Abstract
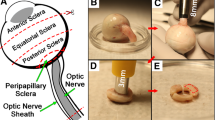
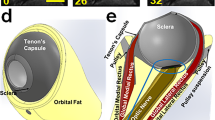
Reported mechanical properties of orbital connective tissue and fat have been too sparse to model strain–stress relationships underlying biomechanical interactions in strabismus. We performed rheological tests to develop a multi-mode upper convected Maxwell (UCM) model of these tissues under shear loading. From 20 fresh bovine orbits, 30 samples of connective tissue were taken from rectus pulley regions and 30 samples of fatty tissues from the posterior orbit. Additional samples were defatted to determine connective tissue weight proportion, which was verified histologically. Mechanical testing in shear employed a triborheometer to perform: strain sweeps at 0.5–2.0 Hz; shear stress relaxation with 1% strain; viscometry at 0.01−0.5 s−1 strain rate; and shear oscillation at 1% strain. Average connective tissue weight proportion was 98% for predominantly connective tissue and 76% for fatty tissue. Connective tissue specimens reached a long-term relaxation modulus of 668 Pa after 1,500 s, while corresponding values for fatty tissue specimens were 290 Pa and 1,100 s. Shear stress magnitude for connective tissue exceeded that of fatty tissue by five-fold. Based on these data, we developed a multi-mode UCM model with variable viscosities and time constants, and a damped hyperelastic response that accurately described measured properties of both connective and fatty tissues. Model parameters differed significantly between the two tissues. Viscoelastic properties of predominantly connective orbital tissues under shear loading differ markedly from properties of orbital fat, but both are accurately reflected using UCM models. These viscoelastic models will facilitate realistic global modeling of EOM behavior in binocular alignment and strabismus.
Article PDF
Similar content being viewed by others
References
Barbenel JC, Evan JH, Finlay JB (1973) Stress-strain-time relations for soft connective tissues, perspectives in biomedical engineering. MacMillan, London
Beatty MF (1987) Topics in finite elasticity: hyperelasticity of rubber, elastomers, and biological tissues—with examples. Appl Mech Rev 40: 1699–1734
Bellezza AJ, Rintalan CJ, Thompson HW, Downs JC, Hart RT, Burgoyne CF (2003) Deformation of the lamina cribrosa and anterior scleral canal wall in early experimental Glaucoma. Invest Ophthalmol Vis Sci 44(2): 623–637
Bilston LE, Liu Z, Phan-Thien N (1998) Linear viscoelastic properties of bovine brain tissue in shear. Biorheology 34: 377–385
Bilston LE, Liu Z, Phan-Thien N (2001) Large strain behaviour of brain tissue in shear: some new experimental data and differential constitutive model. Biorheology 38: 335–345
Bilston LE, Thibault LE (1996) The mechanical properties of the human cervical spinal cord. Ann Biomed Eng 24: 67–74
Buchtal F, Kaiser E (1951) The rheology of the cross striated muscle fiber with particular reference to isotonic conditions. Dan Biol Med 21: 328–336
Chang GL, Hung TK, Feng WW (1996) An in-vivo measurement and analysis of viscoelastic properties of the spinal cord of cats. ASME J Biomech Eng 110: 115–122
Chaudhry HR, Bukiet B, Findley T, Ritter AB (1998) Evaluation of residual stress in rabbit skin and the relevant material constants. J Theor Biol 192: 191–195
Collins CC, Carlson MR, Scott AB, Jampolsky A (1981) Extraocular muscle forces in normal human subjects. Invest Ophthalmol Vis Sci 20: 652–664
De Hoff P (1978) On the nonlinear viscoelastic behaviour of soft biological tissues. J Biomech 11: 35–40
Demer JL, Miller JM, Poukens V, Vinters HV, Glasgow BJ (1995) Evidence for fibromuscular pulleys of the recti extraocular muscles. Invest Ophthalmol Vis Sci 36(6): 1125–1136
Demer JL, Oh SY, Clark RA, Poukens V (2003) Evidence for a pulley of the inferior oblique muscle. Invest Ophthalmol Vis Sci 44: 3856–3865
Demer JL, Poukens V, Miller JM, Micevych P (1997) Innervation of extraocular pulley smooth muscle in monkeys and humans. Invest Ophthalmol Vis Sci 38(9): 1774–1785
Downs JC, Suh JKF, Kevin AT, Anthony JB, Claude FB, Richard TH (2003) Viscoelastic characterization of peripapillary sclera: material properties by quadrant in rabbit and monkey eyes. J Biomech Eng 125(1): 124–131
Downs JC, Suh JKF, Thomas KA, Bellezza AJ, Hart RT, Burgoyne CF (2005) Viscoelastic material properties of the peripapillary sclera in normal and early-glaucoma monkey eyes. Invest Ophthalmol Vis Sci 46(2): 540–546
Estes JM, McElhaney JH (1970) Response of brain tissue to compressive loading. ASME 70-BHF, 13
Farshad M, Barbezat M, Flueler P, Schmidlin F, Graber P, Niedere P (1999) Material characterization of the pig kidney in relation with the biomechanical analysis of renal trauma. J Biomech 32: 417–425
Fung YC (1967) Elasticity of soft tissue in simple elongation. Am J Physiol 213: 1532–1544
Fung YC (1972) Stress-strain-history relations of soft tissues in simple elongation, biomechanics: its foundations and objectives. Prentice-Hall, Englewood Cliffs
Fung YC (1993) Biomechanics: mechanical properties of living tissues. Springer, New York
Galford JE, McElhaney JH (1970) A visco-elastic study of scalp, brain, and dura. J Biomech 3: 211–221
Holenstein R, Niederer P, Anliker M (1980) A viscoelastic model for use in predicting arterial pulse waves. ASME J Biomech Eng 102: 302–318
Huyghe JH, Van Campen DH, Arts T, Heethaar RM (1991) The constitutive behaviour of passive heart muscle tissue: a quasi-linear viscoelastic formation. J Biomech 24: 841–849
Jenkins RB, Little RW (1974) A constitutive equation for parallel fibered elastic tissue. J Biomech 397–402
Johnson GA, Livesay GA, Woo SLY, Rajagopal KR (1996) A single integral finite strain viscoelastic model of ligaments tendons. J Biomech Eng 118: 221–226
Kono R, Poukens V, Demer JL (2002) Quantitative analysis of the structure of the human extraocular muscle pulley system. Invest Ophthalmol Vis Sci 43(9): 2923–2932
Kono R, Poukens V, Demer JL (2005) Superior oblique muscle layers in monkeys and humans. Invest Ophthalmol Vis Sci 46(8): 2790–2799
Lanir Y (1979) The rheological behavior of skin: experimental results and a structural model. Biorheology 16: 191–202
Lin HC, Kwan MKW, Woo SLY (1987) On the stress relaxation properties of the anterior cruciate ligament(ACL). Adv Bioeng 5–6
Liu Z, Bilston LE (2000) On the viscoelastic character of liver tissues: experiments and modeling of the linear behavior. Biorheology 37: 191–201
Maynewman K, McCulloch AD (1998) Homogenization modeling for the mechanics of perfused myocardium. Prog Biophys Mol Biol 69: 463–481
Mendis KK, Stalnaker RL, Roberts VL (1995) A constitutive relation for large deformation finite element modeling of brain tissue. J Biomech Eng 117: 279–285
Miller JM (1989) Functional anatomy of normal human rectus muscles. Vis Res 29: 223–240
Miller JM, Demer JL, Poukens V, Pavlovski DS, Nguyen HN, Rossi EA (2003) Extraocular connective tissue architecture. J Vis 2: 12–23
Miller JM, Robins D (1987) Extraocular muscle sideslip and orbital geometry in monkeys. Vis Res 27(3): 381–392
Miller K, Chinzei K (2002) Mechanical properties of brain tissue in tension. J Biomech 35(4): 483–490
Miller K, Chinzei K (1997) Constitutive modeling of brain tissue: experiment and theory. J Biomech Eng 30: 1115–1121
Nasseri S, Bilston LE, Phan-Thien N (2002) Viscoelastic properties of pig kidney in shear, experimental results and modeling. Rheol Acta 41: 180–192
Oh SY, Poukens V, Cohen MS, Demer JL (2001) Structure-function correlation of laminar vascularity in human rectus extraocular muscles. Invest Ophthalmol Vis Sci 42(1): 17–22
Oh SY, Poukens V, Demer JL (2001) Quantitative analysis of rectus extraocular muscle layers in monkey and humans. Invest Ophthalmol Vis Sci 42(1): 10–16
Pamidi MR, Advani SH (1978) Nonlinear constitutive equations for human brain tissue. J Biomech Eng 100: 44–48
Phan-Thien N, Nasseri S, Bilston LE (2000) Oscillatory squeezing flow of a biological material. Rheol Acta 39: 409–417
Phan-Thien N, Safari-Ardi M, Morales PA (1997) Oscillatory and simple shear flows of a flour-water dough: a constitutive model. Rheol Acta 36: 38–48
Pinto JG, Fung YC (1973) Mechanical properties of heart muscle in the passive state. J Biomech 6: 597–616
Pinto JG, Patitucci PJ (1980) Visco-elasticity of passive cardiac muscle. ASME J Biomech Eng 102: 57–61
Pradas MM, Calleja RD (1990) Nonlinear viscoelastic behavior of the flexor tendon of the human hand. J Biomech 23: 773–781
Robinson DA, O’Meara DM, Scott AB, Collins CC (1969) Mechanical components of human eye movement. J Appl Physiol 26: 548–553
Schmidlin FR, Schmid P, Kurtyke T, Iselin CE, Graber P (1996) Force transmission and stress distribution in a computer simulated model of kidney: an analysis of the injury mechanisms in renal trauma. J Trauma 40: 791–796
Schoemaker I, Hoefnagel PPW, Mastenbroek TJ, Kolff CF, Schutte S, van der Helm FCT, Picken SJ, Gerritsen AFC, Wielopolski PA, Spekreijse H, Simonsz HJ (2006) Elasticity, viscosity, and deformation of orbital fat. Invest Ophthalmol Vis Sci 47(11): 4819–4826
Shoemaker PA, Schneider D, Lee MC, Fung YC (1986) A constitutive model for two dimensional soft tissues and its application to experimental data. J Biomech 19: 695–702
Simonsz HJ (1994) Force-length recording of eye muscles during local anesthesia surgery in 32 strabismus patients. Strabismus 2: 197–218
Simonsz HJ, Harting F, De Wall BJ, Verbeeten BWJM (1985) Sideways displacement and curved path of recti eye muscles. Arch Ophthalmol (103):124–128
Treloar L (1975) The physics of rubber elasticity. Clarendon Press, Oxford
Viidik A (1966) Biomechanics and functional adaptation of tendons and joint ligaments. In: Evans FG (eds) Studies on the anatomy and function of bone and joints. Springer, New York
Weiss JA, Maker BN, Govindjee S (1996) Finite element implementation of incompressible, transversely isotropic hyperelasticity. Comput Methods Appl Mech Eng 135(1–2): 107–128
Woo SLY, Simon BR, Kuei SC, Akeson WH (1980) Quasilinear viscoelastic properties of normal articular cartilage. J Biomech Eng 102: 85–90
Yoo LH, Kim H, Gupta V, Demer JL (2009) Quasi-linear viscoelastic behavior of bovine extra-ocular muscle tissue. Invest Ophthalmol Vis Sci, iovs. 50(8): 3721–3728
Acknowledgments
We thank Manning Beef, LLC, Pico Rivera, CA, for their generous contribution of bovine specimens. We thank Jose Martinez, Claudia Tamayo and Ramiro Carlos for assistance with specimen preparation. We thank Vadims Poukens, M.D., Ph.D. for histological preparation and Andrew Shin for assistance with experimental procedures.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Support: Supported by US Public Health Service, National Eye Institute: grants EY08313 and EY00331, and Research to Prevent Blindness. J. Demer is Leonard Apt Professor of Ophthalmology.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Yoo, L., Gupta, V., Lee, C. et al. Viscoelastic properties of bovine orbital connective tissue and fat: constitutive models. Biomech Model Mechanobiol 10, 901–914 (2011). https://doi.org/10.1007/s10237-010-0281-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-010-0281-z