Abstract
Objective
We evaluated the sensitivity of pulsed Arterial Spin Labelling (pASL) for the detection of changes in regional cerebral blood perfusion (CBP) during and after intra-venous (i.v.) infusion of an opioid agonist (fentanyl) and an opioid antagonist (naloxone).
Materials and methods
Twenty-three subjects were scanned four times, receiving i.v. infusion of fentanyl, naloxone, placebo and a second fentanyl administration, in four separate scanning sessions in randomised order. End-tidal CO2, respiration rate and heart rate were recorded continuously throughout each scan. pASL time series were collected using single shot EPI for 15 min (including 5 min of baseline prior to infusion).
Results
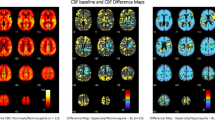
Significant increases in CBP were detected during and after administration of fentanyl, (when compared to placebo and naloxone), in most areas of high concentration of mu-opioid receptors (thalamus, lingual gyrus, para-hippocampal gyrus, and insula); near-significant increases were also observed in the insula. No increases in perfusion were observed during or after naloxone infusion. No correlation was found between regional rCBF changes and end-tidal CO2, respiration rate or heart rate. Good reliability was found between the first and second fentanyl sessions but the regions of high reliability did not overlap completely with those of highest perfusion change.
Conclusion
pASL is a suitable method for examining rapid, dynamic effects of opioid administration on brain physiology.









Similar content being viewed by others
References
Kornetsky C (1995) Action of opioid drugs on the brain reward system. NIDA Res Monogr 147:33–52
Martin-Soelch C, Chevalley AF, Kunig G, Missimer J, Magyar S, Mino A, Schultz W, Leenders KL (2001) Changes in reward-induced brain activation in opiate addicts. Eur J Neurosci 14:1360–1368
Ozaki S, Narita M, Narita M, Iino M, Sugina J, Matsumura Y, Suzuki T (2002) Suppression of the morphine induced rewarding effect in the rat with neuropathic pain: implication of the reduction in mu-opioid receptor function in the ventral tegmental area. J Neurochem 82:1192–1198
Koob GF, Sanna PP, Bloom FE (1998) Neuroscience of addiction. Neuron 21:467–476
Borras MC, Becerra L, Ploghaus A, Gostic JM, DaSilva A, Gonzalez RG, Borsook D (2004) FMRI measurement of CNS responses to naloxone infusion and subsequent mild noxious thermal stimuli in healthy volunteers. J Neurophysiol 91:2723–2733
Schad CA, Justice JB Jr, Holtzman SG (2002) Endogenous opioids in dopaminergic cell body regions modulate amphetamine-induced increases in extracellular dopamine levels in the terminal regions. J Pharmacol Exp Ther 300:932–938
Bullmore E, Suckling J, Zelaya F, Long C, Honey G, Reed L, Routledge C, Ng V, Fletcher P, Brown J, Williams SCR (2003) Practice and difficulty evoke anatomically and pharmacologically dissociable activation dynamics. Cereb Cortex 13:144–154
Honey G, Suckling J, Zelaya FO, Williams SCR, Routledge C, Brown J, Jackson S, Fletcher P, Bullmore E (2003) Dopaminergic drug effects on physiological connectivity in a human cortico-striato-thalamic system. Brain 126(8):1767–1781
Mulderink TA, Gitelman DR, Mesulam MM, Parrish TB (2002) On the use of caffeine as a contrast booster for BOLD fMRI studies. Neuroimage 15(1):37–44
Perthen JE, Lansing AE, Liau J, Liu TT, Buxton RB (2008) Caffeine-induced uncoupling of cerebral blood flow and oxygen metabolism: a calibrated BOLD fMRI study. Neuroimage 40:237–247
Williams DS, Detre JA, Leigh JS, Koretsky AP (1992) Magnetic resonance imaging of perfusion using spin inversion of arterial water. Proc Natl Acad Sci USA 89:212–216
Villiger JW, Ray LJ, Taylor KM (1983) Characteristics of [3] fentanyl binding to the opiate receptor. Neuropharmacology 22:447–452
Aguirre GK, Detre JE, Zarahan E, Alsop DC (2002) Experimental design and the relative sensitivity of BOLD and perfusion fMRI. Neuroimage 15:488–500
Frost JJ (2001) PET imaging of the opioid receptor: the early years. Nucl Med Biol 28:509–513
Schlaepfer TE, Strain EC, Greenber BD, Preston KL, Lancaster E, Bigelow GE, Barta PE, Pearlson GD (1998) Site of opioid action in the human brain: Mu and Kappa agonists’ subjective and cerebral blood flow effects. Am J Psychiatry 155(4):470–473
Firestone LL, Gyulai F, Mintun M, Adler LJ, Urso K, Winter PM (1996) Human brain activity response to fentanyl imaged by positron emission tomography. Anesth Analg 82:1247–1251
Adler LJ, Gyulai FE, Diehl DJ, Mintun MA, Winter PM, Firestone LL (1997) Regional brain activity changes associated with fentanyl analgesia elucidated by positron emission tomography. Anesth Analg 84:120–126
Kurata J, Gyulai FE, Firestone LL (1999) Use of positron emission tomography to measure brain activity responses to fentanyl analgesia. Curr Rev Pain 3(5):359–366
Detre J, Alsop DC (1999) Perfusion magnetic resonance imaging with continuous arterial spin labelling: methods and clinical applications in the central nervous system. Eur J Radiol 30:115–124
Detre J, Wang J (2002) Technical aspects and utility of fMRI using BOLD and ASL. Clin Neurophysiol 113:621–634
O’Gorman R, Mehta M, Asherson P, Zelaya F, Brookes KJ, Toone BK, Alsop DC, Williams SCR (2008) Increased cerebral perfusion in adult attention deficit hyperactivity disorder is normalised by stimulant treatment: a non-invasive MRI pilot study. Neuroimage 42(1):36–41
Van Hell HH, Bossong MG, Jagger G, Kristo G, van Osch MP, Zelaya FO, Kahn RS, Ramsey NF (2011) Evidence for involvement of the insula in the psychotropic effects of THC in humans: a double-blind, randomized pharmacological MRI study. Int J Neuropsychopharmacol 14:1–12
Fernandez-Seara MA, Aznarez-Sanado M, Mengual E, Irigoyen J, Heukamp F, Pastor MA (2011) Effects on resting state cerebral blood flow and functional connectivity induced by metoclopramide: a perfusion MRI study in healthy volunteers. Br J Pharmacol 163(8):1639–1652
MacIntosh BJ, Pattinson KT, Gallichan D, Ahmad I, Miller KL, Feinberg DA, Wise RG, Jezzard P (2008) Measuring the effects of remifentanil on cerebral blood flow and arterial arrival time using 3D GRASE MRI with pulsed arterial spin labelling. J Cereb Blood Flow Metab 28(8):1514–1522
Kofke WA, Blissit P, Rao H, Wang J, Addya K, Detre J (2007) Remifentail-induced cerebral blood flow effects in normal humans: dose and ApoE genotype. Anesth Analg 105(1):167–175
Baumgartner U, Buchholz HG, Bellosevic A, Magerl W, Siessmeir T, Rolke R, Hohnemann S, Piel M, Rosch F, Wester HJ, Henriksen G, Stoeter P, Bartenstein P, Treede RD, Schreckenberger M (2006) High opiate receptor binding potential in the human lateral pain system. Neuroimage 30:692–699
First MB, Spitzer RL, Gibbon M, Williams JBW (2002) Structured clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition (SCID-I/NP). Biometrics Research, New York State Psychiatric Institute, New York
Wong EC, Buxton R, Frank L (1997) Implementation of quantitative perfusion imaging techniques for functional brain mapping using pulsed arterial spin labelling. NMR Biomed 10:237–249
Zacny JP, Lichtor JL, Zaragoza JG, de Wit H (1992) Subjective and behavioural responses to intravenous fentanyl in healthy volunteers. Psychopharmacology (Berl) 107(2–3):319–326
Glascher J, Gitelman D (2008) Contrast weights in flexible factorial design with multiple groups of subjects, SPM Notes, SPM library on line library. http://www.ion.ucl.ac.uk/spm/
Worsley KJ, Andermann M, Koulis T, MacDonald D, Evans AC (1999) Detecting changes in nonisotropic images. Hum Brain Mapp 8:98–101
Hayasaka S, Phan KL, Liberzon I, Worsley KJ, Nichols TE (2004) Nonstationary cluster-size inference with random field and permutation methods. Neuroimage 22:676–687
Caceres A, Hall D, Zelaya FO, Williams SC, Mehta M (2009) Measuring fMRI reliability with the intra-class correlation coefficient. Neuroimage 45(3):758–768
Shrout PE, Fleiss JL (1979) Intra-class correlations: uses in assessing rater reliability. Psychol Bull 86(2):420–428
McGraw KO, Wong SP (1996) Forming inferences about some intraclass correlation coefficients. Psychol Methods 1(1):30–46
Martinez D, Slifstein M, Broft A, Mawlawi O, Hwang DR, Huang Y, Cooper T, Kegeles L, Zarahn E, Abi-Dargham A, Haber SN, Laruelle M (2003) Imaging human mesolimbic dopamine transmission with positron emission tomography. Part II: amphetamine-induced dopamine release in the functional subdivisions of the striatum. J Cereb Blood Flow Metab 23(3):285–300
Lappin JM, Reeves SJ, Mehta MA, Egerton A, Coulson M, Grasby PM (2009) Dopamine release in the human striatum: motor and cognitive tasks revisited. J Cereb Blood Flow Metab 29(3):554–564
Brett M, Anton J, Valabregue R, Poline J (2002) Region of interest analysis using an SPM toolbox. NeuroImage 16(2):2–6
Pfeiffer A, Pasi A, Mehraein P, Herz A (1982) Opiate receptor binding sites in human brain. Brain Res 248(1):87–96
Zubieta JK, Gorelick DA, Stauffer R, Ravert HT, Dannals RF, Frost JJ (1996) Increased mu opioid receptor binding detected by PET in cocaine-dependent men is associated with cocaine craving. Nat Med 2(11):1225–1229
Peroutka SJ, Moskowitz MA, Reinhard JF Jr, Snyder SH (1980) Neurotransmitter receptor binding in bovine cerebral microvessels. Science 208(4444):610–612
Devine JO, Armstead WM (1995) The role of nitric oxide in opioid-induced pial artery vasodilation. Brain Res 675(1–2):257–263
Velasco M, Brito F, Jimenez F, Gallegos M, Velasco AL, Velasco F (1998) Effect of fentanyl and naloxone on a thalamic induced painful response in intractable epileptic patients. Stereotact Funct Neurosurg 71(2):90–102
Stefanovic B, Warnking JM, Rylander KM, Pike GB (2006) The effect of global cerebral vasodilation on focal activation hemodynamics. Neuroimage 30(3):726–734
Franceschini MA, Radhakrishnan H, Thakur K, Wu W, Ruvinskaya S, Carp S, Boas DA (2010) The effect of different anaesthetics on neurovascular coupling. Neuroimage 51(4):1367–1377
Melichar JK, Nutt DJ, Malizia AL (2003) Naloxone displacement at opioid receptor sites measured in vivo in the human brain. Eur J Pharmacol 459:217–219
Liu TT, Wong EC (2005) A signal processing model for arterial spin labelling functional MRI. Neuroimage 24:207–215
Wise RG, Ide K, Poulin MJ, Tracey I (2004) Resting fluctuations in arterial carbon dioxide induce significant low frequency fluctuations in BOLD signal. Neuroimage 21(4):1652–1664
Young KA, Holcomb LA, Bonkale WL, Hicks PB, Yazdan U, German DC (2007) 5HTTLPR: polymorphism and enlargement of the pulvinar: unlocking the backdoor to the limbic system. Biol Psychiatry 61:813–818
Handley R, Zelaya FO, Reinders AATS, Marques TR, Mehta MA, O’Gorman R, Alsop DC, Taylor H, Johnston A, Williams SCR, McGuire P, Pariante C, Kapur S, Dazzan P (2011) Acute effects of single dose aripiprazole and haloperidol on resting cerebral blood flow (rCBF) in the human brain. Hum Brain Mapp. Accepted (in press)
Acknowledgments
The authors are grateful to Pfizer Pharmaceuticals UK for funding towards this project, to Mr Jeffrey Dalton for help with the implementation of software for the measurement of physiological variables and to Dr Eric Wong for providing a hyperbolic secant adiabatic RF pulse for use with arterial spin labelling.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. A. Mehta and L. J. Reed contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zelaya, F.O., Zois, E., Muller-Pollard, C. et al. The response to rapid infusion of fentanyl in the human brain measured using pulsed arterial spin labelling. Magn Reson Mater Phy 25, 163–175 (2012). https://doi.org/10.1007/s10334-011-0293-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-011-0293-4