittal SK (2009) Adenoviral vector-based strategies for cancer therapy. Curr Drug Ther 4:117–138
Kang S, Kim JH, Kim SY, Kang D, Je S, Song JJ (2014) Establishment of a mouse melanoma model system for the efficient infection and replication of human adenovirus type 5-based oncolytic virus. Biochem Biophys Res Commun 453:480–485
Acknowledgments
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) and funded by the Ministry of Education, Science, and Technology (NRF-2013R1A1A2A100005494) and a faculty research grant of Yonsei University College of Medicine for 2014 (6-2014-0138). Zhezhu Han, Seungha Lee and Suyeon Je were supported by the Brain Korea 21 Plus project for Medical Science (Yonsei University, College of Medicine, Seoul, Republic of Korea).
Authors’ contributions
ZZH and SHL carried out overall research, experimental studies and data acquisition. SYJ participated in the animal study and helped to western blots. CYE participated in the immunohistochemistry and data acquisition. HJC carried out the data acquisition, and helped to draft the manuscript. JJS participated in the overall study design and drafted and revised the manuscript. JHK proposed the study and participated in its design and helped to draft, and assisted writing the manuscript. All authors had already read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Zhezhu Han and Seungha Lee contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10495_2015_1208_MOESM1_ESM.tif
Supplementary figure 1. Screening of human survivin short hairpin RNAs (shRNAs), with sequences of five shRNA oligomers targeting survivin. The selected target sequence is indicated in bold (bottom of top panel). Five oligomers of the target and the positive control shRNA were transfected into HeLa cells. The knockdown efficiency of each oligomer was measured using quantitative real-time PCR to amplify survivin. Relative expression levels of survivin were plotted after normalization to the scrambled shRNA as a negative control (bottom panel) (TIFF 1082 kb)
10495_2015_1208_MOESM2_ESM.tif
Supplementary figure 2. The number of infectious viral particles was determined as a measure of oncolytic adenoviral replication. MiaPaCa-2 cells were infected with Ad-3484-NC or Ad-3484-shSurvivin or Ad-3484-TRAIL-shSurvivin adenovirus at an MOI of 50 for various times. After infection, the supernatants were examined for virus production. Error bars represent standard errors from three independent experiments (TIFF 419 kb)
Rights and permissions
About this article
Cite this article
Han, Z., Lee, S., Je, S. et al. Survivin silencing and TRAIL expression using oncolytic adenovirus increase anti-tumorigenic activity in gemcitabine-resistant pancreatic cancer cells. Apoptosis 21, 351–364 (2016). https://doi.org/10.1007/s10495-015-1208-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1208-z