Abstract
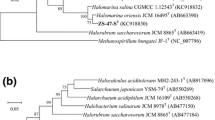
A halophilic archaeal strain, SA3T, was isolated from sediment of a solar saltern in Gomso Bay, Republic of Korea. Cells of strain SA3T were observed to be coccoid-shaped, to lyse in distilled water, Gram stain-negative and to form red-pigmented colonies. Strain SA3T was found to require at least 18 % (w/v) NaCl for growth. Optimal growth was observed at 24 % (w/v) NaCl and 6 % (w/v) MgCl2. The optimum pH and temperature for growth were determined to be pH 7.0 and 40 °C, respectively, while the strain was found to grow within pH and temperature ranges of 5.5–8.0 and 20–45 °C, respectively. The polar lipids were determined to consist of phosphatidylglycerol, phosphatidylglycerol phosphate methyl ester, unidentified phosphoglycolipids and unidentified phospholipids. Phylogenetic analysis based on 16S rRNA gene sequences showed that strain SA3T was most closely related to the members of the genus Natronomonas, Natronomonas moolapensis JCM 14361T (95.2 %) and Natronomonas pharaonis JCM 8858T (95.1 %). The genomic DNA G+C content (61.8 mol%) determined for strain SA3T was slightly lower than those of N. moolapensis JCM 14361T (63.4 mol%) and N. pharaonis JCM 8858T (64.3 mol%). DNA–DNA hybridization values between N. moolapensis JCM 14361T and N. pharaonis JCM 8858T and strain SA3T were <20 %. Based on phenotypic, chemotaxonomic and phylogenetic properties, we describe a new species of the genus Natronomonas, represented by strain SA3T (=JCM 17867T = KCTC 4088T), for which we propose the name Natronomonas gomsonensis sp. nov.


Similar content being viewed by others
References
Antón J, Oren A, Benlloch S, Rodríguez-Valera F, Amann R, Rosselló-Mora R (2002) Salinibacter ruber gen. nov., sp. nov., a novel, extremely halophilic member of the Bacteria from saltern crystallizer ponds. Int J Syst Evol Microbiol 52(2):485–491
Burns DG, Janssen PH, Itoh T, Minegishi H, Usami R, Kamekura M, Dyall-Smith ML (2010) Natronomonas moolapensis sp. nov., non-alkaliphilic isolates recovered from a solar saltern crystallizer pond, and emended description of the genus Natronomonas. Int J Syst Evol Microbiol 60(5):1173–1176
Cui HL, Lin ZY, Dong Y, Zhou PJ, Liu SJ (2007) Halorubrum litoreum sp. nov., an extremely halophilic archaeon from a solar saltern. Int J Syst Evol Microbiol 57(10):2204–2206
DeLong EF (1992) Archaea in coastal marine environments. Proc Natl Acad Sci USA 89(12):5685–5689
Dittmer JC, Lester RL (1964) A simple, specific spray for the detection of phospholipids on thin-layer chromatograms. J Lipid Res 15:126–127
Dussault HP (1955) An improved technique for staining red halophilic bacteria. J Bacteriol 70(4):484–485
Enache M, Itoh T, Fukushima T, Usami R, Dumitru L, Kamekura M (2007) Phylogenetic relationships within the family Halobacteriaceae inferred from rpoB’ gene and protein sequences. Int J Syst Evol Microbiol 57:2289–2295
Ezaki T, Hashimoto Y, Yabuuchi E (1989) Fluorometric deoxyribonucleic acid–deoxyribonucleic acid hybridization in microdilution wells as an alternative to membrane filter hybridization in which radioisotopes are used to determine genetic relatedness among bacterial strains. Int J Syst Bacteriol 39:224–229
Falb M, Pfeiffer F, Palm P, Rodewald K, Hickmann V, Tittor J, Oesterhelt D (2005) Living with two extremes: conclusions from the genome sequence of Natronomonas pharaonis. Genome Res 15(10):1336–1343
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17(6):368–376
Gibbons NE (1974) Family V. Halobacteriaceae fam. nov. In: Buchanan RE, Gibbons NE (eds) Bergey’s manual of meterminative macteriology. Williams & Wilkins, Baltimore, pp 269–273
Gonzalez JM, Saiz-Jimenez C (2002) A fluorimetric method for the estimation of G+C mol% content in microorganisms by thermal denaturation temperature. Environ Microbiol 4:770–773
Gonzalez C, Gutierrez C, Ramirez C (1978) Halobacterium vallismortis sp. nov. An amylolytic and carbohydrate-metabolizing, extremely halophilic bacterium. Can J Microbiol 24(6):710–715
Gonzalez O, Oberwinkler T, Mansueto L, Pfeiffer F, Mendoza E, Zimmer R, Oesterhelt D (2010) Characterization of growth and metabolism of the haloalkaliphile Natronomonas pharaonis. PLoS Comput Biol 6(6):e1000799
Grant WD, Larsen H (1989) Group 111. Extremely halophilic archaeobacteria, Order Halobacteriales ord. nov. In: Staley JT, Bryant MP, Pfennig N, Holt JG (eds) Bergey’s manual of systematic bacteriology. Williams & Wilkins, Baltimore, pp 2216–2233
Gutiérrez C, González C (1972) Method for simultaneous detection of proteinase and esterase activities in extremely halophilic bacteria. Appl Microbiol 24(3):516
Gutiérrez M, Castillo A, Kamekura M, Ventosa A (2008) Haloterrigena salina sp. nov., an extremely halophilic archaeon isolated from a salt lake. Int J Syst Evol Microbiol 58(12):2880–2884
Holding A, Collee J (1971) Routine biochemical tests. Methods Microbiol 6A:1–32
Kamekura M, Dyall-Smith ML, Upasani V, Ventosa A, Kates M (1997) Diversity of alkaliphilic halobacteria: proposals for transfer of Natronobacterium vacuolatum, Natronobacterium magadii, and Natronobacterium pharaonis to Halorubrum, Natrialba, and Natronomonas gen. nov., respectively, as Halorubrum vacuolatum comb. nov., Natrialba magadii comb. nov., and Natronomonas pharaonis comb. nov., respectively. Int J Syst Evol Microbiol 47(3):853–857
Kim O-S, Cho Y-J, Lee K, Yoon S-H, Kim M, Na H, Park S-C, Jeon YS, Lee J-H, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62(3):716–721
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University, Cambridge
Lane D (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, Chichester, pp 115–175
McDade JJ, Weaver RH (1959) Rapid methods for the detection of gelatin hydrolysis. J Bacteriol 77(1):60–64
Nei M, Kumar S, Takahashi K (1998) The optimization principle in phylogenetic analysis tends to give incorrect topologies when the number of nucleotides or amino acids used is small. Proc Natl Acad Sci USA 95:12390–12397
Oren A (2012) Taxonomy of the family Halobacteriaceae: a paradigm for changing concepts in prokaryote systematics. Int J Syst Evol Microbiol 62:263–271
Oren A, Ventosa A, Grant WD (1997) Proposed minimal standards for description of new taxa in the order Halobacteriales. Int J Syst Evol Microbiol 47(1):233–238
Oren A, Duker S, Ritter S (2006) The polar lipid composition of Walsby’s square bacterium. FEMS Microbiol Lett 138:135–140
Oren A, Arahal DR, Ventosa A (2009) Emended descriptions of genera of the family Halobacteriaceae. Int J Syst Evol Microbiol 59:637–642
Ovreås L, Forney L, Daae FL, Torsvik V (1997) Distribution of bacterioplankton in meromictic Lake Saelenvannet, as determined by denaturing gradient gel electrophoresis of PCR-amplified gene fragments coding for 16S rRNA. Appl Environ Microbiol 63(9):3367–3373
Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, Glöckner FO (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35(21):7188–7196
Ross H, Grant W (1985) Nucleic acid studies on halophilic archaebacteria. J Gen Microbiol 131(1):165–173
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Bio Evol 4:406–425
Smibert RM, Krieg NR (1994) Phenotypic characterization. In: Gerhardt P, Murray RGE, Wood WA, Krieg NR (eds) Methods for general and molecular bacteriology, vol 1325. American society for Microbiology, Washington, pp 607–654
Soliman GSH, Trüper HG (1982) Halobacterium pharaonis sp. nov., a new, extremely haloalkaliphilic archaebacterium with low magnesium requirement. Zbl Bakt Hyg I Abt Orig C 3 3(10):318–329
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Tittsler RP, Sandholzer LA (1936) The use of semi-solid agar for the detection of bacterial motility. J Bacteriol 31:575–580
Torreblanca M, Rodriguez-Valera F, Juez G, Ventosa A, Kamekura M, Kates M (1986) Classification of non-alkaliphilic halobacteria based on numerical taxonomy and polar lipid composition, and description of Haloarcula gen. nov. and Haloferax gen. nov. Syst Appl Microbiol 8(1):89–99
Wayne R, Nash W, O’Brien S (1987) Chromosomal evolution of the Canidae. II. Divergence from the primitive carnivore karyotype. Cytogenet Cell Genet 44:134–141
Xin H, Itoh T, Zhou P, Suzuki K, Kamekura M, Nakase T (2000) Natrinema versiforme sp. nov., an extremely halophilic archaeon from Aibi salt lake, Xinjiang. China. Int J Syst Evol Microbiol 50:1297–1303
Acknowledgments
This study was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korea government (MEST) (No. NRF-2013R1A2A2A05006754), the Korea Polar Research Institute (PP12010) and the “Marine and Extreme Genome Research Center Program” of the Ministry of Ministry of Oceans and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tae-Yoon Kim and So-Jeong Kim have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, TY., Kim, SJ., Park, SJ. et al. Natronomonas gomsonensis sp. nov., isolated from a solar saltern. Antonie van Leeuwenhoek 104, 627–635 (2013). https://doi.org/10.1007/s10482-013-9970-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-013-9970-9