Abstract
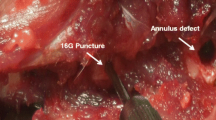
Insufficient nutrient supply has been suggested to be one of the etiologies for intervertebral disc (IVD) degeneration. We are investigating nutrient transport into the IVD as a potential treatment strategy for disc degeneration. Most cellular activities in the IVD (e.g., cell proliferation and extracellular matrix production) are mainly driven by adenosine-5′-triphosphate (ATP) which is the main energy currency. The objective of this study was to investigate the effect of increased mass transfer on ATP production in the IVD by the implantation of polyurethane (PU) mass transfer devices. In this study, the porcine functional spine units were used and divided into intact, device and surgical groups. For the device and surgical groups, two puncture holes were created bilaterally at the dorsal side of the annulus fibrosus (AF) region and the PU mass transfer devices were only implanted into the holes in the device group. Surgical groups were observed for the effects of placing the holes through the AF only. After 7 days of culture, the surgical group exhibited a significant reduction in the compressive stiffness and disc height compared to the intact and device groups, whereas no significant differences were found in compressive stiffness, disc height and cell viability between the intact and device groups. ATP, lactate and the proteoglycan contents in the device group were significantly higher than the intact group. These results indicated that the implantation of the PU mass transfer device can promote the nutrient transport and enhance energy production without compromising mechanical and cellular functions in the disc. These results also suggested that compromise to the AF has a negative impact on the IVD and must be addressed when treatment strategies are considered. The results of this study will help guide the development of potential strategies for disc degeneration.






Similar content being viewed by others
References
Acosta, F. L., L. Metz, H. D. Adkisson, J. Liu, E. Carruthers-Liebenberg, C. Milliman, M. Maloney, and J. C. Lotz. Porcine intervertebral disc repair using allogeneic juvenile articular chondrocytes or mesenchymal stem cells. Tissue Eng. Pt. A 17(23–24):3045–3055, 2011.
Adams, M. A., and P. J. Roughley. What is intervertebral disc degeneration, and what causes it? Spine 31(18):2151–2161, 2006.
An, H. S., P. A. Anderson, V. M. Haughton, J. C. Iatridis, J. D. Kang, J. C. Lotz, R. N. Natarajan, T. R. Oegema, Jr, P. Roughley, L. A. Setton, J. P. Urban, T. Videman, G. B. Andersson, and J. N. Weinstein. Introduction: disc degeneration: summary. Spine (Phila Pa 1976) 29(23):2677–2678, 2004.
Andersson, G. B. J. Epidemiological features of chronic low-back pain. Lancet 354(9178):581–585, 1999.
Bibby, S. R., D. A. Jones, R. M. Ripley, and J. P. Urban. Metabolism of the intervertebral disc: effects of low levels of oxygen, glucose, and pH on rates of energy metabolism of bovine nucleus pulposus cells. Spine (Phila Pa 1976) 30(5):487–496, 2005.
Bibby, S. R. S., D. A. Jones, R. M. Ripley, and J. P. G. Urban. Metabolism of the intervertebral disc: effects of low levels of oxygen, glucose, and pH on rates of energy metabolism of bovine nucleus pulposus cells. Spine 30(5):487–496, 2005.
Boubriak, O. A., N. Watson, S. S. Sivan, N. Stubbens, and J. P. Urban. Factors regulating viable cell density in the intervertebral disc: blood supply in relation to disc height. J. Anat. 222(3):341–348, 2013.
Bron, J. L., A. J. van der Veen, M. N. Helder, B. J. van Royen, T. H. Smit, S. T. E. G. Amsterdam, and R. I. Move. Biomechanical and in vivo evaluation of experimental closure devices of the annulus fibrosus designed for a goat nucleus replacement model. Eur. Spine J. 19(8):1347–1355, 2010.
Burnstock, G. The past, present and future of purine nucleotides as signalling molecules. Neuropharmacology 36(9):1127–1139, 1997.
Chida, J., K. Yamane, T. Takei, and H. Kido. An efficient extraction method for quantitation of adenosine triphosphate in mammalian tissues and cells. Anal. Chim. Acta 727:8–12, 2012.
Croucher, L. J., A. Crawford, P. V. Hatton, R. G. G. Russell, and D. J. Buttle. Extracellular ATP and UTP stimulate cartilage proteoglycan and collagen accumulation in bovine articular chondrocyte pellet cultures. Bba-Mol. Basis Dis. 1502(2):297–306, 2000.
Drazin, D., J. Rosner, P. Avalos, and F. Acosta. Stem cell therapy for degenerative disc disease. Adv. Orthop. 2012:961052, 2012.
Elliott, D. M., C. S. Yerramalli, J. C. Beckstein, J. I. Boxberger, W. Johannessen, and E. J. Vresilovic. The effect of relative needle diameter in puncture and sham injection animal models of degeneration. Spine (Phila Pa 1976) 33(6):588–596, 2008.
Feng, G. J., X. F. Zhao, H. Liu, H. N. Zhang, X. J. Chen, R. Shi, X. Liu, X. D. Zhao, W. L. Zhang, and B. Y. Wang. Transplantation of mesenchymal stem cells and nucleus pulposus cells in a degenerative disc model in rabbits: a comparison of 2 cell types as potential candidates for disc regeneration laboratory investigation. J. Neurosurg.Spine 14(3):322–329, 2011.
Fernando, H. N., J. Czamanski, T. Y. Yuan, W. Gu, A. Salahadin, and C. Y. Huang. Mechanical loading affects the energy metabolism of intervertebral disc cells. J. Orthop. Res. 29(11):1634–1641, 2011.
Gawri, R., F. Mwale, J. Ouellet, P. J. Roughley, T. Steffen, J. Antoniou, and L. Haglund. Development of an organ culture system for Long-Term survival of the intact human intervertebral disc. Spine 36(22):1835–1842, 2011.
Gilson, A., M. Dreger, and J. P. G. Urban. Differential expression level of cytokeratin 8 in cells of the bovine nucleus pulposus complicates the search for specific intervertebral disc cell markers. Arthritis Res. Ther. 12(1):R24, 2010.
Gonzales, S., B. Rodriguez, C. Barrera, and C. Y. C. Huang. Measurement of ATP-induced membrane potential changes in IVD cells. Cell. Mol. Bioeng. 7(4):598–606, 2014.
Gonzales, S., C. Wang, H. Levene, H. S. Cheung, and C. Y. C. Huang. ATP promotes extracellular matrix biosynthesis of intervertebral disc cells. Cell Tissue Res. 359(2):635–642, 2015.
Grunhagen, T., A. Shirazi-Adl, J. C. Fairbank, and J. P. Urban. Intervertebral disk nutrition: a review of factors influencing concentrations of nutrients and metabolites. Orthop. Clin. North Am. 42(4):465–477, 2011.
Grunhagen, T., G. Wilde, D. M. Soukane, S. A. Shirazi-Adl, and J. P. Urban. Nutrient supply and intervertebral disc metabolism. J. Bone Joint Surg. Am. 88(Suppl 2):30–35, 2006.
Gu, W. Y., and H. Yao. Effects of hydration and fixed charge density on fluid transport in charged hydrated soft tissues. Ann. Biomed. Eng. 31(10):1162–1170, 2003.
Guehring, T., G. Wilde, M. Sumner, T. Grunhagen, G. B. Karney, U. K. Tirlapur, and J. P. G. Urban. Notochordal intervertebral disc cells sensitivity to nutrient deprivation. Arthritis Rheum. 60(4):1026–1034, 2009.
Henriksson, H. B., T. Svanvik, M. Jonsson, M. Hagman, M. Horn, A. Lindahl, and H. Brisby. Transplantation of human mesenchymal stems cells into intervertebral discs in a xenogeneic porcine model. Spine 34(2):141–148, 2009.
Hirschberg, C. B., P. W. Robbins, and C. Abeijon. Transporters of nucleotide sugars, ATP, and nucleotide sulfate in the endoplasmic reticulum and Golgi apparatus. Annu. Rev. Biochem. 67:49–69, 1998.
Horner, H. A., and J. P. G. Urban. 2001 Volvo Award winner in basic science studies: effect of nutrient supply on the viability of cells from the nucleus pulposus of the intervertebral disc. Spine 26(23):2543–2549, 2001.
Hsieh, A. H., D. Hwang, D. A. Ryan, A. K. Freeman, and H. Kim. Degenerative anular changes induced by puncture are associated with insufficiency of disc biomechanical function. Spine 34(10):998–1005, 2009.
Huang, C. Y. C., M. A. Deitzer, and H. S. Cheung. Effects of fibrinolytic inhibitors on chondrogenesis of bone-marrow derived mesenchymal stem cells in fibrin gels. Biomech. Model. Mechan. 6(1–2):5–11, 2007.
Huang, C. Y., and W. Y. Gu. Effects of mechanical compression on metabolism and distribution of oxygen and lactate in intervertebral disc. J. Biomech. 41(6):1184–1196, 2008.
Huang, Y. C., J. P. G. Urban, and K. D. K. Luk. Opinion intervertebral disc regeneration: do nutrients lead the way? Nat. Rev. Rheumatol. 10(9):561–566, 2014.
Jackson, A., and W. Gu. Transport Properties of Cartilaginous Tissues. Curr. Rheumatol. Rev. 5(1):40, 2009.
Jackson, A. R., C. Y. Huang, and W. Y. Gu. Effect of endplate calcification and mechanical deformation on the distribution of glucose in intervertebral disc: a 3D finite element study. Comput. Method Biomec. 14(2):195–204, 2011.
Johnson, K., A. Jung, A. Murphy, A. Andreyev, J. Dykens, and R. Terkeltaub. Mitochondrial oxidative phosphorylation is a downstream regulator of nitric oxide effects on chondrocyte matrix synthesis and mineralization. Arthritis Rheum. 43(7):1560–1570, 2000.
Johnson, S., and P. Rabinovitch. Ex vivo imaging of excised tissue using vital dyes and confocal microscopy. Curr. Protoc. Cytom. 9:9–39, 2012.
Johnson, K., C. I. Svensson, D. Van Etten, S. S. Ghosh, A. N. Murphy, H. C. Powell, and R. Terkeltaub. Mediation of spontaneous knee osteoarthritis by progressive chondrocyte ATP depletion in Hartley guinea pigs. Arthritis Rheum. 50(4):1216–1225, 2004.
Katz, J. N. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J. Bone Joint Surg. Am. 88a:21–24, 2006.
Korecki, C. L., J. J. Costi, and J. C. Iatridis. Needle puncture injury affects intervertebral disc mechanics and biology in an organ culture model. Spine 33(3):235–241, 2008.
Levene, H. B. Analysis of Tyrosine-Derived Novel Synthetic Polymer Scaffold Devices for Guided Tissue Regeneration, Rutgers University and the University of Medicine and Dentistry of New Jersey, New Brunswick, NJ, 1999, p. 167.
Likhitpanichkul, M., M. Dreischarf, S. Illien-Junger, B. A. Walter, T. Nukaga, R. G. Long, D. Sakai, A. C. Hecht, and J. C. Iatridis. Fibrin-genipin adhesive hydrogel for annulus fibrosus repair: performance evaluation with large animal organ culture, situ biomechanics, and in vivo degradation tests. Eur. Cells Mater. 28:25–38, 2014.
Long, R. G., S. G. Rotman, W. W. Hom, D. J. Assael, D. W. Grijpma, and J. C. Iatridis. In vitro and biomechanical screening of polyethylene glycol and poly(trimethylene carbonate) block copolymers for annulus fibrosus repair, J. Tissue Eng. Regen. Med. 2016.
Long, R. G., O. M. Torre, W. W. Hom, D. J. Assael, and J. C. Iatridis. Design requirements for annulus fibrosus repair: review of forces, displacements, and material properties of the intervertebral disk and a summary of candidate hydrogels for repair. J. Biomech. Eng. 138(2):021007, 2016.
MacLean, J. J., C. R. Lee, S. Grad, K. Ito, M. Alini, and J. C. Iatridis. Effects of immobilization and dynamic compression on intervertebral disc cell gene expression in vivo. Spine 28(10):973–981, 2003.
Martin, J. A., A. Martini, A. Molinari, W. Morgan, W. Ramalingam, J. A. Buckwalter, and T. O. McKinley. Mitochondrial electron transport and glycolysis are coupled in articular cartilage. Osteoarthr. Cartilage 20(4):323–329, 2012.
Masuda, K., T. R. Oegema, and H. S. An. Growth factors and treatment of intervertebral disc degeneration. Spine 29(23):2757–2769, 2004.
Michalek, A. J., M. R. Buckley, L. J. Bonassar, I. Cohen, and J. C. Iatridis. The effects of needle puncture injury on microscale shear strain in the intervertebral disc annulus fibrosus. Spine J. 10(12):1098–1105, 2010.
Neidlinger-Wilke, C., A. Mietsch, C. Rinkler, H. J. Wilke, A. Ignatius, and J. Urban. Interactions of environmental conditions and mechanical loads have influence on matrix turnover by nucleus pulposus cells. J. Orthop. Res. 30(1):112–121, 2012.
Osada, R., H. Ohshima, H. Ishihara, K. Yudoh, K. Sakai, H. Matsui, and H. Tsuji. Autocrine/paracrine mechanism of insulin-like growth factor-1 secretion, and the effect of insulin-like growth factor-1 on proteoglycan synthesis in bovine intervertebral discs. J. Orthopaed. Res. 14(5):690–699, 1996.
Pattappa, G., Z. Li, M. Peroglio, N. Wismer, M. Alini, and S. Grad. Diversity of intervertebral disc cells: phenotype and function. J. Anat. 221(6):480–496, 2012.
Pratsinis, H., and D. Kletsas. PDGF, bFGF and IGF-I stimulate the proliferation of intervertebral disc cells in vitro via the activation of the ERK and Akt signaling pathways. Eur. Spine J. 16(11):1858–1866, 2007.
Prydz, K., and K. T. Dalen. Synthesis and sorting of proteoglycans—commentary. J. Cell. Sci. 113(2):193–205, 2000.
Schmidt, H., A. Shirazi-Adl, F. Galbusera, and H. J. Wilke. Response analysis of the lumbar spine during regular daily activities–a finite element analysis. J. Biomech. 43(10):1849–1856, 2010.
Smith, L. J., N. L. Nerurkar, K. S. Choi, B. D. Harfe, and D. M. Elliott. Degeneration and regeneration of the intervertebral disc: lessons from development. Dis. Model Mech. 4(1):31–41, 2011.
Sobajima, S., J. F. Kompel, J. S. Kim, C. J. Wallach, D. D. Robertson, M. T. Vogt, J. D. Kang, and L. G. Gilbertson. A slowly progressive and reproducible animal model of intervertebral disc degeneration characterized by MRI, X-ray, and histology. Spine 30(1):15–24, 2005.
Urban, J. P. G., and S. Roberts. Degeneration of the intervertebral disc. Arthritis Res. Ther. 5(3):120–130, 2003.
Urban, J. P. G., S. Smith, and J. C. T. Fairbank. Nutrition of the intervertebral disc. Spine 29(23):2700–2709, 2004.
Waldman, S. D., J. Usprech, L. E. Flynn, and A. A. Khan. Harnessing the purinergic receptor pathway to develop functional engineered cartilage constructs. Osteoarthr. Cartilage 18(6):864–872, 2010.
Walter, B. A., C. L. Korecki, D. Purmessur, P. J. Roughley, A. J. Michalek, and J. C. Iatridis. Complex loading affects intervertebral disc mechanics and biology. Osteoarthr. Cartilage 19(8):1011–1018, 2011.
Wang, Y. F., C. M. Barrera, E. A. Dauer, W. Gu, F. Andreopoulos, and C. C. Huang. Systematic characterization of porosity and mass transport and mechanical properties of porous polyurethane scaffolds. J. Mech. Behav. Biomed. Mater. 65:657–664, 2016.
Wang, C., S. Gonzales, H. Levene, W. Y. Gu, and C. Y. C. Huang. Energy metabolism of intervertebral disc under mechanical loading. J. Orthopaed. Res. 31(11):1733–1738, 2013.
Wang, D. L., S. D. Jiang, and L. Y. Dai. Biologic response of the intervertebral disc to static and dynamic compression in vitro. Spine (Phila Pa 1976) 32(23):2521–2528, 2007.
Wu, Y. R., S. Cisewski, B. L. Sachs, and H. Yao. Effect of cartilage endplate on cell based disc regeneration: a finite element analysis. Mol. Cell. Biomech. 10(2):159–182, 2013.
Yao, H., M. A. Justiz, D. Flagler, and W. Y. Gu. Effects of swelling pressure and hydraulic permeability on dynamic compressive behavior of lumbar annulus fibrosus. Ann. Biomed. Eng. 30(10):1234–1241, 2002.
Zhou, S., Z. Cui, and J. P. Urban. Nutrient gradients in engineered cartilage: metabolic kinetics measurement and mass transfer modeling. Biotechnol. Bioeng. 101(2):408–421, 2008.
Acknowledgments
This study was supported by the Grant AR066240 from the NIH and the University of Miami Provost’s Research Award.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Peter E. McHugh oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Wang, YF., Levene, H.B., Gu, W. et al. Enhancement of Energy Production of the Intervertebral Disc by the Implantation of Polyurethane Mass Transfer Devices. Ann Biomed Eng 45, 2098–2108 (2017). https://doi.org/10.1007/s10439-017-1867-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-017-1867-8