Abstract
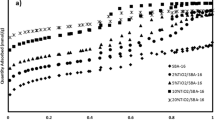
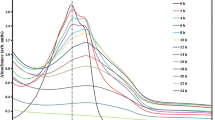
Photocatalyst of TiO2 coated on silica beads was prepared, and the adsorption isotherms and kinetics of photocatalytic decomposition of acetone and acetaldehyde on the photocatalyst were studied at room temperature (ca. 298 K). Adsorption isotherms of both acetone and acetaldehyde conformed to the Langmuir isotherm. FTIR spectra of the gas phase showed that these volatile organic compounds were mainly oxidized to CO2 and H2O. The time courses of concentration change due to photocatalytic decomposition under UV-irradiation were well represented by a proposed model by using the values of Langmuir parameters and the kinetic parameters determined.
Similar content being viewed by others
References
Bouzaza, A and A. Laplanche, J. Photochemistry and Photobiology A:Chemistry, 150, 207–212 (2002).
Daniel M. Blake, NREL’TP-;510–31319 (2001).
El-Maazawi et al., J. of Catalysis, 191, 138 –146 (2000).
Jian-Hua Xu and F. Shiraishi, J. Chem. Technol. Biotechnol., 74, 1096–1100 (1999).
Obuchi et al., Chem. Eng. Sci., 54, 1525–1530 (1999).
Sang Rum Kim and Sung Chang Hong, Applied Catalysis B:Environmental, 35, 305–315 (2002).
Sauer, M.L. and F.D., Ollis, J. of Catalysis, 158, 570–582 (1996).
Sopyan, etal. J. Photochemistry and Photobiology A:Chemistry, 98, 79–86 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tomida, T., Okada, N., Katoh, M. et al. Adsorption and Photocatalytic Decomposition of Volatile Organic Compounds on Photocatalyst of TiO2—Silica Beads. Adsorption 11 (Suppl 1), 865–869 (2005). https://doi.org/10.1007/s10450-005-6037-x
Issue Date:
DOI: https://doi.org/10.1007/s10450-005-6037-x