Abstract
Convergent evolution is one of the main drivers of traits and phenotypes in animals and plants. Here, we investigated the minimum number of independent colonisations of marine and freshwater habitats in derived oribatid mites (Brachypylina), a mainly terrestrial taxon. Furthermore, we investigated whether the reproductive mode (sexual vs. thelytokous) is associated with the habitat type (marine, freshwater) where the animals live. We hypothesized that continuous resource availability in freshwater systems fosters asexual reproduction. We used 18S rDNA sequences to construct a molecular phylogeny of oribatid mites from terrestrial, marine and freshwater habitats. The results indicate that aquatic life in oribatid mites evolved at least 3×: once in Limnozetoidea (including only freshwater taxa) and at least twice in Ameronothroidea. In Ameronothroidea the taxon Ameronothridae n. gen. (nr. Aquanothrus) colonized fresh water independently from Selenoribatidae and Fortuyniidae (mainly marine Ameronothroidea). Reproductive mode was associated neither with marine nor with freshwater life; rather, in both habitats sexual and parthenogenetic taxa occur. However, the reproductive mode was related to the stability of the habitat. Species that live underwater permanently tend to be parthenogenetic whereas taxa whose life cycle is often interrupted by flooding, such as marine oribatid mites, or by desiccation, e.g., freshwater-living Ameronothridae n. gen. (nr. Aquanothrus) (Ameronothroidea) species, are mainly sexual, indicating that continuous access to resources indeed favours parthenogenetic reproduction. Findings of our study therefore suggest that parthenogenetic reproduction is not selected for by disturbances but by unlimited access to resources.

Similar content being viewed by others
References
Ashworth L, Aguilar R, Martén-Rodríguez S, Lopezaraiza-Mikel M, Avila-Sakar G, Rosas-Guerrero V, Quesada M (2015) Pollination syndromes: a global pattern of convergent evolution driven by the most effective pollinator. In: Pontarotti P (ed) Evolutionary biology: biodiversification from genotype to phenotype. Springer International, pp 203–224
Bast J, Schaefer I, Schwander T, Maraun M, Scheu S, Kraaijeveld K (2016) No accumulation of transposable elements in asexual arthropods. Mol Biol Evol 33:697–706
Behan-Pelletier VM (1989) Limnozetes (Acari: Oribatida: Limnozetidae) of north-eastern North America. Can Entomol 121:453–506
Behan-Pelletier VM (1997) The semi-aquatic genus Tegeocranellus (Acari: Oribatida: Ameronothroidea) of North and Central America. Can Entomol 129:537–577
Behan-Pelletier VM (2015) Review of sexual dimorphism in brachypyline oribatid mites. Acarologia 55:127–146
Behan-Pelletier VM, Bissett B (1994) Oribatida of Canadian peatlands. Mem Entomol Soc Canada 126:73–88
Behan-Pelletier VM, Eamer B (2007) Aquatic Oribatida: adaptations, constraints, distribution and ecology. In: Morales-Malacara JB, Behan-Pelletier VM, Ueckermann E, Pérez TM, Estrada-Venegas EG, Badii M (eds) Acarology XI: proceedings of the international congress. Instituto de Biología, Facultad de Ciencias, Universidad Nacional Autónoma de México, Sociedad Latinoamericana de Acarología, México, pp 71–82
Bell G (1982) The masterpiece of nature: the evolution and genetics of sexuality. University of California Press, Berkeley
Conway Morris S (2003) Life’s solution: inevitable humans in a lonely universe. Cambridge University Press, Cambridge
Covarrubias R, Mellado I (1998) Oribatidos de Chile (Acarina: Oribatida). II. Especies asociadas a plantas acuaticas. Acta Entomol Chil 22:37–44
Dabert M, Witalinski W, Kazmierski A, Olszanowski Z, Dabert J (2010) Molecular phylogeny of acariform mites (Acari, Arachnida): strong conflict between phylogenetic signal and long-branch attraction artifacts. Mol Phylogen Evol 56:222–241
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
Debortoli N, Xiang L, Eyres I, Fontaneto D, Hespeels B, Tang CQ, Flot JF, Van Doninck K (2016) Genetic exchange among bdelloid rotifers is more likely due to horizontal gene transfer than to meiotic sex. Curr Biol 26:723–732
Domes K, Norton RA, Maraun M, Scheu S (2007) Re-evolution of sex in oribatid mites breaks Dollo’s law. Proc Nat Acad Sci USA 104:7139–7144
Emery NJ, Clayton NS (2004) The mentality of crows: convergent evolution of intelligence in corvids and apes. Science 306:1903–1907
Emlen DJ (2008) Animal weapons: the evolution of battle. Ann Rev Ecol Evol Syst 39:387–413
Fain A, Lambrechts L (1987) Observations on the acarofauna of fish aquariums. I. Mites associated with Discus fish. Bull Ann Soc R Belge Entomol 123:87–102
Freeman S, Herron JC (2007) Evolutionary analysis. Prentice Hall, New Jersey
Gouy M, Guindon S, Gascuel O (2010) SeaView version 4: a multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol Biol Evol 27:221–224
Graham TB, Norton RA (1997) Uncharismatic microfauna of the Colorado Plateau: notes on distribution and ecology of an undescribed, pothole-dwelling Ameronothroid (Acari: Ameronothridae) mite. In: Learning from the land: GSENM science symposium proceedings, Cedar City, UT, pp 477–483
Grandjean F (1951) Comparaison du genre Limnozetes au genre Hydrozetes (Oribates). Bull Mus Nat Hist Natur Paris 23:200–207
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98
Hamilton WD (1980) Sex versus non-sex versus parasite. Oikos 35:282–290
Hughes HC (1999) Sensory exotica. A world beyond human experince. The MIT Press, Cambridge
Jaenicke J (1978) A hypothesis to account for the maintenance of sex within populations. Evol Theory 3:191–194
Judson OP, Normark BB (1996) Ancient asexual scandals. Trends Ecol Evol 11:41–46
Keightley PD, Otto SP (2006) Interference among deleterious mutations favours sex and recombination in finite populations. Nature 443:89–92
Krantz GW, Behan-Pelletier VM, Cook DR, Harvey MS, Keirans JE, Lindquist EE, Norton RA, OConner BM, Smith IM, Walter DE (2009) A manual of acarology, 3rd edn. Texas Tech University Press, Lubbock
Kreipe V, Corral-Hernández E, Scheu S, Schaefer I, Maraun M (2015) Phylogeny and species delineation in European species of the genus Steganacarus (Acari, Oribatida) using mitochondrial and nuclear markers. Exp Appl Acarol 66:173–186
Lehmitz R, Maraun M (2016) Small-scale spatial heterogeneity of stable isotopes signatures (δ15N, δ13C) in Sphagnum sp. transfers to all trophic levels in oribatid mites. Soil Biol Biochem 100:242–251
Maraun M, Scheu S (2000) The structure of oribatid mite communities (Acari, Oribatida): patterns, mechanisms and implications for future research. Ecography 23:374–383
Maraun M, Heethoff M, Scheu S, Norton RA, Weigmann G, Thomas RH (2003) Radiation in sexual and parthenogenetic oribatid mites (Oribatida, Acari) as indicated by genetic divergence of closely related species. Exp Appl Acarol 29:265–277
Martens K, Rossetti G, Butlin RK, Schön I (2005) Molecular and morphological phylogeny of the ancient asexual Darwinulidae (Crustacea, Ostracoda). Hydrobiologia 538:153–165
Maynard Smith J (1986) Contemplating life without sex. Nature 324:300–301
McGhee G (2011) Convergent evolution: limited forms most beautiful. The MIT Press, Cambridge
Muller HJ (1964) The relation of recombination to mutational advance. Mut Res 1:2–9
Norton RA, Behan-Pelletier VM (2009) Suborder Oribatida. In: Krantz GW, Walter DE (eds) A manual of acarology. Texas Tech University Press, Lubbock, pp 430–564
Norton RA, Palmer SC (1991) The distribution, mechanisms and evolutionary significance of parthenogenesis in oribatid mites. In: Murphy PW, Schuster R (eds) The acari: reproduction, development and life-history strategies. Chapman and Hall, London, pp 107–136
Norton RA, Kethley JB, Johnston DE, O’Connor BM (1993) Phylogenetic perspectives on genetic systems and reproductive modes of mites. In: Wrensch OL, Ebbert MA (eds) Evolution and diversity of sex ratio in insects and mites. Chapman and Hall, New York, pp 8–99
Norton RA, Graham TB, Alberti G (1997) A rotifer-eating ameronothroid (Acari: Ameronothridae) mite from ephemeral pools on the Colorado Plateau. In: Mitchell R, Horn DJ, Needham GR, Welbourn WC (eds) Acarology IX, proceedings IXth international congress on acarology, Ohio, Columbus pp 539–542
Pachl P, Domes K, Schulz G, Norton RA, Scheu S, Schaefer I, Maraun M (2012) Convergent evolution of defense mechanisms in oribatid mites (Acari, Oribatida) shows no “ghosts of predation past”. Mol Phylogen Evol 65:412–420
Pachl P, Lindl AC, Krause A, Scheu S, Schaefer I, Maraun M (2016) The tropics as ancient cradle of oribatid mite diversity. Acarologia (in press)
Pfingstl T (2013) Resistance to fresh and salt water in intertidal mites (Acari: Oribatida): implications for ecology and hydrochorous dispersal. Exp Appl Acarol 61:87–96
Pfingstl T (2015) Morphological diversity in Selenoribates (Acari, Oribatida): new species from coasts of the Red Sea and the Indo-Pacific. Int J Acarol 41:356–370
Pfingstl T (2016) Marine associated mites (Acari: Oribatida: Ameronothroidea) of the African continent: biogeography, new species and distribution records. Syst Appl Acarol 21:320–334
Pfingstl T, Jagersbacher-Baumann J (2016) Indications of parthenogenesis and morphological differentiation in Hawaiian intertidal Fortuynia (Acari, Oribatida) populations. Zool Anz 260:11–24
Poulin R, Randhawa HS (2013) Evolution of parasitism along convergent lines: from ecology to genomics. Parasitology 11:1–10
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Schaefer I, Norton RA, Scheu S, Maraun M (2010) Arthropod colonization of land—linking molecules and fossils in oribatid mites (Acari, Oribatida). Mol Phylogen Evol 57:113–121
Schäffer S, Koblmüller S, Pfingstl T, Sturmbauer C, Krisper G (2010) Ancestral state reconstruction reveals multiple independent evolution of diagnostic morphological characters in the “Higher Oribatida” (Acari), conflicting with current classification schemes. BMC Evol Biol 10:246
Schatz H, Behan-Pelletier VM (2008) Global diversity of oribatids (Oribatida: Acari: Arachnida). Hydrobiologia 595:323–328
Scheu S, Drossel B (2007) Sexual reproduction prevails in a world of structured resources in short supply. Proc R Soc Lond B 274:1225–1231
Schön I, Rossetti G, Martens K (2009) Darwinulid ostracods: ancient asexual scandals or scandalous gossip? In: Schön I, Martens K, Dijk P (eds) Lost sex: The evolutionary biology of parthenogenesis. Springer, Netherlands, Dordrecht, pp 217–240
Schön I, Pinto RL, Halse S, Smith AJ, Martens K, Birky CW (2012) Cryptic species in putative ancient asexual darwinulids (Crustacea, Ostracoda). PLoS ONE 7:e39844
Schubart HOR (1975) Morphologische Grundlagen für die Klärung der Verwandtschaftsverhältnisse innerhalb der Milbenfamilie Ameronothridae (Acari). Zoologica 123:23–91
Schuster R (1979) Soil mites in the marine environment. In: Rodriguez JG (ed) Recent advances in acarology, vol 1. Academic Press, New York, pp 593–602
Schwander T (2016) Evolution: the end of an ancient asexual scandal. Curr Biol 26:233–235
Seniczak S, Seniczak A (2009) Hydrozetes longisetosus sp. nov. (Acari: Oribatida: Hydrozetidae)—the most primitive European species of Hydrozetes from Poland. J Nat Hist 43:951–971
Seniczak A, Solhøy T, Seniczak S, De La Riva-Caballero A (2010) Species composition and abundance of the oribatid fauna (Acari, Oribatida) at two lakes in the Fløyen area, Bergen, Norway. Biol Lett 47:11–19
Seniczak A, Seniczak S, Maraun M, Graczyk R, Mistrzak M (2016) Oribatid mite species numbers increase, densities decline and parthenogenetic species suffer during bog degradation. Exp Appl Acarol 68:409–428
Smith RJ, Kamiya T, Horne DJ (2006) Living males of the “ancient asexual” Darwinulidae (Ostracoda: Crustacea). Proc R Soc Lond B 273:1569–1578
Song Y, Drossel B, Scheu S (2011) Tangled Bank dismissed too early. Oikos 120:1601–1607
Speed MP, Arbuckle K (2016) Quantification provides a conceptual basis for convergent evolution. Biol Rev. doi:10.1111/brv.12257
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Subías LS (2004) Listado sistematico, sinonimico y biogeografico de los acaros oribatidos (Acariformes, Oribatida) del Mundo (1758–2002). Graellsia, 60:3–305 (updated 2016). http://escalera.bio.ucm.es/usuarios/bba/cont/docs/RO_1.pdf
Taberly G (1988) Recherches sur la parthénogenèse thélytoque de deux espèces d’Acariens Oribates: Trhypochthonius tectorum (Berlese) et Platynothrus peltifer (Koch). IV. Observations sur les males ataviques. Acarologia 29:95–107
Walter DE, Proctor HC (2013) Mites: ecology, evolution and behaviour, 2nd edn. Springer, Dordrecht
Weigmann G, Deichsel R (2006) 4. Limnic Oribatida - Süsswasserfauna von Mitteleuropa. In: Chelicerata: Araneae, Acari I, Süßwasserfauna von Mitteleuropa 7. pp 89–112
Weigmann G, Schulte G (1977) The evolution of the family Ameronothridae (Acari, Oribatei). I. Morphological aspects. Acarologia 19:161–166
Williams GC (1975) Sex and evolution. Princeton University Press, Princeton
Winemiller KO, Fitzgerald DB, Bower LM, Pianka ER (2015) Functional traits, convergent evolution, and periodic tables of niches. Ecol Lett 18:737–751
Zakon HH (2002) Convergent evolution on the molecular level. Brain Behav Evol 59:250–261
Acknowledgments
We thank Roy A. Norton for sending species of Ameronothridae n. gen. (nr. Aquanothrus), Hydrozetes and Limnozetes and for commenting on earlier versions of the paper.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
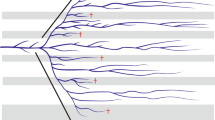
Phylogenetic tree of terrestrial and aquatic oribatid mites (Brachypylina) with three outgroup taxa (Desmonomata) based on the ribosomal 18S rDNA reconstructed with the Maximum Likelihood algorithm RAxML. Numbers on nodes represent bootstrap values; values below 70 are not shown. Marine taxa are marked in red, freshwater taxa in blue and terrestrial taxa in black. (PPTX 60 kb)
Rights and permissions
About this article
Cite this article
Krause, A., Pachl, P., Schulz, G. et al. Convergent evolution of aquatic life by sexual and parthenogenetic oribatid mites. Exp Appl Acarol 70, 439–453 (2016). https://doi.org/10.1007/s10493-016-0089-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-016-0089-3