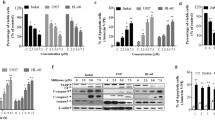
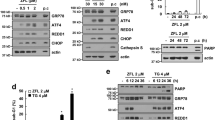
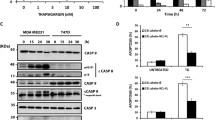
Abstract
Tunicamycin (TN), one of the endoplasmic reticulum stress inducers, has been reported to inhibit tumor cell growth and exhibit anticarcinogenic activity. However, the mechanism by which TN initiates apoptosis remains poorly understood. In the present study, we investigated the effect of TN on the apoptotic pathway in U937 cells. We show that TN induces apoptosis in association with caspase-3 activation, generation of reactive oxygen species (ROS), and downregulation of survivin expression. P38 MAPK (mitogen-activated protein kinase) and the generation of ROS signaling pathway play crucial roles in TN-induced apoptosis in U937 cells. We hypothesized that TN-induced activation of p38 MAPK signaling pathway is responsible for cell death. To test this hypothesis, we selectively inhibited MAPK during treatment with TN. Our data demonstrated that inhibitor of p38 (SB), but not ERK (PD) or JNK (SP), partially maintained apoptosis during treatment with TN. Pre-treatment with NAC and GSH markedly prevented cell death, suggesting a role for ROS in this process. Ectopic expression of survivin in U937 cells attenuated TN-induced apoptosis by suppression of caspase-3 cleavage, mitochondrial membrane potential, and cytochrome c release in U937 cells. Taken together, our results show that TN modulates multiple components of the apoptotic response of human leukemia cells and raise the possibility of a novel therapeutic strategy for hematological malignancies.






Similar content being viewed by others
Abbreviations
- DAPI:
-
4′,6′-Diamidino-2-phenylindole
- DCFDA:
-
2′7′-Dichlorodihydrofluorescein diacetate
- NAC:
-
N-acetylcysteine
- PBS:
-
Phosphate-buffered saline
- PI:
-
Propidium iodide
- ROS:
-
Reactive oxygen species
- UPR:
-
Unfolded protein response
- z-VAD-fmk:
-
Benzyl carbonyl-Val-Ala-Asp-fluoromethyl ketone
References
Noda I, Fujieda S, Seki M, Tanaka N, Sunaga H, Ohtsubo T, Tsuzuki H, Fan GK, Saito H (1999) Inhibition of N-linked glycosylation by tunicamycin enhances sensitivity to cisplatin in human head-and-neck carcinoma cells. Int J Cancer 80:279–284
Dennis JW, Granovsky M, Warren CE (1999) Protein glycosylation in development and disease. BioEssays 21:412–421
Goss PE, Baker MA, Carver JP, Dennis JW (1995) Inhibitors of carbohydrate processing: a new class of anticancer agents. Clin Cancer Res 1:935–944
Bentley J, Quinn DM, Pitman RS, Warr JR, Kellett GL (1997) The human KB multidrug-resistant cell line KB-C1 is hypersensitive to inhibitors of glycosylation. Cancer Lett 115:221–227
Carlberg M, Larsson O (1993) Role of N-linked glycosylation in cell-cycle progression and initiation of DNA synthesis in tumor-transformed human fibroblasts. Anticancer Res 13:167–171
Martínez JA, Torres-Negrón I, Amigó LA, Banerjee DK (1999) Expression of Glc3Man9GlcNAc2-PP-Dol is a prerequisite for capillary endothelial cell proliferation. Cell Mol Biol (Noisy-le-grand) 45:137–152
Hiss D, Gabriels G, Jacobs P, Folb P (1996) Tunicamycin potentiates drug cytotoxicity and vincristine retention in multidrug resistant cell lines. Eur J Cancer 32:2164–2172
Kramer R, Weber TK, Arceci R, Ramchurren N, Kastrinakis WV, Steele G Jr, Summerhayes IC (1995) Inhibition of N-linked glycosylation of P-glycoprotein by tunicamycin results in a reduced multidrug resistance phenotype. Br J Cancer 71:670–675
Lou LX, Geng B, Yu F, Zhang J, Pan CS, Chen L, Qi YF, Ke Y, Wang X, Tang CS (2006) Endoplasmic reticulum stress response is involved in the pathogenesis of stress induced gastric lesions in rats. Life Sci 79:1856–1864
Rao RV, Poksay KS, Castro-Obregon S, Schilling B, Row RH, del Rio G, Gibson BW, Ellerby HM, Bredesen DE (2004) Molecular components of a cell death pathway activated by endoplasmic reticulum stress. J Biol Chem 279:177–187
Reimertz C, Kögel D, Rami A, Chittenden T, Prehn JH (2003) Gene expression during ER stress-induced apoptosis in neurons: induction of the BH3-only protein Bbc3/PUMA and activation of the mitochondrial apoptosis pathway. J Cell Biol 162:587–597
Walker BK, Lei H, Krag SS (1998) A functional link between N-linked glycosylation and apoptosis in Chinese hamster ovary cells. Biochem Biophys Res Commun 250:264–270
Kleizen B, Braakman I (2004) Protein folding and quality control in the endoplasmic reticulum. Curr Opin Cell Biol 16:343–349
Aridor M, Balch WE (1999) Integration of endoplasmic reticulum signaling in health and disease. Nat Med 5:745–751
Xu C, Bailly-Maitre B, Reed JC (2005) Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 115:2656–2664
Kim I, Xu W, Reed JC (2008) Cell death and endoplasmic reticulum stress: disease relevance and herapeutic opportunities. Nat Rev Drug Discov 7:1013–1030
Rutkowski DT, Kaufman RJ (2004) A trip to the ER: coping with stress. Trends Cell Biol 14:20–28
Elbein AD (1987) Inhibitors of the biosynthesis and processing of N-linked oligosaccharide chains. Annu Rev Biochem 56:497–534
Natsume Y, Ito S, Satsu H, Shimizu M (2009) Protective effect of quercetin on ER stress caused by calcium dynamics dysregulation in intestinal epithelial cells. Toxicology 258:164–175
Xu C, Bailly-Maitre B, Reed JC (2005) Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 115:2656–2664
Hattori M, Sakamoto H, Satoh K, Yamamoto T (2001) DNA demethylase is expressed in ovarian cancers and the expression correlates with demethylation of CpG sites in the promoter region of c-erbB-2 and survivin genes. Cancer Lett 169:155–164
Li F, Altieri DC (1999) Transcriptional analysis of human survivin gene expression. Biochem J 344Pt 2:305–311
Hoffman WH, Biade S, Zilfou JT, Chen J, Murphy M (2002) Transcriptional repression of the anti-apoptotic survivin gene by wild type p53. J Biol Chem 277:3247–3257
Mirza A, McGuirk M, Hockenberry TN, Wu Q, Ashar H, Black S, Wen SF, Wang L, Kirschmeier P, Bishop WR, Nielsen LL, Pickett CB, Liu S (2002) Human survivin is negatively regulated by wild-type p53 and participates in p53-dependent apoptotic pathway. Oncogene 21:2613–2622
Simoncini S, Sapet C, Camoin-Jau L, Bardin N, Harlé JR, Sampol J, Dignat-George F, Anfosso F (2005) Role of reactive oxygen species and p38 MAPK in the induction of the pro-adhesive endothelial state mediated by IgG from patients with anti-phospholipid syndrome. Int Immunol 17:489–500
Kim BM, Lee KH, Hong IS, Hong SH (2012) p38 mitogen-activated protein kinase is a key regulator of 5-phenylselenyl- and 5-methylselenyl-methyl-2′-deoxyuridine-induced apoptosis in human HL-60 cells. Biochem Biophys Res Commun 417:237–244
Kim MJ, Lee TH, Kim SH, Choi YJ, Heo J, Kim YH (2010) Triptolide inactivates Akt and induces caspase-dependent death in cervical cancer cells via the mitochondrial pathway. Int J Oncol 37:1177–1185
Fleury C, Mignotte B, Vayssière JL (2002) Mitochondrial reactive oxygen species in cell death signaling. Biochimie 84:131–141
Wen J, You KR, Lee SY, Song CH, Kim DG (2002) Oxidative stress-mediated apoptosis. The anticancer effect of the sesquiterpene lactone parthenolide. J Biol Chem 277:38954–38964
Jin HO, Yoon SI, Seo SK, Lee HC, Woo SH, Yoo DH, Lee SJ, Choe TB, An S, Kwon TJ, Kim JI, Park MJ, Hong SI, Park IC, Rhee CH (2006) Synergistic induction of apoptosis by sulindac and arsenic trioxide in human lung cancer A549 cells via reactive oxygen species-dependent down-regulation of survivin. Biochem Pharmacol 72:1228–1236
Guan L, Han B, Li Z, Hua F, Huang F, Wei W, Yang Y, Xu C (2009) Sodium selenite induces apoptosis by ROS-mediated endoplasmic reticulum stress and mitochondrial dysfunction in human acute promyelocytic leukemia NB4 cells. Apoptosis 14:218–225
Ono Y, Shimazawa M, Ishisaka M, Oyagi A, Tsuruma K, Hara H (2012) Imipramine protects mouse hippocampus against tunicamycin-induced cell death. Eur J Pharmacol 696:83–88
Shore GC, Papa FR, Oakes SA (2011) Signaling cell death from the endoplasmic reticulum stress response. Curr Opin Cell Biol 23:143–149
Okada K, Minamino T, Tsukamoto Y, Liao Y, Tsukamoto O, Takashima S, Hirata A, Fujita M, Nagamachi Y, Nakatani T (2004) Prolonged endoplasmic reticulum stress in hypertrophic and failing heart after aortic constriction: possible contribution of endoplasmic reticulum stress to cardiac myocyte apoptosis. Circulation 110:705–712
Zhao H, Liao Y, Minamino T, Asano Y, Asakura M, Kim J, Asanuma H, Takashima S, Hori M, Kitakaze M (2008) Inhibition of cardiac remodeling by pravastatin is associated with amelioration of endoplasmic reticulum stress. Hypertens Res 31:1977–1987
Marciniak SJ, Yun CY, Oyadomari S, Novoa I, Zhang Y, Jungreis R, Nagata K, Harding HP, Ron D (2004) CHOP induces death by promoting protein synthesis and oxidation in the stressed endoplasmic reticulum. Genes Dev 18:3066–3077
Li Y, Xie M, Yang J, Yang D, Deng R, Wan Y, Yan B (2006) The expression of antiapoptotic protein survivin is transcriptionally upregulated by DEC1 primarily through multiple sp1 binding sites in the proximal promoter. Oncogene 25:3296–3306
Cosgrave N, Hill AD, Young LS (2006) Growth factor-dependent regulation of survivin by c-myc in human breast cancer. J Mol Endocrinol 37:377–390
Zhu N, Gu L, Findley HW, Chen C, Dong JT, Yang L, Zhou M (2006) KLF5 Interacts with p53 in regulating survivin expression in acute lymphoblastic leukemia. J Biol Chem 281:14711–14718
Gritsko T, Williams A, Turkson J, Kaneko S, Bowman T, Huang M, Nam S, Eweis I, Diaz N, Sullivan D, Yoder S, Enkemann S, Eschrich S, Lee JH, Beam CA, Cheng J, Minton S, Muro-Cacho CA, Jove R (2006) Persistent activation of stat3 signaling induces survivin gene expression and confers resistance to apoptosis in human breast cancer cells. Clin Cancer Res 12:11–19
Peng XH, Karna P, Cao Z, Jiang BH, Zhou M, Yang L (2006) Cross-talk between epidermal growth factor receptor and hypoxia-inducible factor-1alpha signal pathways increases resistance to apoptosis by up-regulating survivin gene expression. J Biol Chem 281:25903–25914
Dasgupta P, Kinkade R, Joshi B, Decook C, Haura E, Chellappan S (2006) Nicotine inhibits apoptosis induced by chemotherapeutic drugs by up-regulating XIAP and survivin. Proc Natl Acad Sci USA 103:6332–6337
Kim YH, Lee DH, Jeong JH, Guo ZS, Lee YJ (2008) Quercetin augments TRAIL-induced apoptotic death: involvement of the ERK signal transduction pathway. Biochem Pharmacol 75:1946–1958
Ringshausen I, Schneller F, Bogner C, Hipp S, Duyster J, Peschel C, Decker T (2002) Constitutively activated phosphatidylinositol-3 kinase (PI-3 K) is involved in the defect of apoptosis in B-CLL: association with protein kinase Cdelta. Blood 100:3741–3748
Hu P, Han Z, Couvillon AD, Exton JH (2004) Critical role of endogenous Akt/IAPs and MEK1/ERK pathways in counteracting endoplasmic reticulum stress-induced cell death. J Biol Chem 279:49420–49429
Abdelrahim M, Liu S, Safe S (2005) Induction of endoplasmic reticulum-induced stress genes in Panc-1 pancreatic cancer cells is dependent on Sp proteins. J Biol Chem 280:16508–16513
Acknowledgments
This work was supported by Basic Science Research Program through the NRF funded by the Ministry of Education, Science and Technology (2010-0009064).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lim, E.J., Heo, J. & Kim, YH. Tunicamycin promotes apoptosis in leukemia cells through ROS generation and downregulation of survivin expression. Apoptosis 20, 1087–1098 (2015). https://doi.org/10.1007/s10495-015-1135-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1135-z