Abstract
Aim
Study of intraepidermal nerve fiber density (IENFD) by skin biopsy represents a promising tool in the evaluation of patients with ATTRv polyneuropathy (ATTRv-PN). Herein, we retrospectively analyze intraepidermal innervation and quantitative sensory test (QST) data from an Italian cohort of Italian ATTRv-PN patients and asymptomatic carriers aimed to provide insights into early nerve pathological and functional changes in this disease.
Methods
IENFD and QST data of 14 ATTRv-PN patients and 14 asymptomatic carriers were retrospectively analyzed together with clinical and paraclinical data such as disease stage and severity, neuropathic pain scales, and sural SNAP amplitude.
Results
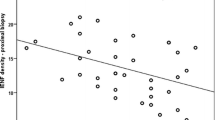
Given an estimated time to the predicted age of onset of symptomatic disease of 20.27 + / − 7.9 years, small nerve fiber loss seems to be unexpectedly early in carriers. Moreover, carriers showed skin denervation at the proximal (thigh) site, suggesting a non-length-dependent neuropathic process. IENFD at ankle correlated with disease severity and other paraclinical variables such as sural nerve potential amplitude and QST parameters. Patients at earlier stages of the disease did not show significant differences in ankle IENFD compared with asymptomatic carriers, but significant differences in terms of QST parameters, small fiber neuropathy symptoms, and neuropathic pain.
Conclusions
Skin biopsy can disclose an early non-length-dependent small fiber loss in ATTRv-PN and, together with QST, could provide a useful insight disease onset and progression.

Similar content being viewed by others
References
Benson MD, Buxbaum JN, Eisenberg DS, Merlini G, Saraiva MJM, Sekijima Y, Sipe JD, Westermark P (2018) Amyloid nomenclature 2018: recommendations by the International Society of Amyloidosis (ISA) nomenclature committee. Amyloid 25:215–219
Adams D, Coelho T, Obici L et al (2015) Rapid progression of familial amyloidotic polyneuropathy: a multinational natural history study. Neurology 85:675–682
Coutinho P, Martins da Silva A, Lopes Lima J, Resende Barbosa A (1980) Forty years of experience with type I amyloid neuropathy (review of 483 cases). In: Glenner GG, Pinho e Costa P, Falcão de Freitas (eds) Amyloid and amyloidosis. Excerpta Medica, Amsterdam
Yang NC, Lee MJ, Chao CC et al (2010) Clinical presentations and skin denervation in amyloid neuropathy due to transthyretin Ala97Ser. Neurology 75:532–538
Masuda T, Ueda M, Suenaga G (2017) Early skin denervation in hereditary and iatrogenic transthyretin amyloid neuropathy. Neurology 88(23):2192–2197
Ebenezer GJ et al (2017) Cutaneous nerve biomarkers in transthyretin familial amyloid polyneuropathy. Ann Neurol 82:44–56
Chao CC, Hsueh HW, Kan HW et al (2019) Skin nerve pathology: biomarkers of premanifest and manifest amyloid neuropathy. Ann Neurol 85(4):560–573
Lauria G, Hsieh ST, Johansson O, Kennedy WR et al (2010) European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society. Eur J Neurol 17(7):903–12 (e44-9)
Provitera V, Gibbons CH, Wendelschafer-Crabb G et al (2016) A multi-center, multinational age- and gender-adjusted normative dataset for immunofluorescent intraepidermal nerve fiber density at the distal leg. Eur J Neurol 23(2):333–338
Provitera V, Gibbons CH, Wendelschafer-Crabb G, Donadio V, Vitale DF, Loavenbruck A, Stancanelli A, Caporaso G, Liguori R, Wang N, Santoro L, Kennedy WR, Nolano M (2018) The role of skin biopsy in differentiating small-fiber neuropathy from ganglionopathy. Eur J Neurol 25(6):848–853
Vollert J, Attal N, Baron R, Freynhagen R, Haanpää M, Hansson P, Jensen TS, Rice AS, Segerdahl M, Serra J, Sindrup SH, Tölle TR, Treede RD, Maier C (2016) Quantitative sensory testing using DFNS protocol in Europe: an evaluation of heterogeneity across multiple centers in patients with peripheral neuropathic pain and healthy subjects. Pain 157(3):750–758
Berk JL, Suhr OB, Obici L et al (2013) Repurposing diflunisal for familial amyloid polyneuropathy: a randomized clinical trial. JAMA 310:2658–2667
Dyck PJ, Sherman WR, Hallcher LM, Service FJ, O’Brien PC, Grina LA, Palumbo PJ, Swanson CJ (1980) Human diabetic endoneurial sorbitol, fructose, and myo-inositol related to sural nerve morphometry. Ann Neurol 8(6):590–596
Spallone V, Morganti R, D’Amato C, Greco C, Cacciotti L, Marfia GA (2012) Validation of DN4 as a screening tool for neuropathic pain in painful diabetic polyneuropathy. Diabet Med 29(5):578–585
Bakkers M, Merkies IS, Lauria G, Devigili G, Penza P, Lombardi R, Hermans MC, van Nes SI, De Baets M, Faber CG (2009) Intraepidermal nerve fiber density and its application in sarcoidosis. Neurology 73(14):1142–1148
England JD, Gronseth GS, Franklin G, Carter GT, Kinsella LJ, Cohen JA, Asbury AK, Szigeti K, Lupski JR, Latov N, Lewis RA, Low PA, Fisher MA, Herrmann DN, Howard JF Jr, Lauria G, Miller RG, Polydefkis M, Sumner AJ (2009) American Academy of Neurology. Practice Parameter: evaluation of distal symmetric polyneuropathy: role of laboratory and genetic testing (an evidence-based review). Report of the American Academy of Neurology, American Association of Neuromuscular and Electrodiagnostic Medicine, and American Academy of Physical Medicine and Rehabilitation. Neurology 72(2):185–92
Conceição I, Damy T, Romero M, Galán L, Attarian S, Luigetti M, Sadeh M, Sarafov S, Tournev I, Ueda M (2019) Early diagnosis of ATTR amyloidosis through targeted follow-up of identified carriers of TTR gene mutations. Amyloid 26(1):3–9
Adams D, Koike H, Slama M, Coelho T (2019) Hereditary transthyretin amyloidosis: a model of medical progress for a fatal disease. Nat Rev Neurol 15(7):387–404
Kapoor M, Foiani M, Heslegrave A, Zetterberg H, Lunn MP, Malaspina A, Gillmore JD, Rossor AM, Reilly MM (2019) Plasma neurofilament light chain concentration is increased and correlates with the severity of neuropathy in hereditary transthyretin amyloidosis. J Peripher Nerv Syst 24(4):314–319
Hanyu N, Ikeda S, Nakadai A, Yanagisawa N, Powell HC (1989) Peripheral nerve pathological findings in familial amyloid polyneuropathy: a correlative study of proximal sciatic nerve and sural nerve lesions. Ann Neurol 25(4):340–350
Fujitake J, Mizuta H, Fujii H, Ishikawa Y, Katsuyama E, Takasu K, Saida K, Tatsuoka Y (2003) Late-onset familial amyloid polyneuropathy: an autopsy study of two Japanese brothers. Amyloid 10(3):198–205
Granata G, Luigetti M, Coraci D et al (2014) Ultrasound evaluation in transthyretin-related amyloid neuropathy. Muscle Nerve 50(3):372–376
Kollmer J, Hund E, Hornung B, Hegenbart U, Schonland SO, Kimmich C et al (2015) In vivo detection of nerve injury in familial amyloid polyneuropathy by magnetic resonance neurography. Brain 138:549–562
Podnar S, Sarafov S, Tournev I, Omejec G, Zidar J (2017) Peripheral nerve ultrasonography in patients with transthyretin amyloidosis. Clin Neurophysiol 128:505–511
Salvalaggio A, Coraci D, Cacciavillani M, Obici L, Mazzeo A, Luigetti M, Pastorelli F, Grandis M, Cavallaro T, Bisogni G, Lozza A, Gemelli C, Gentile L, Ermani M, Fabrizi GM, Plasmati R, Campagnolo M, Castellani F, Gasparotti R, Martinoli C, Padua L, Briani C (2020) Nerve ultrasound in hereditary transthyretin amyloidosis: red flags and possible progression biomarkers. J Neurol 268(1):189–198
Russo M, Obici L, Bartolomei I, Cappelli F, Luigetti M, Fenu S, Cavallaro T, Chiappini MG, Gemelli C, Pradotto LG, Manganelli F, Leonardi L, My F, Sampaolo S, Briani C, Gentile L, Stancanelli C, Di Buduo E, Pacciolla P, Salvi F, Casagrande S, Bisogni G, Calabrese D, Vanoli F, Di Iorio G, Antonini G, Santoro L, Mauro A, Grandis M, Di Girolamo M, Fabrizi GM, Pareyson D, Sabatelli M, Perfetto F, Rapezzi C, Merlini G, Mazzeo A, Vita G (2020) ATTRv amyloidosis Italian Registry: clinical and epidemiological data. Amyloid 27(4):259–265
Luigetti M, Romozzi M, Bisogni G, Cardellini D, Cavallaro T, Di Paolantonio A, Fabrizi GM, Fenu S, Gentile L, Grandis M, Marucci G, Massucco S, Mazzeo A, Pareyson D, Romano A, Russo M, Schenone A, Tagliapietra M, Tozza S, Vita G, Sabatelli M (2020) hATTR Pathology: Nerve Biopsy Results from Italian Referral Centers. Brain Sci 10(11):E780
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
We confirm that we have read the journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Competing interests
Any of the authors have conflict of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leonardi, L., Galosi, E., Vanoli, F. et al. Skin biopsy and quantitative sensory assessment in an Italian cohort of ATTRv patients with polyneuropathy and asymptomatic carriers: possible evidence of early non-length dependent denervation. Neurol Sci 43, 1359–1364 (2022). https://doi.org/10.1007/s10072-021-05434-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-021-05434-5