Abstract
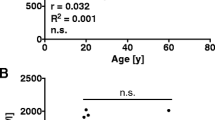
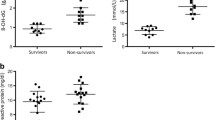
Lysophosphatidylcholine (LPC) has been suggested to serve as a useful prognostic marker for sepsis. However, existing LPC assays are complicated, time-consuming, and of limited application in real clinical situations. Thus, we investigated the serum LPC levels in sepsis patients using an enzymatic assay and analyzed the correlations between the serum LPC concentration and clinical characteristics. We prospectively collected blood samples from suspected sepsis patients, commencing on day 1 of sepsis. We analyzed all samples using an enzymatic assay. Additionally, we analyzed the serum LPC concentrations in a control group of 21 healthy blood donors. A total of 105 patients who fulfilled the sepsis criteria were included. The mean serum LPC concentration was 43.49 ± 33.09 μmol/L in sepsis patients, which was much lower than that of 21 healthy controls (234.68 ± 30.33 μmol/L, p < 0.001). Bacteremic sepsis was associated with a lower serum LPC concentration than non-bacteremic sepsis (34.81 ± 26.85 vs. 49.05 ± 35.63 μmol/L, p < 0.05). No difference in serum LPC concentration was evident between survivors and non-survivors. The serum LPC concentration tended to decrease with the severity of sepsis. The day 1 serum LPC concentration was decreased in patients with sepsis, especially when bacteremia was present. However, the serum LPC level did not correlate with disease severity and did not predict mortality from sepsis.


Similar content being viewed by others
References
Hotchkiss RS, Karl IE (2003) The pathophysiology and treatment of sepsis. N Engl J Med 348:138–150
Bulger EM, Maier RV (2000) Lipid mediators in the pathophysiology of critical illness. Crit Care Med 28(4 Suppl):N27–N36
Quinn MT, Parthasarathy S, Steinberg D (1988) Lysophosphatidylcholine: a chemotactic factor for human monocytes and its potential role in atherogenesis. Proc Natl Acad Sci USA 85:2805–2809
Nakano T, Raines EW, Abraham JA, Klagsbrun M, Ross R (1994) Lysophosphatidylcholine upregulates the level of heparin-binding epidermal growth factor-like growth factor mRNA in human monocytes. Proc Natl Acad Sci USA 91:1069–1073
Liu-Wu Y, Hurt-Camejo E, Wiklund O (1998) Lysophosphatidylcholine induces the production of IL-1β by human monocytes. Atherosclerosis 137:351–357
Coutant F, Perrin-Cocon L, Agaugué S, Delair T, André P, Lotteau V (2002) Mature dendritic cell generation promoted by lysophosphatidylcholine. J Immunol 169:1688–1695
Sakai M, Miyazaki A, Hakamata H, Sasaki T, Yui S, Yamazaki M et al (1994) Lysophosphatidylcholine plays an essential role in the mitogenic effect of oxidized low density lipoprotein on murine macrophages. J Biol Chem 269:31430–31435
Gómez-Muñoz A, O’Brien L, Hundal R, Steinbrecher UP (1999) Lysophosphatidylcholine stimulates phospholipase D activity in mouse peritoneal macrophages. J Lipid Res 40:988–993
De Vries HE, Ronken E, Reinders JH, Buchner B, Van Berkel TJ, Kuiper J (1998) Acute effects of oxidized low density lipoprotein on metabolic responses in macrophages. FASEB J 12:111–118
Ngwenya BZ, Yamamoto N (1986) Effects of inflammation products on immune systems. Lysophosphatidylcholine stimulates macrophages. Cancer Immunol Immunother 21:174–182
Ramos MA, Kuzuya M, Esaki T, Miura S, Satake S, Asai T et al (1998) Induction of macrophage VEGF in response to oxidized LDL and VEGF accumulation in human atherosclerotic lesions. Arterioscler Thromb Vasc Biol 18:1188–1196
McMurray HF, Parthasarathy S, Steinberg D (1993) Oxidatively modified low density lipoprotein is a chemoattractant for human T lymphocytes. J Clin Invest 92:1004–1008
Asaoka Y, Oka M, Yoshida K, Nishizuka Y (1991) Lysophosphatidylcholine as a possible second messenger synergistic to diacylglycerol and calcium ion for T-lymphocyte activation. Biochem Biophys Res Commun 178:1378–1385
Nishi E, Kume N, Ochi H, Moriwaki H, Wakatsuki Y, Higashiyama S et al (1997) Lysophosphatidylcholine increases expression of heparin-binding epidermal growth factor-like growth factor in human T lymphocytes. Circ Res 80:638–644
Nishi E, Kume N, Ueno Y, Ochi H, Moriwaki H, Kita T (1998) Lysophosphatidylcholine enhances cytokine-induced interferon gamma expression in human T lymphocytes. Circ Res 83:508–515
Asaoka Y, Oka M, Yoshida K, Sasaki Y, Nishizuka Y (1992) Role of lysophosphatidylcholine in T-lymphocyte activation: involvement of phospholipase A2 in signal transduction through protein kinase C. Proc Nat Acad Sci USA 89:6447–6451
Savage JE, Theron AJ, Anderson R (1993) Activation of neutrophil membrane-associated oxidative metabolism by ultraviolet radiation. J Invest Dermatol 101:532–536
Nishioka H, Horiuchi H, Arai H, Kita T (1998) Lysophosphatidylcholine generates superoxide anions through activation of phosphatidylinositol 3-kinase in human neutrophils. FEBS Lett 441:63–66
Kabarowski JH, Zhu K, Le LQ, Witte ON, Xu Y (2001) Lysophosphatidylcholine as a ligand for the immunoregulatory receptor G2A. Science 293:702–705
Le LQ, Kabarowski JH, Weng Z, Satterthwaite AB, Harvill ET, Jensen ER et al (2001) Mice lacking the orphan G protein-coupled receptor G2A develop a late-onset autoimmune syndrome. Immunity 14:561–571
Drobnik W, Liebisch G, Audebert FX, Frohlich D, Gluck T, Vogel P et al (2003) Plasma ceramide and lysophosphatidylcholine inversely correlate with mortality in sepsis patients. J Lipid Res 44:754–761
Yan JJ, Jung JS, Lee JE, Lee J, Huh SO, Kim HS et al (2004) Therapeutic effects of lysophosphatidylcholine in experimental sepsis. Nat Med 10:161–167
Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA et al (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 101:1644–1655
Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH (1999) Manual of clinical microbiology, 7th edn. American Society for Microbiology, Washington, DC
Riedel S, Melendez JH, An AT, Rosenbaum JE, Zenilman JM (2011) Procalcitonin as a marker for the detection of bacteremia and sepsis in the emergency department. Am J Clin Pathol 135:182–189
Jones AE, Fiechtl JF, Brown MD, Ballew JJ, Kline JA (2007) Procalcitonin test in the diagnosis of bacteremia: a meta-analysis. Ann Emerg Med 50:34–41
Sierra R, Rello J, Bailén MA, Benítez E, Gordillo A, León C et al (2004) C-reactive protein used as an early indicator of infection in patients with systemic inflammatory response syndrome. Intensive Care Med 30:2038–2045
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2011–0006208).
We thank clinical researcher E.M. Cho for reviewing the medical records.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, W.H., Park, T., Park, Y.Y. et al. Clinical significance of enzymatic lysophosphatidylcholine (LPC) assay data in patients with sepsis. Eur J Clin Microbiol Infect Dis 31, 1805–1810 (2012). https://doi.org/10.1007/s10096-011-1505-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-011-1505-6