Abstract
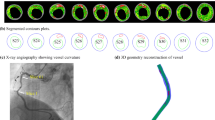
Plaque rupture has been considered to be the result of its structural failure. The aim of this study is to suggest a possible link between higher stresses and rupture sites observed from in vivo magnetic resonance imaging (MRI) of transient ischemic attack (TIA) patients, by using stress analysis methods. Three patients, who had recently suffered a TIA, underwent in vivo multi-spectral MR imaging. Based on plaque geometries reconstructed from the post-rupture status, six pre-rupture plaque models were generated for each patient dataset with different reconstructions of rupture sites to bridge the gap of fibrous cap from original MRI images. Stress analysis by fluid structure interaction simulation was performed on the models, followed by analysis of local stress concentration distribution and plaque rupture sites. Furthermore, the sensitivity of stress analysis to the pre-rupture plaque geometry reconstruction was examined. Local stress concentrations were found to be located at the plaque rupture sites for the three subjects studied. In the total of 18 models created, the locations of the stress concentration regions were similar in 17 models in which rupture sites were always associated with high stresses. The local stress concentration region moved from circumferential center to the shoulder region (slightly away from the rupture site) for a case with a thick fibrous cap. Plaque wall stress level in the rupture locations was found to be much higher than the value in non-rupture locations. The good correlation between local stress concentrations and plaque rupture sites, and generally higher plaque wall stress level in rupture locations in the subjects studied could provide indirect evidence for the extreme stress-induced plaque rupture hypothesis. Local stress concentration in the plaque region could be one of the factors contributing to plaque rupture.






Similar content being viewed by others
References
Casscells, W., M. Naghavi, and J. T. Willerson. Vulnerable atherosclerotic plaque: a multifocal disease. Circulation 107:2072–2075, 2003.
Cheng, G. C., H. M. Loree, R. D. Kamm, M. C. Fishbein, and R. T. Lee. Distribution of circumferential stress in ruptured and stable atherosclerotic lesions. A structural analysis with histopathological correction. Circulation 87:1179–1187, 1993.
Creane, A., E. Maher, S. Sultan, N. Hynes, D. Kelly, and C. Lally. Finite element modeling of diseased carotid bifurcations generated from in vivo computerized tomographic angiography. Comput. Biol. Med. 40:419–429, 2010.
Gao, H., and Q. Long. Effects of varied lipid core volume and fibrous cap thickness on stress distribution in carotid arterial plaques. J. Biomech. 41:3053–3059, 2008.
Gao, H., Q. Long, M. Graves, J. H. Gillard, and Z. Y. Li. Carotid arterial plaque stress analysis using fluid-structure interactive simulation based on in vivo magnetic resonance images of four patients. J. Biomech. 42:1416–1423, 2009.
Gao, H., Q. Long, U. Sadat, M. J. Graves, J. H. Gillard, and Z. Y. Li. Stress analysis of carotid atheroma in a TIA patient using MRI-based fluid-structure interaction method. Br. J. Radiol. 82:S46–S54, 2009.
Lee, R. T., H. M. Loree, G. C. Cheng, E. H. Lieberman, N. Jaramillo, and F. J. Schoen. Computational structural analysis based on intravascular ultrasound imaging before in vitro angioplasty: prediction of plaque rupture locations. J. Am. Coll. Cardiol. 21:777–782, 1993.
Li, Z. Y., S. P. S. Howarth, T. Tang, M. J. Graves, J. U-King-Im, R. A. Trivedi, P. J. Kirkpatrick, and J. H. Gillard. Structural analysis and magnetic resonance imaging predict plaque vulnerability: a study comparing symptomatic and asymptomatic individuals. J. Vasc. Surg. 45:768–775, 2007.
Li, Z. Y., S. Howarth, R. A. Trivedi, J. M. U-King-Im, M. J. Graves, A. Brown, L. Wang, and J. H. Gillard. Stress analysis of carotid plaque rupture based on in vivo high resolution MRI. J. Biomech. 39:2611–2622, 2006.
Libby, P., P. M. Ridker, and A. Maseri. Inflammation and atherosclerosis. Circulation 105(9):1135–1143, 2002.
Loree, H. M., R. D. Kamm, R. G. Stringfellow, and R. T. Lee. Effects of fibrous cap thickness on peak circumferential stress in model atherosclerotic vessels. Circ. Res. 71:850–858, 1992.
Maher, E., A. Creane, S. Sultan, N. Hynes, C. Lally, and D. Kelly. Tensile and compressive properties of fresh human carotid atherosclerotic plaques. J. Biomech. 42:2760–2767, 2009.
Masson, I., P. Boutouyrie, S. Laurent, J. D. Humphrey, and M. Zidi. Characterization of arterial wall mechanical behaviour and stresses from human clinical data. J. Biomech. 41:2618–2627, 2008.
Naghavi, M., P. Libby, E. Falk, S. Casscells, and S. Litovsky. From vulnerable plaque to vulnerable patient: a call for new definition and risk assessment strategies: part I. Circulation 108:1664–1672, 2003.
Ohayon, J., P. Teppaz, G. Finet, and G. Rioufol. In vivo prediction of human coronary plaque rupture locations using intravascular ultrasound and the finite element method. Coron Artery Dis 12:655–663, 2001.
Redgrave, J. N., P. Gallagher, J. K. Lovett, and P. M. Rothwell. Critical cap thickness and rupture in symptomatic carotid plaques. Stroke 39:1722–1729, 2008.
Richardson, P. D. Biomechanics of plaque rupture: progress, problems, and new frontiers. Ann. Biomed. Eng. 30:524–536, 2002.
Rosamond, W., K. Flegal, G. Friday, K. Furie, A. Go, K. Greenlund, N. Haase, M. Ho, V. Howard, B. Kissela, S. Kittner, D. Lloyd-Jones, M. McDermott, J. Meigs, C. Moy, G. Nichol, C. J. O’Donnel, V. Roger, J. Rumsfel, P. Sorlie, J. Steinberger, T. Thom, S. Wasserthiel-Smoller, and Y. Hong. Heart disease and stroke statistics—2007 update. Circulation 115:69–171, 2007.
Sadat, U., Z. Teng, V. E. Young, S. R. Walsh, Z. Y. Li, M. J. Graves, K. Varty, and J. H. Gillard. Association between biomechanical structural stresses of atherosclerotic carotid plaques and subsequent ischaemic cerebrovascular events—a longitudinal in vivo magnetic resonance imaging-based finite element study. Eur. J. Vasc. Endovasc. Surg. 40:485–491, 2010.
Shah, P. K. Mechanisms of plaque vulnerability and rupture. J. Am. Coll. Cardiol. 41(suppl S):15–22, 2003.
Tang, D., Z. Teng, G. Canton, C. Yang, M. Ferguson, X. Huang, J. Zheng, P. K. Woodard, and C. Yuan. Sites of rupture in human atherosclerotic carotid plaques are associated with high structural stresses: an in vivo MRI-based 3D fluid–structure interaction study. Stroke 40:3258–3263, 2009.
Tang, D., C. Yang, J. Zheng, K. P. Woodard, J. E. Saffitz, G. A. Sicard, T. K. Pilgram, and C. Yuan. Quantifying effects of plaque structure and material properties on stress distributions in human atherosclerotic plaques using 3D FSI models. J. Biomech. Eng. 127:1185–1194, 2005.
Tang, D., C. Yang, J. Zheng, P. K. Woodard, G. A. Sicard, J. E. Saffitz, and C. Yuan. 3D MRI-based multi-component FSI models for atherosclerotic plaques a 3-D FSI model. Ann. Biomed. Eng. 32:947–960, 2004.
Trivedi, R. A., J. M. U-King-Im, M. J. Graves, J. Horsley, M. Goddard, P. J. Kirkpatrick, and J. H. Gillard. MRI-derived measurements of fibrous cap and lipid-core thickness: the potential for identifying vulnerable carotid plaques in vivo. Neuroradiology 46:738–743, 2004.
Yuan, C., S. X. Zhang, N. L. Polissar, D. Echelard, G. Ortiz, W. D. Joseph, E. Ellington, M. S. Ferguson, and T. S. Hatsukami. Identification of fibrous cap rupture with magnetic resonance imaging is highly associated with recent transient ischemic attack or stroke. Circulation 105:181–185, 2002.
Acknowledgments
This project was supported by the British Heart Foundation (FS/06/048). Dr Umar Sadat is supported by a Medical Research Council (UK) and Royal College of Surgeons of England Joint Clinical Research Training Fellowship.
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Peter E. McHugh oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Gao, H., Long, Q., Das, S.K. et al. Stress Analysis of Carotid Atheroma in Transient Ischemic Attack Patients: Evidence for Extreme Stress-Induced Plaque Rupture. Ann Biomed Eng 39, 2203–2212 (2011). https://doi.org/10.1007/s10439-011-0314-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-011-0314-5