Abstract
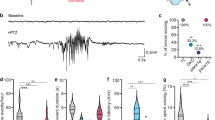
Increased neuronal excitability causes seizures with debilitating symptoms. Effective and noninvasive treatments are limited for easing symptoms, partially due to the complexity of the disorder and lack of knowledge of specific molecular faults. An unexplored, novel target for seizure therapeutics is the cGMP/protein kinase G (PKG) pathway, which targets downstream K+ channels, a mechanism similar to Retigabine, a recently FDA-approved antiepileptic drug. Our results demonstrate that increased PKG activity decreased seizure duration in C. elegans utilizing a recently developed electroconvulsive seizure assay. While the fly is a well-established seizure model, C. elegans are an ideal yet unexploited model which easily uptakes drugs and can be utilized for high-throughput screens. In this study, we show that treating the worms with either a potassium channel opener, Retigabine or published pharmaceuticals that increase PKG activity, significantly reduces seizure recovery times. Our results suggest that PKG signaling modulates downstream K+ channel conductance to control seizure recovery time in C. elegans. Hence, we provide powerful evidence, suggesting that pharmacological manipulation of the PKG signaling cascade may control seizure duration across phyla.





Similar content being viewed by others
References
Barrese V, Miceli F, Soldovieri MV, Ambrosino P, Iannotti FA, Cilio MR et al (2010) Neuronal potassium channel openers in the management of epilepsy: role and potential of retigabine. Clin Pharmacol 2:225–236. https://doi.org/10.2147/cpaa.s15369
Brodie MJ, Barry SJ, Bamagous GA, Norrie JD, Kwan P (2012) Patterns of treatment response in newly diagnosed epilepsy. Neurology 78(20):1548–1554. https://doi.org/10.1212/WNL.0b013e3182563b19
Calahorro F, Ruiz-Rubio M (2011) Caenorhabditis elegans as an experimental tool for the study of complex neurological diseases: Parkinson’s disease, Alzheimer’s disease and autism spectrum disorder. Invert Neurosci 11(2):73–83. https://doi.org/10.1007/s10158-011-0126-1
Caplan SL, Milton SL, Dawson-Scully K (2013) A cGMP-dependent protein kinase (PKG) controls synaptic transmission tolerance to acute oxidative stress at the Drosophila larval neuromuscular junction. J Neurophysiol 109(3):649–658. https://doi.org/10.1152/jn.00784.2011
Chalfie M, Tu Y, Euskirchen G, Ward WW, Prasher DC (1994) Green fluorescent protein as a marker for gene expression. Science 263(5148):802–805
Daniels SA, Ailion M, Thomas JH, Sengupta P (2000) egl-4 acts through a transforming growth factor-beta/SMAD pathway in Caenorhabditis elegans to regulate multiple neuronal circuits in response to sensory cues. Genetics 156(1):123–141
Dawson-Scully K, Bukvic D, Chakaborty-Chatterjee M, Ferreira R, Milton SL, Sokolowski MB (2010) Controlling anoxic tolerance in adult Drosophila via the cGMP–PKG pathway. J Exp Biol 213(Pt 14):2410–2416. https://doi.org/10.1242/jeb.041319
Friedman LK, Slomko AM, Wongvravit JP, Naseer Z, Hu S, Wan WY et al (2015) Efficacy of retigabine on acute limbic seizures in adult rats. J Epilepsy Res 5(2):46–59. https://doi.org/10.14581/jer.15010
Fujiwara M, Sengupta P, McIntire SL (2002) Regulation of body size and behavioral state of C. elegans by sensory perception and the EGL-4 cGMP-dependent protein kinase. Neuron 36(6):1091–1102
Guerrini R, Rosati A, Giordano F, Genitori L, Barba C (2013) The medical and surgical treatment of tumoral seizures: current and future perspectives. Epilepsia 54(Suppl 9):84–90. https://doi.org/10.1111/epi.12450
Hahm JH, Kim S, Paik YK (2009) Endogenous cGMP regulates adult longevity via the insulin signaling pathway in Caenorhabditis elegans. Aging Cell 8(4):473–483. https://doi.org/10.1111/j.1474-9726.2009.00495.x
Hamamichi S, Rivas RN, Knight AL, Cao S, Caldwell KA, Caldwell GA (2008) Hypothesis-based RNAi screening identifies neuroprotective genes in a Parkinson’s disease model. Proc Natl Acad Sci USA 105(2):728–733. https://doi.org/10.1073/pnas.0711018105
Kaun KR, Hendel T, Gerber B, Sokolowski MB (2007) Natural variation in Drosophila larval reward learning and memory due to a cGMP-dependent protein kinase. Learn Mem 14(5):342–349. https://doi.org/10.1101/lm.505807
Kim JB (2014) Channelopathies. Korean J Pediatr 57(1):1–18. https://doi.org/10.3345/kjp.2014.57.1.1
Krzyzanowski MC, Brueggemann C, Ezak MJ, Wood JF, Michaels KL, Jackson CA et al (2013) The C. elegans cGMP-dependent protein kinase EGL-4 regulates nociceptive behavioral sensitivity. PLoS Genet 9(7):e1003619. https://doi.org/10.1371/journal.pgen.1003619
Lee SK (2014) Old versus new: why do we need new antiepileptic drugs? J Epilepsy Res 4(2):39–44
L’Etoile ND, Coburn CM, Eastham J, Kistler A, Gallegos G, Bargmann CI (2002) The cyclic GMP-dependent protein kinase EGL-4 regulates olfactory adaptation in C. elegans. Neuron 36(6):1079–1089
Levitan D, Doyle TG, Brousseau D, Lee MK, Thinakaran G, Slunt HH et al (1996) Assessment of normal and mutant human presenilin function in Caenorhabditis elegans. Proc Natl Acad Sci USA 93(25):14940–14944
Link CD, Taft A, Kapulkin V, Duke K, Kim S, Fei Q et al (2003) Gene expression analysis in a transgenic Caenorhabditis elegans Alzheimer’s disease model. Neurobiol Aging 24(3):397–413
Liu J, Ward A, Gao J, Dong Y, Nishio N, Inada H et al (2010) C. elegans phototransduction requires a G protein-dependent cGMP pathway and a taste receptor homolog. Nat Neurosci 13(6):715–722. https://doi.org/10.1038/nn.2540
Loscher W, Schmidt D (2011) Modern antiepileptic drug development has failed to deliver: ways out of the current dilemma. Epilepsia 52(4):657–678. https://doi.org/10.1111/j.1528-1167.2011.03024.x
Mary Jane E, Catharyn TL, Andrea MS, Larisa MS (2012) Epilepsy across the spectrum: promoting health and understanding. The National Academies Press, Washington, DC
Mery F, Belay AT, So AK, Sokolowski MB, Kawecki TJ (2007) Natural polymorphism affecting learning and memory in Drosophila. Proc Natl Acad Sci USA 104(32):13051–13055. https://doi.org/10.1073/pnas.0702923104
Opperman KJ, Mulcahy B, Giles AC, Risley MG, Birnbaum RL, Tulgren ED et al (2017) The HECT family ubiquitin ligase EEL-1 regulates neuronal function and development. Cell Rep 19(4):822–835. https://doi.org/10.1016/j.celrep.2017.04.003
Raizen DM, Cullison KM, Pack AI, Sundaram MV (2006) A novel gain-of-function mutant of the cyclic GMP-dependent protein kinase egl-4 affects multiple physiological processes in Caenorhabditis elegans. Genetics 173(1):177–187. https://doi.org/10.1534/genetics.106.057380
Renger JJ, Yao WD, Sokolowski MB, Wu CF (1999) Neuronal polymorphism among natural alleles of a cGMP-dependent kinase gene, foraging, in Drosophila. J Neurosci 19(19):RC28
Risley MG, Kelly SP, Jia K, Grill B, Dawson-Scully K (2016) Modulating behavior in C. elegans using electroshock and antiepileptic drugs. PLoS ONE 11(9):e0163786. https://doi.org/10.1371/journal.pone.0163786
Rundfeldt C (1997) The new anticonvulsant retigabine (D-23129) acts as an opener of K+ channels in neuronal cells. Eur J Pharmacol 336(2–3):243–249
Saeki S, Yamamoto M, Iino Y (2001) Plasticity of chemotaxis revealed by paired presentation of a chemoattractant and starvation in the nematode Caenorhabditis elegans. J Exp Biol 204(Pt 10):1757–1764
Szaflarski JP, Nazzal Y, Dreer LE (2014) Post-traumatic epilepsy: current and emerging treatment options. Neuropsychiatr Dis Treat 10:1469–1477. https://doi.org/10.2147/ndt.s50421
Timmons L, Court DL, Fire A (2001) Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 263(1–2):103–112
White JG, Southgate E, Thomson JN, Brenner S (1986) The structure of the nervous system of the nematode Caenorhabditis elegans. Philos Trans R Soc Lond B Biol Sci 314(1165):1–340
Zhang Y, Lu H, Bargmann CI (2005) Pathogenic bacteria induce aversive olfactory learning in Caenorhabditis elegans. Nature 438(7065):179–184. https://doi.org/10.1038/nature04216
Acknowledgements
Research was supported by a compound transfer grant (CTP) Grant from Pfizer WI225058 for KD-S. Some strains were provided by the CGC, which is funded by National Institute of Health (NIH) Office of Research Infrastructure Programs (P40 OD010440).
Funding
Research was supported by a compound transfer grant (CTP) grant from Pfizer WI225058 for KD-S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Rights and permissions
About this article
Cite this article
Risley, M.G., Kelly, S.P., Minnerly, J. et al. egl-4 modulates electroconvulsive seizure duration in C. elegans. Invert Neurosci 18, 8 (2018). https://doi.org/10.1007/s10158-018-0211-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10158-018-0211-9