Abstract
Background
Surgical treatments for renal cell carcinoma reduces kidney volume to some degree and may derive postsurgical chronic kidney disease. We made a new marker for postoperative renal function using CT volumetry. To determine the impact of various parameters including this marker, we observed pre- and postsurgical renal function of experienced cases.
Methods
From 2004 to 2014, we underwent total or partial nephrectomy for 181 patients with renal carcinoma in a single institution. Of the total, 138 cases with presurgical CT volumetry were included in this study. We evaluated parameters for assessments of peri- and postoperative renal function including age, gender, serum creatinine, eGFR, performed surgery, pathology, estimated residual kidney volume and associated disease. Presence or absence of acute kidney injury (AKI) and chronic kidney disease (CKD) were also evaluated before, immediately after and 5 years after surgery.
Results
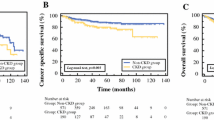
Multiple logistic regression analysis identified AKI, preoperative eGFR and estimated residual kidney volume as significant prognostic factors for the postoperative CKD. Moreover, cases with triple positive of these factors suffer postoperative CKD more significantly than those with one or two positives.
Conclusion
Using these predictive factors, we may determine patients with high risk for CKD who require an early intervention of renal protective treatment.



Similar content being viewed by others
References
Hori M, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H, et al. Cancer incidence and incidence rates in Japan in 2009: a study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 2015;45(9):884–91.
Einstein DM, Herts BR, Weaver R, Obuchowski N, Zepp R, Singer A. Evaluation of renal masses detected by excretory urography: cost-effectiveness of sonography versus CT. AJR Am J Roentgenol. 1995;164(2):371–5.
Colombo JR Jr, Haber GP, Jelovsek JE, Lane B, Novick AC, Gill IS. Seven years after laparoscopic radical nephrectomy: oncologic and renal functional outcomes. Urology. 2008;71(6):1149–54.
Shinder BM, Rhee K, Farrell D, Farber NJ, Stein MN, Jang TL, et al. Surgical management of advanced and metastatic renal cell carcinoma: a multidisciplinary approach. Front Oncol. 2017;7:107.
Van Poppel H, Da Pozzo L, Albrecht W, Matveev V, Bono A, Borkowski A, et al. A prospective, randomised EORTC intergroup phase 3 study comparing the oncologic outcome of elective nephron-sparing surgery and radical nephrectomy for low-stage renal cell carcinoma. Eur Urol. 2011;59(4):543–52.
Huang WC, Levey AS, Serio AM, Snyder M, Vickers AJ, Raj GV, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7(9):735–40.
Patel HD, Kates M, Pierorazio PM, Gorin MA, Jayram G, Ball MW, et al. Comorbidities and causes of death in the management of localized T1a kidney cancer. Int J Urol. 2014;21(11):1086–92.
Weight CJ, Larson BT, Gao T, Campbell SC, Lane BR, Kaouk JH, et al. Elective partial nephrectomy in patients with clinical T1b renal tumors is associated with improved overall survival. Urology. 2010;76(3):631–7.
Tan HJ, Norton EC, Ye Z, Hafez KS, Gore JL, Miller DC. Long-term survival following partial vs radical nephrectomy among older patients with early-stage kidney cancer. JAMA. 2012;307(15):1629–35.
Tobert CM, Riedinger CB, Lane BR. Do we know (or just believe) that partial nephrectomy leads to better survival than radical nephrectomy for renal cancer? World J Urol. 2014;32(3):573–9.
Huang WC, Donin NM, Levey AS, Campbell SC. Chronic kidney disease and kidney cancer surgery: new perspectives. J Urol. 2020;203(3):475–85. https://doi.org/10.1097/JU.0000000000000326.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92.
Weng PH, Hung KY, Huang HL, Chen JH, Sung PK, Huang KC. Cancer-specific mortality in chronic kidney disease: longitudinal follow-up of a large cohort. Clin J Am Soc Nephrol. 2011;6(5):1121–8.
Dunkler D, Gao P, Lee SF, Heinze G, Clase CM, Tobe S, et al. Risk prediction for early CKD in type 2 diabetes. Clin J Am Soc Nephrol. 2015;10(8):1371–9.
Gansevoort RT, Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. Lower estimated GFR and higher albuminuria are associated with adverse kidney outcomes. A collaborative meta-analysis of general and high-risk population cohorts. Kidney Int. 2011;80(1):93–104.
Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012;2(2):1303–53.
Funahashi Y, Yoshino Y, Sassa N, Matsukawa Y, Takai S, Gotoh M. Comparison of warm and cold ischemia on renal function after partial nephrectomy. Urology. 2014;84(6):1408–12.
Gong IH, Hwang J, Choi DK, Lee SR, Hong YK, Hong JY, et al. Relationship among total kidney volume, renal function and age. J Urol. 2012;187(1):344–9.
Simmons MN, Fergany AF, Campbell SC. Effect of parenchymal volume preservation on kidney function after partial nephrectomy. J Urol. 2011;186(2):405–10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB approval number RK-170711-7) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supp Fig 1. The association of age and eGFR at each time (before: A, 6 months: B, 12 months: C, 36 months: and D, 60 months: E after surgery).

10157_2020_1984_MOESM1_ESM.jpg
Supp Fig 1 The association of age and eGFR at each time (before: A, 6 months: B, 12 months: C, 36 months: and D, 60 months: E after surgery) (JPG 949 kb)
Supp Fig 2. ROC curves for estimated residual kidney volume (A), eGFR (B), and Age (C) to predict CKD development.

10157_2020_1984_MOESM2_ESM.jpg
Supp Fig 2 ROC curves for estimated residual kidney volume (A), eGFR (B), and Age (C) to predict CKD development (JPG 836 kb)
Supp Fig 3. The association of renal ischemic time with AKI after the surgery (A), CKD development (B), and eGFR after 6 months the surgery (C).

10157_2020_1984_MOESM3_ESM.jpg
Supp Fig 3 The association of renal ischemic time with AKI after the surgery (A), CKD development (B), and eGFR after 6 months the surgery (C) (JPG 623 kb)
About this article
Cite this article
Hori, Y., Obinata, D., Funakoshi, D. et al. Preoperative CT volumetry of estimated residual kidney for prediction of postoperative chronic kidney disease in patients with renal cell carcinoma. Clin Exp Nephrol 25, 315–321 (2021). https://doi.org/10.1007/s10157-020-01984-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-020-01984-8