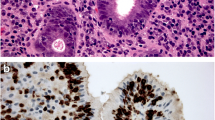
Abstract
Background
Although both eosinophilic esophagitis (EoE) and Barrett’s esophagus (BE) are considered to be associated with T helper (Th) 2-mediated immune responses, the association between EoE and BE is unclear. We investigated the clinical relationship between EoE and BE.
Methods
We conducted a single-center retrospective observational study. The study included 95 patients with EoE and randomly selected age- and sex-matched controls who underwent esophagogastroduodenoscopy during a medical health check-up at Osaka City University in a ratio of 1:2 for comparison. We compared the clinical characteristics and the prevalence rate of BE, reflux esophagitis (RE), hiatal hernia, and atrophic gastritis between EoE patients and controls by univariate analysis. Furthermore, we performed multivariate logistic regression analysis to investigate the association of these factors with EoE.
Results
On univariate analysis, the prevalence rate of BE was significantly lower in patients with EoE than in controls (2.1% vs. 13.2%; p = 0.00528). In contrast, the prevalence rate of RE was higher in EoE patients than in controls, but it was not statistically significant (absence and Grades A, B, and C: 74.7%, 18.9%, 5.3%, and 1.1% vs. 83.7%, 12.6%, 3.7%, and 0%; p = 0.193, respectively). Multivariate analysis showed that BE was negatively associated with EoE (odds ratio: 0.132; 95% confidence interval: 0.0302–0.573; p = 0.00686).
Conclusions
BE is negatively associated with EoE in Japanese subjects. The mechanism behind the inverse relationship between EoE and BE should be examined.


Similar content being viewed by others
References
Abe Y, Nomura E, Sato T, Ueno Y. The diagnosis of eosinophilic esophagitis. Gastroenterol Endosc. 2014;56:3378–93.
Liacouras CA, Furuta GT, Hirano I, et al. Eosinophilic esophagitis: updated consensus recommendations for children and adults. J Allergy Clin Immunol. 2011;128:3–20.
Safroneeva E, Coslovsky M, Kuehni CE, et al. Eosinophilic oesophagitis: relationship of quality of life with clinical, endoscopic and histological activity. Aliment Pharmacol Ther. 2015;42:1000–10.
Fujiwara Y, Sugawa T, Tanaka F, et al. A multicenter study on the prevalence of eosinophilic esophagitis and PPI-responsive esophageal eosinophilic infiltration. Intern Med. 2012;51:3235–9.
Fujishiro H, Amano Y, Kushiyama Y, et al. Eosinophilic esophagitis investigated by upper gastrointestinal endoscopy in Japanese patients. J Gastroenterol. 2011;46:1142–4.
Stewart MJ, Shaffer E, Urbanski SJ, et al. The association between celiac disease and eosinophilic esophagitis in children and adults. BMC Gastroenterol. 2013;13:96.
Sherrill JD, Gao PS, Stucke EM, et al. Variants of thymic stromal lymphopoietin and its receptor associate with eosinophilic esophagitis. J Allergy Clin Immunol. 2010;126:160–5.
Fitzgerald RC, Onwuegbusi BA, Bajaj-Elliott M, et al. Diversity in the oesophageal phenotypic response to gastro-oesophageal reflux: immunological determinants. Gut. 2002;50:451–9.
Kohata Y, Fujiwara Y, Machida H, et al. Role of Th-2 cytokines in the development of Barrett’s esophagus in rats. J Gastroenterol. 2011;46:883–93.
Lucendo AJ, Molina-Infante J, Arias Á, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United Eur Gastroenterol J. 2017;5:335–58.
Japan Esophageal Society. Japanese classification of esophageal cancer, 11th Edition: part II and III. Esophagus. 2017;14:37–65.
Matsuzaki J, Suzuki H, Asakura K, et al. Etiological difference between ultrashort- and short-segment Barrett’s esophagus. J Gastroenterol. 2011;46:332–8.
Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80.
Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969;1:87–97.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Francalanci P, De Angelis P, Minnei F, et al. Eosinophilic esophagitis and Barrett’s esophagus: an occasional association or an overlap disease? Esophageal ‘double trouble’ in two children. Digestion. 2008;77:16–9.
Mukkada V, Atkins D, Furuta GT. Uncertain association of Barrett’s esophagus with eosinophilic esophagitis. Clin Gastroenterol Hepatol. 2008;6:832–3.
Saboorian MH, Genta RM, Marcus PB, et al. Inverse association of esophageal eosinophilia and Barrett esophagus. J Clin Gastroenterol. 2012;46:752–7.
Kim HP, Vance RB, Shaheen NJ, et al. The prevalence and diagnostic utility of endoscopic features of eosinophilic esophagitis: a meta-analysis. Clin Gastroenterol Hepatol. 2012;10:988–96.
Manabe N, Haruma K, Imamura H, et al. Does short-segment columnar-lined esophagus elongate during a mean follow-up period of 5.7 years? Dig Endosc. 2011;23:166–72.
Sun D, Wang X, Gai Z, et al. Bile acids but not acidic acids induce Barrett’s esophagus. Int J Clin Exp Pathol. 2015;8:1384–92.
Rothenberg ME. Molecular, genetic, and cellular bases for treating eosinophilic esophagitis. Gastroenterology. 2015;148:1143–57.
Palles C, Chegwidden L, Li X, et al. Polymorphisms near TBX5 and GDF7 are associated with increased risk for Barrett’s esophagus. Gastroenterology. 2015;148:367–78.
Wang KK, Sampliner RE. Updated guidelines 2008 for the diagnosis, surveillance and therapy of Barrett’s esophagus. Am J Gastroenterol. 2008;103:788–97.
Amano Y, Ishimura N, Furuta K, et al. Which landmark results in a more consistent diagnosis of Barrett’s esophagus, the gastric folds or the palisade vessels? Gastrointest Endosc. 2006;64:206–11.
Watari J, Hori K, Toyoshima F, et al. Association between obesity and Barrett’s esophagus in a Japanese population: a hospital-based, cross-sectional study. BMC Gastroenterol. 2013;13:143.
Acknowledgements
ST, FT, and YF were involved in study conception and design. ST and FT had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of data analysis. KO and SF acquired the data. ST and FT analyzed and interpreted the data. Drafting of the manuscript was performed by ST and FT. Critical revision of the manuscript for important intellectual content was conducted by ST, FT, KO, SH, YN, NK, KT, HY, TT, SF, TW, and YF. All authors have approved the final draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Statement
This study was conducted in accordance with the Declaration of Helsinki and its amendments. The study protocol was approved by the Ethics Committee of the Osaka City University Graduate School of Medicine (No. 3951).
Conflict of interest
The authors declare no conflict of interests for this article.
Rights and permissions
About this article
Cite this article
Takashima, S., Tanaka, F., Otani, K. et al. Barrett’s esophagus is negatively associated with eosinophilic esophagitis in Japanese subjects. Esophagus 16, 168–173 (2019). https://doi.org/10.1007/s10388-018-0648-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-018-0648-2