Abstract
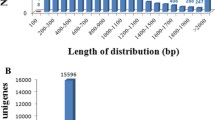
Grain aphid (Sitobion avenae F.) is the most dominant and destructive pest of wheat, which causes significant yield loss of cereal plants each year by inflicting damage both through the direct effects of feeding and by vectoring debilitating plant viruses. In this study, we performed de novo transcriptome sequencing of grain aphid via Roche 454 GS-FLX pyrosequencing. A total of 1,106,696 reads were obtained and assembled into 32,277 unigenes, of which 25,389, 21,635, and 16,211 unigenes matched the Nt, Nr, and Swiss-Prot databases, respectively. Functional annotation of these unigenes revealed not only the presence of genes that encode the key components of RNAi machinery such as Dicer and Argonaute but also the genes encoding the TAR RNA binding protein (TRBP) and the SID-1 protein, which function in assisting the RNA-induced silencing complex (RISC) formation in microRNA (miRNA) pathway and mediating a systemic RNA interference (RNAi) effect though a cellular uptake mechanism. Furthermore, among a set of 66 unigenes selected for a double-stranded RNA (dsRNA) artificial diet assay, four novel effective RNAi targets, which led to high mortality of aphids due to the down-regulation of the expression of the respective target gene, were identified. Moreover, the expansion of systemic RNAi effect in grain aphid was observed by adding the fluorescently labeled dsRNA in an artificial diet assay.




Similar content being viewed by others
Abbreviations
- BLAST:
-
Basic Local Alignment Search Tool
- KOG:
-
Clusters of orthologous groups
- dsRNA:
-
Double-stranded RNA
- ESTs:
-
Expressed sequence tags
- GO:
-
Gene ontology
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- Nr:
-
Non-redundant database
- Nt:
-
Nucleic acid database
- qPCR:
-
Quantitative polymerase chain reaction
- RNAi:
-
RNA interference
- RPKM:
-
Read per kilobase per million
- RT-PCR:
-
Reverse-transcript polymerase chain reaction
References
Aqueel MA, Leather SR (2011) Effect of nitrogen fertilizer on the growth and survival of Rhopalosiphum padi (L.) and Sitobion avenae (F.) (Homoptera: Aphididae) on different wheat cultivars. Crop Prot 30:216–221
Awmack CS, Harrington R (2000) Elevated CO2 affects the interactions between aphid pests and host plant flowering. Agric For Entomol 2:57–61
Baum JA, Bogaert T, Clinton W, Heck GR, Feldmann P, Ilagan O, Johnson S, Plaetinck G, Munyikwa T, Pleau M, Vaughn T, Roberts J (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25:1322–1326
Bautista MA, Miyata T, Miura K, Tanaka T (2009) RNA interference-mediated knockdown of a cytochrome P450, CYP6BG1, from the diamondback moth, Plutella xylostella, reduces larval resistance to permethrin. Insect Biochem Mol Biol 39:38–46
Bhatia V, Bhattacharya R, Uniyal PL, Singh R, Niranjan RS (2012) Host generated siRNAs attenuate expression of serine protease Gene in Myzus persicae. PLoS One 7:e46343
Blackman RL, Eastop VF (1984) Aphids on the world’s crops: an identification and information guide. Wiley, New York
Chen YA, Lin CC, Wang CD, Wu HB, Wang PI (2007) An optimized procedure greatly improves EST vector contamination removal. BMC Genomics 8:416
Chevreux B, Pfisterer T, Drescher B, Driesel AJ, Müller WE, Wetter T, Suhai S (2004) Using the miraEST assembler for reliable and automated mRNA transcript assembly and SNP detection in sequenced ESTs. Genome Res 14:1147–1159
Chou HH, Michael HH (2001) DNA sequence quality trimming and vector removal. Bioinformatics 17:1093–1104
Conesa A, Gotz S, García-Gómez JM, Terol J, Talón M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
DI Deraison C, Duportets L, Gorojankina T, Rahb Y, Jouanin L (2002) Cloning and characterization of a gut-specific cathepsin L from the aphid Aphis gossypii. Insect Mol Biol 13:265–177
Dzitoyeva S, Dimitrijevic N, Manev H (2001) Intra-abdominal injection of double-stranded RNA into anesthetized adult Drosophila triggers RNA interference in the central nervous system. Mol Psychiatry 6:665
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9:102–114
Fire A, Xu S, Montgomery M, Kostas S, Driver S, Mello C (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Gaur RK, Krupp G (1993) Enzymatic RNA synthesis with deoxynucleoside 5′-O (1-thiotriphosphates). FEBS Lett 315:56–60
Huvenne H, Smagghe G (2010) Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: a review. J Insect Physiol 56:227–235
International Aphid Genomics Consortium (2010b) Aphid White Paper II: proposal to complete development of the aphid model. IAGC–White Paper II-FINAL VERSION-September 13th 2010.
International Aphid Genomics Consortium (2010) Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol 8:e1000313
Jaubert-Possamai S, Le Trionnaire G, Bonhomme J, Christophides GK, Rispe C, Tagu D (2007) Gene knockdown by RNAi in the pea aphid Acyrthosiphon pisum. BMC Biotechnol 7:63
Jinek M, Doudna JA (2008) A three-dimensional view of the molecular machinery of RNA interference. Nature 457:405–412
Jose AM, Hunter CP (2007) Transport of sequence-specific RNA interference information between cells. Annu Rev Gene 41:305–330
Lassmann T, Hayashizaki Y, Daub CO (2009) TagDust—a program to eliminate artifacts from next generation sequencing data. Bioinformatics 25:2839–2840
Legeai F, Shigenobu S, Gauthier JP, Colbourne J, Rispe C, Collin O, Richards S, Wilson ACC, Tagu D (2010) AphidBase: a centralized bioinformatic resource for annotation of the pea aphid genome. Insect Mol Biol 19:5–12
Liao Z, Jia QD, Li F, Han ZJ (2010) Identification of two piwi genes and their expression profile in honeybee, Apis mellifera. Arch Insect Biochem Physiol 74:91–102
Livak KJ, Thomas DS (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lynch JA, Desplan C (2006) A method for parental RNA interference in the wasp Nasonia vitripennis. Nat Protoc 1:486–494
Mao YB, Cai WJ, Wang JW, Hong GJ, Tao XY, Wang LJ, Huang YP, Chen XY (2007) Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25:1307–1313
Martín D, Maestro O, Cruz J, Mané-Padrós D, Bellés X (2006) RNAi studies reveal a conserved role for RXR in molting in the cockroach Blattella germanica. Insect Physiol 52:410–416
Meister G, Tuschl T (2004) Mechanisms of gene silencing by double-stranded RNA. Nature 431:343–349
Meyering-Vos M, Müller A (2007) A Structure of the sulfakinin cDNA and gene expression from the mediterranean field cricket Gryllus bimaculatus. Insect Mol Biol 16:445–454
Meyering-Vos M, Merz S, Sertkol M, Hoffmann KH (2006) Functional analysis of the allatostatin-a type gene in the cricket Gryllus bimaculatus and the armyworm Spodoptera frugiperda. Insect Biochem Mol Biol 36:492–504
Michel H, Behr J, Harrenga A, Kannt A (1998) Cytochrome c oxidase: structure and spectroscopy. Annu Rev Bioph Biom 27:329–356
Morrison WP, Peairs FB (1998) Response model concept and economic impact. Response model for an introduced pest: the Russian wheat aphid. Lanham: MD: Entomol Soc Am.
Mutti NS, Park Y, Reese JC, Reeck GR (2006) RNAi knockdown of a salivary transcript leading to lethality in the pea aphid, Acyrthosiphon pisum. J Insect Sci 6:1–7
Nakakita H, Katsumata Y, Ozawa T (1971) The effect of phosphine on respiration of rat liver mitochondria. J Biochem 69:589–593
Oerke EC, Dehne HW, Schönbeck F, Weber A (1994) Crop production and crop protection: estimated losses in major food and cash crops. Elsevier, Amsterdam, pp 179–296
Pitino M, Maffei ME, Coleman AD, Ridout CJ, Hogenhout SA (2011) Silencing of aphid genes by dsRNA feeding from plants. PLoS One 6:e25709
Price DR, Gatehouse JA (2008) RNAi-mediated crop protection against insects. Trends Biotechnol 26:393–400
Shakesby AJ, Wallace IS, Isaacs HV, Pritchard J, Roberts DM, Douglas AE (2009) A water-specific aquaporin involved in aphid osmoregulation. Insect Biochem Mol Biol 39:1–10
Siomi H, Siomi MC (2009) On the road to reading the RNA-interference code. Nature 457:396–404
Smith CM, Belay T, Stauffer C, Stary P, Kubeckova I, Starkey S (2004) Identifiaction of Russian wheat aphid (Homoptera: Aphididae) biotypes virulent to the Dn4 resistance gene. J Econ Entomol 97:112–117
Sternlicht MD, Werb Z (2001) How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dve Bi 17:463–516
Stoger E, Willianms S, Christou P, Down RE, Gatehouse JA (1999) Expression of the insecticidal lectin from snowdrop (Galanthus nivalis agglutinin; GNA) in transgenic wheat plants: effects on predation by the grain aphid Sitobion avenae. Mol Breed 5:65–73
Taanman JW, Williams SL (2001) Assembly of cytochrome c oxidase: what can we learn from patients with cytochrome c oxidase deficiency? Biochem Soc T 29:446–451
Tagu D, Klingler JP, Moya A, Simon JC (2008) Early progress in aphid genomics and consequences for plant-aphid interactions studies. Mol Plant-Microbe Interact 21(6):701–708
Terenius O, Papanicolaou A, Garbutt JS, Eleftherianos I, Huvenne H, Kanginakudru S, Albrechtsen M, An C, Aymeric JL, Barthel A (2011) RNA interference in Lepidoptera: an overview of successful and unsuccessful studies and implications for experimental design. J Insect Physiol 57:231–245
Tian H, Peng H, Yao Q, Chen H, Xie Q, Tang B, Zhang W (2009) Developmental control of a lepidopteran pest Spodoptera exigua by ingestion of bacteria expressing dsRNA of a non-midgut gene. PLoS One 4:e6225
Tomoyasu Y, Miller SC, Tomita S, Schoppmeier M, Grossmann D, Bucher G (2008) Exploring systemic RNA interference in insects: a genome-wide survey for RNAi genes in Tribolium. Genome Biol 9:R10
Turner CT, Davy MW, MacDiarmid RM, Plummer KM, Birch NP, Newcomb RD (2006) RNA interference in the light brown apple moth, Epiphyas postvittana (Walker) induced by double-stranded RNA feeding. Insect Mol Biol 15:383–391
Van Roessel, A.H. Brand (2004) Spreading silence with Sid. Genome Biology, 5 (2004), p. 208Wang CP, Chen Q, Luo K, Zhao HY, Zhang GS, Tlali, RM (2011) Evaluation of resistance in wheat germplasm to the aphids, Sitobion avenae based on Technique for Order Preference by Similarity to Ideal Solution TOPSIS and cluster methods. Afr J Agric Res 6: 1592–1599
Whangbo JS, Hunter CP (2008) Environmental RNA interference. Trends Genet 24:297–305
Whyard S, Singh AD, Wong S (2009) Ingested double-stranded RNAs can act as species-specific insecticides. Insect Biochem Mol Biol 39:824–832
Williams RW, Rubin GM (2002) ARGONAUTE1 is required for efficient RNA interference in Drosophila embryos. Proc Natl Acad Sci U S A 99:6889–6894
Xia LQ, Ma Y, He Y, Jones HD (2012) GM wheat development in China: current status and challenges to commercialization. J Exp Bot 63:1785–1790
Xu WN, Han ZJ (2008) Cloning and phylogenetic analysis of sid-1-like genes from aphids. J Insect Sci 8:1–6
Ye J, Fang L, Zheng HK, Zhang Y, Chen J, Zhang ZJ, Wang J, Li ST, Li RQ, Bolund L, Wang J (2006) WEGO: a web tool for plotting GO annotations. Nucleic Acids Res 34:293–297
Yu XD, Pickett J, Ma Y, Bruce T, Napier J, Jones HD, Xia LQ (2012) Metabolic engineering of plant-derived (E)-β-farnesene synthase genes for a novel type of aphid-resistant genetically-modified crop plants. J Integr Plant Biol 54:282–299
Yu XD, Wang GP, Huang SL, Ma YZ, Xia LQ (2014) Engineering plants for aphid resistance: current status and future perspectives. Theor Appl Genet 127:2065–2083
Zha WJ, Peng XX, Chen RZ, Du B, Zhu LL, He GC (2011) Knockdown of midgut genes by dsRNA-transgenic plant-mediated RNA interference in the hemipteran insect Nilaparvata lugens. PLoS One 6:e20504
Zhang M, Zhou Y, Wang H, Jones HD, Gao Q, Wang DH, Ma YZ, Xia LQ (2013) Identifying potential RNAi targets in grain aphid (Sitobion avenae F.) based on transcriptome profiling of its alimentary canal after feeding on wheat plants. BMC Genomics 14:560
Zhou X, Wheeler MM, Oi FM, Scharf ME (2008) RNA interference in the termite Reticulitermes flavipes through ingestion of double-stranded RNA. Insect Biochem Mol Biol 38:805–815
Acknowledgments
The authors thank Prof. Huw Jones at Rothamsted Research, UK, for his critical review of this manuscript. This work was supported by grants from the Research Initiative on Transgenic Plants from the Ministry of Agriculture of China (2014ZX0800201B), Natural Science Foundation of China (grant nos. 31171618 and 31371702), and the Chinese State Key Laboratory for Biology of Plant Diseases and Insects (grant no. SKLOF201307). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
The primer sets of 66 unigenes selected for dsRNA amplification. (XLSX 19 kb)
Table S2
The primer sets of 14 unigenes selected for qPCR amplifying to detecting the expression correlation between RPKM and qPCR of unigenes. (XLSX 22 kb)
Table S3
The annotations of 66 unigenes selected for dsRNA feeding experiments. (XLSX 31 kb)
Figure S1
The length distribution of contigs, singletons, and unigenes, and reads number distribution of the unigenes. (GIF 27 kb)
Figure S2
A Venn diagram illustrating shared and unique unigenes annotated in Nr, Swiss-Prot, KOG, GO, and KEGG public databases. Among 32,277 unigenes assembled, 27,093 unigenes were annotated in at least one of the public databases, including 21,635, 16,211, 15,957, 13,876, and 11,731 in Nr, Swiss-Prot, KOG, KEGG, and GO databases, respectively. (GIF 6 kb)
Rights and permissions
About this article
Cite this article
Wang, D., Liu, Q., Li, X. et al. Double-stranded RNA in the biological control of grain aphid (Sitobion avenae F.). Funct Integr Genomics 15, 211–223 (2015). https://doi.org/10.1007/s10142-014-0424-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-014-0424-x