Abstract
Objective
Hemifacial spasm (HFS) is a movement disorder characterized by involuntary muscle contractions on one side of the face. It is associated with disturbances in the brain’s functional architecture. Despite this, the structural alterations in the brain related to HFS remain poorly understood. In this study, we investigated the cortical morphology changes in patients with HFS compared to healthy controls (HCs).
Methods
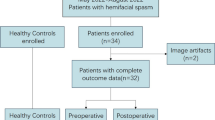
We analyzed 3D T1-weighted MRI images from 33 patients with left-sided primary HFS and 33 age- and sex-matched HCs. Measurements of cortical thickness (CTh), sulcal depth, local gyrification index (lGI), and fractal dimension were taken using a computational anatomy toolbox. A general linear model, accounting for age, gender, and total brain volume, was applied for statistical analyses. Significant clusters were then assessed for correlations with clinical parameters.
Results
The HFS patients displayed several cortical abnormalities when compared to HCs, including reduced CTh in the contralateral precentral gyrus and left orbitofrontal cortex, decreased sulcal depth in the left orbitofrontal cortex, and increased lGI in the right insula and superior temporal cortex. However, fractal dimension did not differ significantly between the groups. Additionally, in HFS patients, a notable negative correlation was found between the sulcal depth in the left orbitofrontal cortex and the Beck Depression Inventory-II scores.
Conclusions
Our findings reveal that HFS is associated with specific surface-based morphological changes in the brain. These alterations contribute to a deeper understanding of the neurophysiological mechanisms involved in HFS and may have implications for future research and treatment strategies.




Similar content being viewed by others
Data availability
Data are available upon reasonable requests of collaboration.
Code availability
None.
References
Jannetta PJ (1998) Typical or atypical hemifacial spasm. J Neurosurg 89:346–347
Jo KW, Kong DS, Park K (2013) Microvascular decompression for hemifacial spasm: long-term outcome and prognostic factors, with emphasis on delayed cure. Neurosurg Rev 36:297–301 (discussion 301–292)
Tan EK, Chan LL (2006) Young onset hemifacial spasm. Acta Neurol Scand 114:59–62
McLaughlin MR, Jannetta PJ, Clyde BL, Subach BR, Comey CH, Resnick DK (1999) Microvascular decompression of cranial nerves: lessons learned after 4400 operations. J Neurosurg 90:1–8
Gardner WJ (1962) Concerning the mechanism of trigeminal neuralgia and hemifacial spasm. J Neurosurg 19:947–958
Miller LE, Miller VM (2012) Safety and effectiveness of microvascular decompression for treatment of hemifacial spasm: a systematic review. Br J Neurosurg 26:438–444
Eksantivongs S, Poungvarin N, Viriyavejakul A, Punyamee J (1994) Hemifacial spasm: an electrophysiological evidence of facial motorneurons hyperexcitability. J Med Assoc Thai 77:627–632
Barker FG 2nd, Jannetta PJ, Bissonette DJ, Shields PT, Larkins MV, Jho HD (1995) Microvascular decompression for hemifacial spasm. J Neurosurg 82:201–210
Kakizawa Y, Seguchi T, Kodama K, Ogiwara T, Sasaki T, Goto T, Hongo K (2008) Anatomical study of the trigeminal and facial cranial nerves with the aid of 3.0-tesla magnetic resonance imaging. J Neurosurg 108:483–490
Wang A, Jankovic J (1998) Hemifacial spasm: clinical findings and treatment. Muscle Nerve 21:1740–1747
Bao F, Wang Y, Liu J, Mao C, Ma S, Guo C, Ding H, Zhang M (2015) Structural changes in the CNS of patients with hemifacial spasm. Neuroscience 289:56–62
Guo Y, Peng K, Liu Y, Zhong L, Dang C, Yan Z, Wang Y, Zeng J, Zhang W, Ou Z, Liu G (2021) Topological alterations in white matter structural networks in blepharospasm. Mov Disord 36:2802–2810
Tu Y, Yu T, Wei Y, Sun K, Zhao W, Yu B (2016) Structural brain alterations in hemifacial spasm: a voxel-based morphometry and diffusion tensor imaging study. Clin Neurophysiol 127:1470–1474
Peelle JE, Cusack R, Henson RN (2012) Adjusting for global effects in voxel-based morphometry: gray matter decline in normal aging. Neuroimage 60:1503–1516
Henley SM, Ridgway GR, Scahill RI, Kloppel S, Tabrizi SJ, Fox NC, Kassubek J (2010) Group EIW Pitfalls in the use of voxel-based morphometry as a biomarker: examples from huntington disease. AJNR Am J Neuroradiol 31:711–719
Voets NL, Hough MG, Douaud G, Matthews PM, James A, Winmill L, Webster P, Smith S (2008) Evidence for abnormalities of cortical development in adolescent-onset schizophrenia. Neuroimage 43:665–675
Hutton C, Draganski B, Ashburner J, Weiskopf N (2009) A comparison between voxel-based cortical thickness and voxel-based morphometry in normal aging. Neuroimage 48:371–380
Palaniyappan L, Liddle PF (2012) Differential effects of surface area, gyrification and cortical thickness on voxel based morphometric deficits in schizophrenia. Neuroimage 60:693–699
Luders E, Thompson PM, Narr KL, Toga AW, Jancke L, Gaser C (2006) A curvature-based approach to estimate local gyrification on the cortical surface. Neuroimage 29:1224–1230
Armstrong E, Schleicher A, Omran H, Curtis M, Zilles K (1995) The ontogeny of human gyrification. Cereb Cortex 5:56–63
Im K, Lee JM, Yoon U, Shin YW, Hong SB, Kim IY, Kwon JS, Kim SI (2006) Fractal dimension in human cortical surface: multiple regression analysis with cortical thickness, sulcal depth, and folding area. Hum Brain Mapp 27:994–1003
Mitchell T, Lehericy S, Chiu SY, Strafella AP, Stoessl AJ, Vaillancourt DE (2021) Emerging neuroimaging biomarkers across disease stage in Parkinson disease: a review. JAMA Neurol 78:1262–1272
Sampedro F, Martinez-Horta S, Perez-Perez J, Horta-Barba A, Lopez-Mora DA, Camacho V, Fernandez-Leon A, Gomez-Anson B, Carrio I, Kulisevsky J (2019) Cortical atrophic-hypometabolic dissociation in the transition from premanifest to early-stage Huntington’s disease. Eur J Nucl Med Mol Imaging 46:1111–1116
Tomic A, Agosta F, Sarasso E, Svetel M, Kresojevic N, Fontana A, Canu E, Petrovic I, Kostic VS, Filippi M (2021) Brain structural changes in focal dystonia-what about task specificity? A multimodal MRI study. Mov Disord 36:196–205
Pietracupa S, Bologna M, Bharti K, Pasqua G, Tommasin S, Elifani F, Paparella G, Petsas N, Grillea G, Berardelli A, Pantano P (2019) White matter rather than gray matter damage characterizes essential tremor. Eur Radiol 29:6634–6642
Xu H, Guo C, Li H, Gao L, Zhang M, Wang Y (2019) Structural and functional amygdala abnormalities in hemifacial spasm. Front Neurol 10:393
Albanese A, Bhatia K, Bressman SB, Delong MR, Fahn S, Fung VS, Hallett M, Jankovic J, Jinnah HA, Klein C, Lang AE, Mink JW, Teller JK (2013) Phenomenology and classification of dystonia: a consensus update. Mov Disord 28:863–873
Jankovic J, Kenney C, Grafe S, Goertelmeyer R, Comes G (2009) Relationship between various clinical outcome assessments in patients with blepharospasm. Mov Disord 24:407–413
Yotter RA, Nenadic I, Ziegler G, Thompson PM, Gaser C (2011) Local cortical surface complexity maps from spherical harmonic reconstructions. Neuroimage 56:961–973
Lerner A, Bagic A, Hanakawa T, Boudreau EA, Pagan F, Mari Z, Bara-Jimenez W, Aksu M, Sato S, Murphy DL, Hallett M (2009) Involvement of insula and cingulate cortices in control and suppression of natural urges. Cereb Cortex 19:218–223
Morecraft RJ, Stilwell-Morecraft KS, Rossing WR (2004) The motor cortex and facial expression: new insights from neuroscience. Neurologist 10:235–249
Cattaneo L, Pavesi G (2014) The facial motor system. Neurosci Biobehav Rev 38:135–159
Chung JY, Yoon HW, Song MS, Park H (2006) Event related fMRI studies of voluntary and inhibited eye blinking using a time marker of EOG. Neurosci Lett 395:196–200
Setthawatcharawanich S, Sathirapanya P, Limapichat K, Phabphal K (2011) Factors associated with quality of life in hemifacial spasm and blepharospasm during long-term treatment with botulinum toxin. Qual Life Res 20:1519–1523
Davidson RJ, Putnam KM, Larson CL (2000) Dysfunction in the neural circuitry of emotion regulation–a possible prelude to violence. Science 289:591–594
Rolls ET (2021) The neuroscience of emotional disorders. Handb Clin Neurol 183:1–26
Drevets WC (2007) Orbitofrontal cortex function and structure in depression. Ann N Y Acad Sci 1121:499–527
Berman BD, Horovitz SG, Morel B, Hallett M (2012) Neural correlates of blink suppression and the buildup of a natural bodily urge. Neuroimage 59:1441–1450
Rousseau C, Fautrelle L, Papaxanthis C, Fadiga L, Pozzo T, White O (2016) Direction-dependent activation of the insular cortex during vertical and horizontal hand movements. Neuroscience 325:10–19
Acknowledgements
We thank all the participants in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This research was approved by the Ethics Committee of Ruijin Hospital, Shanghai Jiaotong University School of Medicine.
Consent to participate
Informed consent was obtained from all participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Z., Xiong, F., Gao, F. et al. Cortical changes in the brain of patients with hemifacial spasm. Neurol Sci (2024). https://doi.org/10.1007/s10072-024-07353-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10072-024-07353-7