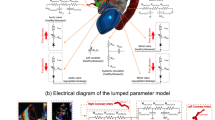
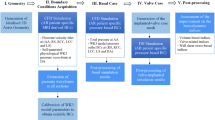
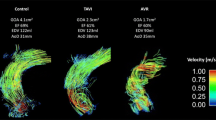
Abstract
Transcatheter aortic valve implantation (TAVI) is a micro-invasive surgery used to treat patients with aortic stenosis (AS) efficiently. However, the uneven valve expansion can cause a non-circular annulus, which is one of the main factors leading to complications after TAVI. As a preliminary work, the main purpose of this study was to evaluate the risk of adverse aortic events in patients with a non-circular aortic annulus after TAVI. This study numerically investigated the distribution of four wall shear stress (WSS)-based indicators and three helicity-based indicators in eight patient-specific aortas with different annulus including circular, type I elliptical and type II elliptical shapes. Both elliptical annulus features can significantly enhance the intensity of the helicity (h2) in the ascending aorta (p < 0.001). However, for the type I elliptical annulus, the spiral flow structure was changed into low-velocity and disturbed flow pattern close to the inner side of the aortic arch. For the type II elliptical annulus, the spiral flow remained but became skewed in distribution. The elliptical annulus feature could increase the general level WSS-based indicators, especially in the ascending aorta. However, due to the disturbance of spiral flow or second helical flow in ascending aortas, areas with low TAWSS accompanied by high oscillatory shear index (OSI) and cross flow index (CFI) were observed in all the ascending aortas with non-circular annulus. The elliptical annulus feature can change the hemodynamic environment in the aortic arch, especially in the ascending aorta. Although both elliptical annulus features enhanced the strength of helicity, the uniform distribution of the helical flow was disturbed, especially in the ascending aorta, indicating the potential risk of adverse aortic events may increase. Therefore, for the patients without paravalvular leak but elliptical annulus shape after TAVI treatment, surgeons may be needed to consider further dilatation to make the non-circular annulus become circular.










Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article/supplementary material, and further inquiries can be directed to the corresponding author/s.
References
Alloui Z, Vasseur P (2015) Natural convection of Carrea_Yasuda non-Newtonian fluids in a vertical cavity heated from the sides. Int J Heat Mass Transf 84:912–924
Andersson M, Lantz J, Ebbers T, Karlsson M (2017) Multidirectional WSS disturbances in stenotic turbulent flows: a pre- and post-intervention study in an aortic coarctation. J Biomech 51:8–16. https://doi.org/10.1016/j.jbiomech.2016.11.064
Armour CH, Menichini C, Milinis K et al (2020) Location of reentry tears affects false lumen thrombosis in aortic dissection following TEVAR. J Endovasc Ther 27:396–404. https://doi.org/10.1177/1526602820917962
Bissell MM, Hess AT, Biasiolli L et al (2013) Aortic dilation in bicuspid aortic valve disease: flow pattern is a major contributor and differs with valve fusion type. Circ Cardiovasc Imaging 6:499–507. https://doi.org/10.1161/CIRCIMAGING.113.000528
Bonfanti M, Balabani S, Greenwood JP et al (2017) Computational tools for clinical support: a multi-scale compliant model for haemodynamic simulations in an aortic dissection based on multi-modal imaging data. J R Soc Interface. https://doi.org/10.1098/rsif.2017.0632
Bonfanti M, Franzetti G, Homer-Vanniasinkam S et al (2020) A combined in vivo, in vitro, in silico approach for patient-specific haemodynamic studies of aortic dissection. Ann Biomed Eng 48:2950–2964. https://doi.org/10.1007/s10439-020-02603-z
Cheng Z, Riga C, Chan J et al (2013) Initial findings and potential applicability of computational simulation of the aorta in acute type B dissection. J Vasc Surg 57:35S-43S. https://doi.org/10.1016/j.jvs.2012.07.061
Cheng Z, Juli C, Wood NB et al (2014) Predicting flow in aortic dissection: comparison of computational model with PC-MRI velocity measurements. Med Eng Phys 36:1176–1184. https://doi.org/10.1016/j.medengphy.2014.07.006
Chi Q, He Y, Luan Y et al (2017) Numerical analysis of wall shear stress in ascending aorta before tearing in type A aortic dissection. Comput Biol Med 89:236–247. https://doi.org/10.1016/j.compbiomed.2017.07.029
Chiu J-J, Chien S (2011) Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol Rev 91:327–387. https://doi.org/10.1152/physrev.00047.2009
Cho YI, Kensey KR (1991) Effects of the non-Newtonian viscosity of blood on flows in a diseased arterial vessel. Part 1: steady flows. Biorheology 28:241–262. https://doi.org/10.3233/BIR-1991-283-415
Coppola G, Caro C (2008) Oxygen mass transfer in a model three-dimensional artery. J R Soc Interface 5:1067–1075. https://doi.org/10.1098/rsif.2007.1338
Crick SJ, Sheppard MN, Ho SY et al (1998) Anatomy of the pig heart: comparisons with normal human cardiac structure. J Anat 193:105–119. https://doi.org/10.1046/j.1469-7580.1998.19310105.x
Davies PF (2009) Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat Clin Pract Cardiovasc Med 6:16–26. https://doi.org/10.1038/ncpcardio1397
De Nisco G, Kok AM, Chiastra C et al (2019) The atheroprotective nature of helical flow in coronary arteries. Ann Biomed Eng 47:425–438. https://doi.org/10.1007/s10439-018-02169-x
Delgado V, Ng ACT, van de Veire NR et al (2010) Transcatheter aortic valve implantation: role of multi-detector row computed tomography to evaluate prosthesis positioning and deployment in relation to valve function. Eur Heart J 31:1114–1123. https://doi.org/10.1093/eurheartj/ehq018
Deng X, Marois Y, How T et al (1995) Luminal surface concentration of lipoprotein (LDL) and its effect on the wall uptake of cholesterol by canine carotid arteries. J Vasc Surg 21:135–145. https://doi.org/10.1016/s0741-5214(95)70252-0
Dolan JM, Sim FJ, Meng H, Kolega J (2012) Endothelial cells express a unique transcriptional profile under very high wall shear stress known to induce expansive arterial remodeling. Am J Physiol Cell Physiol 302:C1109–C1118. https://doi.org/10.1152/ajpcell.00369.2011
Ethier CR (2002) Computational modeling of mass transfer and links to atherosclerosis. Ann Biomed Eng 30:461–471. https://doi.org/10.1114/1.1468890
Gallo D, De Santis G, Negri F et al (2012) On the use of in vivo measured flow rates as boundary conditions for image-based hemodynamic models of the human aorta: implications for indicators of abnormal flow. Ann Biomed Eng 40:729–741
Hoogendoorn A, Kok AM, Hartman EMJ et al (2020) Multidirectional wall shear stress promotes advanced coronary plaque development: comparing five shear stress metrics. Cardiovasc Res 116:1136–1146. https://doi.org/10.1093/cvr/cvz212
Jackson SP, Nesbitt WS, Westein E (2009) Dynamics of platelet thrombus formation. J Thromb Haemost 7(Suppl 1):17–20. https://doi.org/10.1111/j.1538-7836.2009.03401.x
Kodali SK, Williams MR, Smith CR et al (2012) Two-year outcomes after transcatheter or surgical aortic-valve replacement. N Engl J Med 366:1686–1695. https://doi.org/10.1056/NEJMoa1200384
Kousera CA, Wood NB, Seed WA et al (2013) A numerical study of aortic flow stability and comparison with in vivo flow measurements. J Biomech Eng 135:11003. https://doi.org/10.1115/1.4023132
Ku DN, Giddens DP, Zarins CK, Glagov S (1985) Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arteriosclerosis 5:293
LaBarbera M (1990) Principles of design of fluid transport systems in zoology. Science 249:992–1000. https://doi.org/10.1126/science.2396104
Liu X, Fan Y, Xu XY, Deng X (2012) Nitric oxide transport in an axisymmetric stenosis. J R Soc Interface 9:2468–2478
Marrocco-trischitta MM, Sturla F (2021) Blood flow helical pattern in type III arch configuration as a potential risk factor for type B aortic dissection. Eur J Cardio-Thoracic Surg 00:1–8. https://doi.org/10.1093/ejcts/ezab307
Menichini C, Cheng Z, Gibbs RGJ, Xu XY (2018) A computational model for false lumen thrombosis in type B aortic dissection following thoracic endovascular repair. J Biomech 66:36–43. https://doi.org/10.1016/j.jbiomech.2017.10.029
Mohamied Y, Sherwin SJ, Weinberg PD (2017) Understanding the fluid mechanics behind transverse wall shear stress. J Biomech 50:102–109. https://doi.org/10.1016/j.jbiomech.2016.11.035
Morbiducci U, Ponzini MR, Redaelli A (2007) Helical flow as fluid dynamic signature for atherogenesis risk in aortocoronary bypass. A Numeric Study J Biomech 40:519–534
Nesbitt WS, Westein E, Tovar-Lopez FJ et al (2009) A shear gradient-dependent platelet aggregation mechanism drives thrombus formation. Nat Med 15:665–673. https://doi.org/10.1038/nm.1955
Pasta S, Cannata S, Gentile G et al (2020) Simulation study of transcatheter heart valve implantation in patients with stenotic bicuspid aortic valve. Med Biol Eng Comput 58:815–829. https://doi.org/10.1007/s11517-020-02138-4
Prashantha B, Anish S (2019) Computational investigations on the hemodynamic performance of a new swirl generator in bifurcated arteries. Comput Methods Biomech Biomed Engin 22:364–375. https://doi.org/10.1080/10255842.2018.1556974
Scharfschwerdt M, Meyer-Saraei R, Schmidtke C, Sievers HH (2014) Hemodynamics of the Edwards Sapien XT transcatheter heart valve in noncircular aortic annuli. J Thorac Cardiovasc Surg 148:126–132. https://doi.org/10.1016/j.jtcvs.2013.07.057
Stonebridge PABC (1991) Spiral laminar flow in arteries? Lancet 338(8779):1360–1361
Tarbell JM (2003) Mass transport in arteries and the localization of atherosclerosis. Annu Rev Biomed Eng 5:79–118. https://doi.org/10.1146/annurev.bioeng.5.040202.121529
Tasso P, Raptis A, Matsagkas M et al (2018) Abdominal aortic aneurysm endovascular repair: profiling post-implantation morphometry and hemodynamics with image-based computational fluid dynamics. J Biomech Eng Doi 10(1115/1):4040337
Unbehaun A, Pasic M, Dreysse S et al (2012) Transapical aortic valve implantation: incidence and predictors of paravalvular leakage and transvalvular regurgitation in a series of 358 patients. J Am Coll Cardiol 59:211–221. https://doi.org/10.1016/j.jacc.2011.10.857
Wen J, Ding G, Jiang W et al (2014) Numerical simulation of compliant artery bypass grafts using fluid-structure interaction framework. ASAIO J 60:533–540. https://doi.org/10.1097/MAT.0000000000000101
Wen J, Ho H, Peng L et al (2022) Effect of anastomosis angles on retrograde perfusion and hemodynamics of hybrid treatment for thoracoabdominal aortic aneurysm. Ann Vasc Surg 79:298–309. https://doi.org/10.1016/j.avsg.2021.08.009
White SJ, Hayes EM, Lehoux S et al (2011) Characterization of the differential response of endothelial cells exposed to normal and elevated laminar shear stress. J Cell Physiol 226:2841–2848. https://doi.org/10.1002/jcp.22629
Wootton DM, Ku DN (1999) Fluid mechanics of vascular systems, diseases, and thrombosis. Annu Rev Biomed Eng 1:299–329
Zegdi R, Ciobotaru V, Noghin M et al (2008) Is it reasonable to treat all calcified stenotic aortic valves with a valved stent? Results from a human anatomic study in adults. J Am Coll Cardiol 51:579–584. https://doi.org/10.1016/j.jacc.2007.10.023
Zhao ZG, Jilaihawi H, Feng Y, Chen M (2015) Transcatheter aortic valve implantation in bicuspid anatomy. Nat Rev Cardiol 12:123–128. https://doi.org/10.1038/nrcardio.2014.161
Zhu GY, Wei Y, Su YL et al (2019) Impacts of internal carotid artery revascularization on flow in anterior communicating artery aneurysm: a preliminary multiscale numerical investigation. Appl Sci. https://doi.org/10.3390/app9194143
Funding
This study was supported in part by the Applying Basic Research Project of Sichuan Province, China [No. 2022NSFSC1369], the National Natural Science Foundation of China [No. 11802253], NHC Key Laboratory of Nuclear Technology Medical Transformation (MIANYANG CENTRAL HOSPITAL) [No.2021HYX031] and Sichuan Medical Research Youth Innovation Project of China [No. Q21075].
Author information
Authors and Affiliations
Contributions
JW and SP conceived and designed the study, obtained the funding, gave the final approval of the article and took overall responsibility. JW, QG, JNC and XYL carried out analysis and interpretation. QG, JNC and XYL were involved in modeling. SP collected the data and critically reviewed the article. JW wrote the article.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wen, J., Gao, Q., Chen, J. et al. Risk evaluation of adverse aortic events in patients with non-circular aortic annulus after transcatheter aortic valve implantation: a numerical study. Biomech Model Mechanobiol 22, 1379–1394 (2023). https://doi.org/10.1007/s10237-023-01725-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-023-01725-2