Abstract
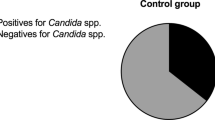
Candida infection among multiple sclerosis (MS) patients has not been studied in depth. We determined whether there is an association between serological evidence of Candida infection and MS. Blood specimens were obtained from 80 MS patients and 240 matched controls. Immunofluorescence analysis and ELISA were used to detect Candida species antibodies and slot-blot to detect antigens. Using immunofluorescence analysis, moderate to high concentrations of serum antibodies to Candida famata were present in 30 (37.5%) MS patients vs. 30 (12.5%) controls (p < 0.001). Results for Candida albicans were 47.5% (38/80) in MS patients vs. 21.3% (51/240) in controls (p < 0.001), for Candida parapsilosis 37% (28/80) vs. 17.1% (41/240) (p < 0.001) and for Candida glabrata 46.3% (37/80) vs. 17.5% (42/240) (p < 0.001), respectively. After adjusting for age and gender, the odds ratios (95% confidence intervals) for MS, according to the presence of Candida antigens were: 2.8 (0.3–23.1, p = 0.337) for Candida famata; 1.5 (0.7–3.4, p = 0.290) for Candida albicans; 7.3 (3.2–16.6, p < 0.001) for Candida parapsilosis; and 3.0 (1.5–6.1, p = 0.002) for Candida glabrata. The results were similar after excluding ten patients on immunosuppressants. The results of this single study suggest that Candida species infection may be associated with increased odds of MS.
Similar content being viewed by others
References
Pugliatti M, Sotgiu S, Rosati G (2002) The worldwide prevalence of multiple sclerosis. Clin Neurol Neurosurg 104:182–191
Benito-León J, Morales JM, Rivera-Navarro J (2002) Health-related quality of life and its relationship to cognitive and emotional functioning in multiple sclerosis patients. Eur J Neurol 9:497–502
Benito-León J, Morales JM, Rivera-Navarro J, Mitchell A (2003) A review about the impact of multiple sclerosis on health-related quality of life. Disabil Rehabil 25:1291–1303
Mitchell AJ, Benito-León J, Gonzalez JM, Rivera-Navarro J (2005) Quality of life and its assessment in multiple sclerosis: integrating physical and psychological components of well-being. Lancet Neurol 4:556–566
Morales-Gonzalez JM, Benito-León J, Rivera-Navarro J, Mitchell AJ; GEDMA Study Group (2004) A systematic approach to analyse health-related quality of life in multiple sclerosis: the GEDMA study. Mult Scler 10:47–54
Rivera-Navarro J, Morales-González JM, Benito-León J; Madrid Demyelinating Diseases Group (GEDMA) (2003) Informal caregiving in multiple sclerosis patients: data from the Madrid demyelinating disease group study. Disabil Rehabil 25:1057–1064
Ascherio A, Munger K (2008) Epidemiology of multiple sclerosis: from risk factors to prevention. Semin Neurol 28:17–28
Gilden DH (2005) Infectious causes of multiple sclerosis. Lancet Neurol 4:195–202
Contini C, Seraceni S, Cultrera R, Castellazzi M, Granieri E, Fainardi E (2008) Molecular detection of Parachlamydia-like organisms in cerebrospinal fluid of patients with multiple sclerosis. Mult Scler 14:564–566
Ascherio A (2008) Epstein-Barr virus in the development of multiple sclerosis. Expert Rev Neurother 8:331–333
Butcher J (1976) The distribution of multiple sclerosis in relation to the dairy industry and milk consumption. NZ Med J 83:427–430
Malosse D, Perron H, Sasco A, Seigneurin JM (1992) Correlation between milk and dairy product consumption and multiple sclerosis prevalence: a worldwide study. Neuroepidemiology 11:304–312
Lagneau PE, Lebtahi K, Swinne D (1996) Isolation of yeasts from bovine milk in Belgium. Mycopathologia 135:99–102
Gass JD (1993) Acute zonal occult outer retinopathy. Donders lecture: the Netherlands Ophthalmological Society, Maastricht, Holland, June 19, 1992. J Clin Neuroophthalmol 13:79–97
Gass JD, Agarwal A, Scott IU (2002) Acute zonal occult outer retinopathy: a long-term follow-up study. Am J Ophthalmol 134:329–339
Hintzen RQ, van den Born LI (2006) Acute zonal occult outer retinopathy and multiple sclerosis. J Neurol Neurosurg Psychiatry 77:1373–1375
Carrasco L, Ramos M, Galisteo R, Pisa D, Fresno M, González ME (2005) Isolation of Candida famata from a patient with acute zonal occult outer retinopathy. J Clin Microbiol 43:635–640
Pisa D, Ramos M, Molina S, García P, Carrasco L (2007) Evolution of antibody response and fungal antigens in the serum of a patient infected with Candida famata. J Med Microbiol 56:571–578
Pisa D, Ramos M, García P, Escoto R, Barraquer R, Molina S, Carrasco L (2008) Fungal infection in patients with serpiginous choroiditis or acute zonal occult outer retinopathy. J Clin Microbiol 46:130–135
Trick WE, Fridkin SK, Edwards JR, Hajjeh RA, Gaynes RP, Hospitals National Nosocomial Infections Surveillance System (2002) Secular trend of hospital-acquired candidemia among intensive care unit patients in the United States during 1989-1999. Clin Infect Dis 35:627–630
Pfaller MA, Diekema DJ (2002) Role of sentinel surveillance of candidemia: trends in species distribution and antifungal susceptibility. J Clin Microbiol 40:3551–3557
Lipton SA, Hickey WF, Morris JH, Loscalzo J (1984) Candidal infection in the central nervous system. Am J Med 76:101–108
Laín A, Elguezabal N, Moragues MD, García-Ruiz JC, del Palacio A, Pontón J (2008) Contribution of serum biomarkers to the diagnosis of invasive candidiasis. Expert Rev Mol Diagn 8:315–325
McDonald WI, Compston A, Edan G, Goodkin D, Hartung HP, Lublin FD et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the international panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Ellepola AN, Morrison CJ (2005) Laboratory diagnosis of invasive candidiasis. J Microbiol 43:65–84
Southern P, Horbul J, Maher D, Davis DA (2008) C. albicans colonization of human mucosal surfaces. PLoS ONE 3:e2067
Yang YL (1993) Virulence factors of Candida species. J Microbiol Immunol Infect 36:223–228
Kurtzke JF (1993) Epidemiologic evidence for multiple sclerosis as an infection. Clin Microbiol Rev 6:382–427
Belgi G, Friedmann PS (2002) Traditional therapies: glucocorticoids, azathioprine, methotrexate, hydroxyurea. Clin Exp Dermatol 27:546–554
Acknowledgments
We wish to express our sincere thanks to Dr. Nancy Grajales Hernández and Gerardo García Perales, as well as other members of the Multiple Sclerosis association of Madrid (Spain). We also thank Drs. Mari Luz Borbolla and Manuel Algora (Transfusion Center of the Community of Madrid) for providing the control samples. Fátima Palomares is also acknowledged for her involvement during the early stages of this study. Finally, we also acknowledge the contribution provided by Dr. Elan D. Louis who critiqued late stage drafts of the paper. The study was supported by grants from the Fundación de Investigación Médica Mútua Madrileña and Fundación Ramón Areces. Dr. Benito-León is supported by NIH R01 NS024859 from the National Institutes of Health, Bethesda, MD, USA.
Competing interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
JB-L and LC conceived, coordinated and designed the original study. JB-L conducted the analysis of data and drafted the manuscript. JBL, DP, RA, PC, and MDS collected data and samples. DP and RA conducted the serological testing. All authors offered critical input into the manuscript and all have read and approved the final version.
Rights and permissions
About this article
Cite this article
Benito-León, J., Pisa, D., Alonso, R. et al. Association between multiple sclerosis and Candida species: evidence from a case-control study. Eur J Clin Microbiol Infect Dis 29, 1139–1145 (2010). https://doi.org/10.1007/s10096-010-0979-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-010-0979-y