Abstract
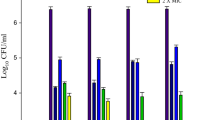
This study has evaluated the effects of photodynamic inactivation (PDI) using erythrosine as photosensitizer and green light-emitting diode (LED) on biofilms of Candida albicans alone and in combination with Enterococcus faecalis and Streptococcus mutans. We have also evaluated the effect of sucrose on biofilm formation and bacterial growth and sensitivity to PDI. Biofilms were formed in suspension of 106 cells/ml on plates before being grown in broth culture with and without sucrose and incubated for 48 h. Next, the treatment was applied using erythrosine at a concentration of 400 μM for 5 min and green LED (532 ± 10 nm) for 3 min on biofilms alone and in combination. The plates were washed and sonicated to disperse the biofilms, and serial dilutions were carried and aliquots seeded in Sabouraud agar before incubation for 48 h. Next, the colony-forming units per milliliter (CFU/ml; log10) were counted and analyzed statistically (ANOVA, Tukey test, P ≤ 0.05). Results show that S. mutans favors the growth of C. albicans in biofilms with sucrose, with treatment not being effective. However, when the biofilm was grown without sucrose, we found a reduction in biofilm formation and a significant decrease in the PDI treatment (P < 0.0001). In conclusion, both growth and sensitivity to PDI in biofilms of C. albicans are strongly influenced by bacterial combination, and the presence of sucrose affected directly the growth and sensitivity of the biofilm to PDI as sucrose is the substrate for construction of the exopolysaccharide matrix.




Similar content being viewed by others
References
Kuramitsu HK, He X, Lux R, Anderson MH, Shi W (2007) Interspecies interactions within oral microbial communities. Microbiol Mol Biol Rev 71(4):653–670. doi:10.1128/MMB12.00024-07
Kolenbrander PE, Andersen RN, Blehert DS, Egland PG, Foster JS, Palmer RJ (2002) Communication among oral bacteria. Microbiol Mol Biol Rev 66(3):486–505. doi:10.1128/MMBR.66.3.486-505.2002
Haffajee AD, Socransky SS (2006) Introduction to microbial aspects of periodontal biofilm communities, development and treatment. Periodontol 2000(42):7–12. doi:10.1111/j.1600-0757.2006.00190.x
Mager DL, Ximenez-Fyvie LA, Haffajee AD, Socransky SS (2003) Distribution of selected bacterial species on intraoral surfaces. J Clin Periodontol 30(7):644–654. doi:10.1034/j.1600-051X.2003.00376.x
Cruz MR, Graham CE, Gagliano BC, Lorenz MC, Garsin DA (2013) Enterococcus faecalis inhibits hyphal morphogenesis and virulence of Candida albicans. Infect Immun 81(1):189–200. doi:10.1128/IAI.00914-12
Shekh RM, Roy U (2012) Biochemical characterization of an anti-Candida factor produced by Enterococcus faecalis. BMC Microbiol 12(1):132. doi:10.1186/1471-2180-12-132
Brusca MI, Irastorza RM, Cattoni DI, Ozu M, Chara O (2013) Mechanisms of interaction between Candida albicans and Streptococcus mutans: an experimental and mathematical modelling study. Acta Odontol Scand 71(3–4):416–423. doi:10.3109/00016357.2012.690530
Seneviratne CJ, Jin L, Samaranayake LP (2008) Biofilm lifestyle of Candida: a mini review. Oral Dis 7:582–590. doi:10.1111/j.1601-0825.2007.01424.x
Souza RC, Junqueira JC, Rossoni RD, Pereira CA, Munin E, Jorge AOC (2010) Comparison of the photodynamic fungicidal efficacy ogf methylene blue, toluidine blue, malachite green and low-power laser irradiation alone against Candida albicans. Lasers Med Sci 3:385–389. doi:10.1007/s10103-009-0706-z
Branda SS, Vik S, Friedman L, Kolter R (2005) Biofilms: the matrix revisited. Trends Microbiol 13(1):20–26. doi:10.1016/j.tim.2004.11.006
Hube B (2004) From comensal to pathogen: stage-and tissue-specific gene expression of Candida albicans. Curr Opin Microbiol 4:336–334. doi:10.1016/j.mib.2004.06.003
Souza SC, Junqueira JC, Balducci I, Ito-Koga CY, Munin E, Jorge AOC (2006) Photosensitization of different Candida species by low power laser light. Photochem Photobiol B Biol 83(1):34–38. doi:10.1016/j.jphotobiol.2005.12.002
Lambrechts SA, Aalders MC, Marle JV (2005) Mechanistic study of the photodynamic inactivation of Candida albicans by a cationic porphyrin. Antimicrob Agents Chemother 5:2026–2034. doi:10.1128/AAC.49.5.2026–2034.2005
Lam M, Jou PC, Lattif AA, Lee Y, Malbasa CL, Mukherjee PK et al (2011) Photodynamic therapy with Pc 4 induces apoptosis of Candida albicans. Photochem Photobiol 4:904–909. doi:10.1111/j.1751-1097.2011.00938.x
Smijs TG, Pavel S (2011) The susceptibility of dermatophytes to photodynamic treatment with special focus on Trichophyton rubrum. Photochem Photobiol 1:2–13. doi:10.1111/j.1751-1097.2010.00848.x
Freire F, Costa ACBP, Pereira CA, Junior MB, Junqueira JC, Jorge AOC (2013) Comparison of the effect of rose bengal- and eosin Y-mediated photodynamic inactivation on planktonic cells and biofilms of Candida albicans. Lasers Med Sci 3:949–955. doi:10.1007/s10103-013-1435-x
Lyon JP, Moreira LM, Moraes PC, Santos FV, Resende MA (2011) Photodynamic therapy for pathogenic fungi. Mycoses 5:265–271. doi:10.1111/j.1439- 507.2010.01966.x
Koo H, Xiao J, Klein MI, Jeon JG (2010) Exopolysaccharides produced by Streptococcus mutans glucosyltransferases modulate the establishment of microcolonies within multispecies biofilms. J Bacteriol 192(12):3024–3032. doi:10.1128/JB.01649-09
Koo H, Falsetta ML, Klein MI (2013) The exopolysaccharide matrix: a virulence determinant of cariogenic biofilm. J Dent Res 92(12):1065–1073. doi:10.1177/0022034513504218
Klein MI, DeBaz L, Agidi S, Lee H, Xie G, Lin AHM, Hamaker BR, Lemos JA, Koo H (2010) Dynamics of Streptococcus mutans transcriptome in response to starch and sucrose during biofilm development. PLoS One 5(10):e13478. doi:10.1371/journal.pone.0013478
Costa AC, de Campos Rasteiro VM, Pereira CA, da Silva Hashimoto ES, Beltrame M Jr, Junqueira JC, Jorge AO (2011) Susceptibility of Candida albicans and Candida dubliniensis to erythrosine- and LED-mediated photodynamic therapy. Arch Oral Biol 56(11):1299–1305. doi:10.1016/j.archoralbio.2011.05.013
Costa AC, Rasteiro VM, Pereira CA, Rossoni RD, Junqueira JC, Jorge AO (2012) The effects of rose bengal- and erythrosine-mediated photodynamic therapy on Candida albicans. Mycoses 55(1):56–63. doi:10.1111/j.1439-0507.2011.02042.x
Costa AC, Campos VMR, Hashimoto ESS, Araújo CF, Pereira CA, Junqueira JC, Jorge AO (2012) Effect of erythrosine- and LED-mediated photodynamic therapy on buccal candidiasis infection of immunosuppressed mice and Candida albicans adherence to buccal epithelial cells. Oral Surg Oral Med Oral Pathol Oral Radiol 114(1):67–74. doi:10.1016/j.oooo.2012.02.002
Garsin DA, Lorenz MC (2013) Candida albicans and Enterococcus faecalis in the gut: synergy in commensalism? Gut Microbes 4(5):409–415. doi:10.4161/gmic.26040
Falsetta ML, Klein MI, Colonne PM, Scott-Anne K, Gregoire S, Pai CH, Gonzalez-Begne M, Watson G, Krysan DJ, Bowen WH, Koo H (2014) Symbiotic relationship between Streptococcus mutans and Candida albicans synergizes virulence of plaque biofilms in vivo. Infect Immun 82(5):1968–1981. doi:10.1128/IAI.00087-14
Sztajer H, Szafranski SP, Tomasch J, Reck M, Nimtz M, Rohde M, Wagner-Döbler (2013) Cross-feeding and interkingdom communication in dual-species biofilms of Streptococcus mutans and Candida albicans. ISME J. doi:10.1038/ismej.2014.73
Acknowledgements
The authors would like to thank the State of São Paulo University (UNESP) for the resources and encouragement of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Funding
The authors declare that they did not receive funding for the development of this research.
Ethical procedures
All ethical procedures were followed. Because the research used ATCC strains of microorganisms and did not involve any kinds of animal, the approval committee of ethics and informed consent do not apply to work.
Rights and permissions
About this article
Cite this article
Tomé, F.M., Paula Ramos, L.D., Freire, F. et al. Influence of sucrose on growth and sensitivity of Candida albicans alone and in combination with Enterococcus faecalis and Streptococcus mutans to photodynamic therapy. Lasers Med Sci 32, 1237–1243 (2017). https://doi.org/10.1007/s10103-017-2201-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-017-2201-2